Oragenics Co-Authors a Publication Describing the Dual Mechanism of Action of Lantibiotics
December 08 2021 - 8:00AM
Business Wire
Molecular modeling offers additional insights
enabling the design and development of novel antibiotics.
Oragenics, Inc. (NYSE American: OGEN) (“Oragenics” or the
“Company”) in collaboration with researchers from Florida
International University’s Department of Physics and Biomolecular
Sciences Institute, today announced the publication of a study
entitled “Lipid II Binding and Transmembrane Properties of Various
Antimicrobial Lanthipeptides.” The study was published in the
Journal of Chemical Theory and Computation
(https://doi.org/10.1021/acs.jctc.1c00666). The alarming rise in
antibiotic resistant infections in recent years and the dire need
for the development of new antimicrobials is globally recognized.
Oragenics’ lantibiotics are promising candidates to fight
antibiotic-resistant bacteria due to their unique and dual mode of
action, including interference with cell wall synthesis by binding
to Lipid II and creation of unique pores in bacterial membranes. In
this publication, atomic-scale molecular dynamics computational
studies were used to compare both the Lipid II binding ability and
the membrane interactions of several lanthipeptides, demonstrating
the unique properties of Oragenics’ lantibiotics and unveiling a
rational path to improve the therapeutic profile of our
compounds.
“We continue to expand our lantibiotic pipeline in order to
create new ways of fighting antibiotic-resistant infections,”
stated Frederick W. Telling, Ph.D., Executive Chairman of
Oragenics. Dr. Telling added, “This publication provides us with
additional insight on how to exploit the unique properties of our
compounds, which we hope may ultimately result in the development
of drugs with a broader spectrum of activity that include
Gram-negative infections.”
About Oragenics, Inc.
Oragenics, Inc. is a development-stage company dedicated to
fighting infectious diseases including coronaviruses and
multidrug-resistant organisms. Its lead product is Terra CoV-2, a
vaccine candidate to prevent COVID-19 and variants of the
SARS-CoV-2 virus. The Terra CoV-2 program leverages coronavirus
spike protein research licensed from the NIH and the National
Research Council of Canada NRC with a focus on addressing
supply-chain challenges, and offering more patient-friendly
administration, such as intranasal. Its lantibiotics program
features a novel class of antibiotics against infectious diseases
that have developed resistance to commercial antibiotics.
Forward-Looking Statements
This communication contains “forward-looking statements” within
the meaning of the safe harbor provisions of the U.S. Private
Securities Litigation Reform Act of 1995. These forward-looking
statements are based on management’s beliefs and assumptions and
information currently available. The words "believe," "expect,"
"anticipate," "intend," "estimate," "project" and similar
expressions that do not relate solely to historical matters
identify forward-looking statements. Investors should be cautious
in relying on forward-looking statements because they are subject
to a variety of risks, uncertainties, and other factors that could
cause actual results to differ materially from those expressed in
any such forward-looking statements. These factors include, but are
not limited to, the following: the Company’s ability to advance the
development of Terra CoV-2 and lantibiotics under the timelines and
in accord with the milestones it projects; the Company’s ability to
obtain funding, non-dilutive or otherwise, for the development of
the vaccine product candidate, Terra CoV-2 and our lantibiotics,
whether through its own cash on hand, or another alternative
source; the regulatory application process, research and
development stages, and future clinical data and analysis relating
to Terra CoV-2 and lantibiotics, including any meetings, decisions
by regulatory authorities, such as the FDA and investigational
review boards, whether favorable or unfavorable; the potential
application of Terra CoV-2 to variants and other coronaviruses; the
Company’s ability to obtain, maintain and enforce necessary patent
and other intellectual property protection; the nature of
competition and development relating to COVID-19 immunization and
therapeutic treatments and demand for vaccines and antibiotics; the
Company’s expectations as to administration, manufacturing, storage
and distribution; other potential adverse impacts due to the global
COVID-19 pandemic, such as delays in regulatory review,
interruptions to manufacturers and supply chains, adverse impacts
on healthcare systems and disruption of the global economy; and
general economic and market conditions and risks, as well as other
uncertainties described in our filings with the U.S. Securities and
Exchange Commission. All information set forth in this press
release is as of the date hereof. You should consider these factors
in evaluating the forward-looking statements included in this press
release and not place undue reliance on such statements. We do not
assume any obligation to publicly provide revisions or updates to
any forward-looking statements, whether as a result of new
information, future developments or otherwise, should circumstances
change, except as otherwise required by law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20211208005417/en/
Oragenics, Inc. Michael Sullivan, Chief Financial Officer
813-286-7900 msullivan@oragenics.com or LHA Investor
Relations Kim Golodetz 212-838-3777 kgolodetz@lhai.com
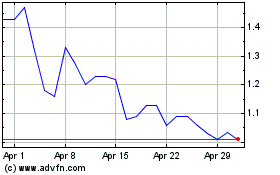
Oragenics (AMEX:OGEN)
Historical Stock Chart
From Mar 2024 to Apr 2024
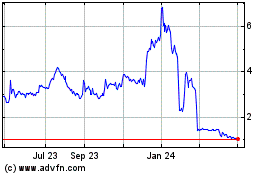
Oragenics (AMEX:OGEN)
Historical Stock Chart
From Apr 2023 to Apr 2024