rms Company Scales Medical Device Production with Addition of 3D Systems’ DMP Flex 350 Dual
January 19 2023 - 8:30AM

Today, 3D Systems (NYSE:DDD) announced rms Company, one of the
largest contract manufacturers of high-quality, tight-tolerance
medical devices in the world, has added the DMP Flex 350 Dual to
its production workflow. rms has more than 30 3D Systems’ DMP 3D
printers in its additive manufacturing facility enabling the
company to provide a complete Powder to Package capability to
medical device OEMs. Powder to Package encompasses the entire
manufacturing and post-processing workflow required to go from a
digital file to a sterilized and packaged final product ready for
the surgical suite. With the addition of the DMP Flex 350 Dual, rms
believes they will be able to expand the types of implants they
produce and accelerate throughput — helping to address the evolving
application needs of the industry.
Over the course of four years, 3D Systems has
helped rms expand its portfolio of direct metal printers from one
machine to more than 30 — backed by a process that ensures
repeatability across machines and supply chains — enabling true
scaled production. As a result, rms has been able to cement its
reputation for additively manufacturing high-quality titanium
implants. With the addition of the DMP Flex 350 Dual which enables
up to 50% productivity increases, rms will be able to increase
throughput for smaller implants including those intended for spinal
applications. They believe the same productivity increases will
also open new opportunities in the production of large joint and
extremity implants while allowing flexibility for existing
projects.
“The increase in efficiency we get from the DMP
Flex 350 Dual system creates new opportunities for larger and
taller parts which were economically challenging with a single
laser system,” said Ryan Kircher, senior additive manufacturing
engineer, rms Company. “Another key reason rms Company decided to
invest in a DMP Flex 350 Dual is that it is engineered in a way
that allows us to produce the high precision, high-quality products
we expect from our current installation of DMP systems without
changing the processing parameters we validated when we initially
developed our AM capabilities. This will allow us to explore not
only new products but also reduce production times and costs on the
products we are already making today.”
“The rms additive division owes a large part of our
success to our customers,” said Troy Olson, director of operations
– additive manufacturing division, rms Company. “Over the past
several years, our customers have come to expect a continual flow
of high-quality, additively manufactured medical devices. Our
Powder to Package solution provides a complete end-to-end value
proposition that allows us to control the entire manufacturing and
packaging process flow. A key element of our process flow is the
reliability and performance we get from our stable of 30-plus 3D
Systems’ DMP ProX® 320 and DMP Flex 350 machines. Uptime on these
machines is crucial for us to maintain our customer commitments.
Our printers operate on a 24/7 production cadence, which leaves
little room for downtime. 3D Systems has taken these first-in-class
single laser platforms and have expanded on that technology with
the DMP Flex 350 Dual. During our validation process, the DMP Flex
350 Dual showed no measurable differences in mechanical properties
or dimensional accuracy. We are excited to add this new capability
to our existing printing operation. As we continue to scale our
additive manufacturing business, the 3D Systems DMP Flex 350 Dual
will be at the forefront of our growth strategy.”
The DMP Flex 350 Dual is the most recent addition
to 3D Systems’ Direct Metal Printing (DMP) portfolio. This
dual-laser configuration maintains the benefits of the single-laser
configuration including flexible application use and quick-swap
build modules, and a central server to manage print jobs,
materials, settings, and maintenance for 24/7 productivity.
Additionally, the company’s unique vacuum chamber significantly
reduces argon gas consumption while delivering best-in-class oxygen
purity (<25 ppm). The printer also includes Oqton’s 3DXpert —
the all-in-one software for industrial additive manufacturing that
enables efficient preparation, optimization, lattice generation,
and 3D printing of high-quality parts by streamlining the workflow
from design to printing.
“Our Application Innovation Group worked alongside
the rms team to develop, characterize and validate the DMP Flex 350
Dual as a seamless solution within their production environment,”
said Jeph Ruppert, director, application innovation group, 3D
Systems. “Our Direct Metal Printing (DMP) platform is recognized as
an industry-leading technology for its ability to produce
exceptionally high feature resolution parts of high chemical purity
which makes it ideal for medical devices. Our collective
application and technology expertise combined with the unparalleled
capabilities of the DMP Flex 350 Dual is helping drive innovation
for rms. They’ve established themselves as a world-class medical
device manufacturer, and we are proud to be part of that
journey.”
“Our collaboration with rms has spanned several
years, and during that time rms has grown at a rate and scale that
is truly impressive,” said Menno Ellis, executive vice president,
healthcare solutions, 3D Systems. “They have not only delivered
excellent solutions to the medical device community but have also
grown their in-house additive knowledge, helping guide the industry
to a more mature and robust position. Today, rms is expanding upon
its capabilities and capacity with the addition of the DMP Flex 350
Dual. I look forward to seeing how this will enhance the breadth of
applications they will be able to address for their customers, and
the impact it will have for growth.”
Forward-Looking StatementsCertain
statements made in this release that are not statements of
historical or current facts are forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of
1995. Forward-looking statements involve known and unknown risks,
uncertainties and other factors that may cause the actual results,
performance or achievements of the company to be materially
different from historical results or from any future results or
projections expressed or implied by such forward-looking
statements. In many cases, forward-looking statements can be
identified by terms such as "believes," "belief," "expects," "may,"
"will," "estimates," "intends," "anticipates" or "plans" or the
negative of these terms or other comparable terminology.
Forward-looking statements are based upon management’s beliefs,
assumptions, and current expectations and may include comments as
to the company’s beliefs and expectations as to future events and
trends affecting its business and are necessarily subject to
uncertainties, many of which are outside the control of the
company. The factors described under the headings "Forward-Looking
Statements" and "Risk Factors" in the company’s periodic filings
with the Securities and Exchange Commission, as well as other
factors, could cause actual results to differ materially from those
reflected or predicted in forward-looking statements. Although
management believes that the expectations reflected in the
forward-looking statements are reasonable, forward-looking
statements are not, and should not be relied upon as a guarantee of
future performance or results, nor will they necessarily prove to
be accurate indications of the times at which such performance or
results will be achieved. The forward-looking statements included
are made only as of the date of the statement. 3D Systems
undertakes no obligation to update or revise any forward-looking
statements made by management or on its behalf, whether as a result
of future developments, subsequent events or circumstances or
otherwise, except as required by law.
About 3D Systems More than 35
years ago, 3D Systems brought the innovation of 3D printing to the
manufacturing industry. Today, as the leading additive
manufacturing solutions partner, we bring innovation, performance,
and reliability to every interaction - empowering our customers to
create products and business models never before possible. Thanks
to our unique offering of hardware, software, materials, and
services, each application-specific solution is powered by the
expertise of our application engineers who collaborate with
customers to transform how they deliver their products and
services. 3D Systems’ solutions address a variety of advanced
applications in healthcare and industrial markets such as medical
and dental, aerospace & defense, automotive, and durable goods.
More information on the company is available at
www.3dsystems.com.
A photo accompanying this announcement is available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/6667e96a-3bb8-474e-9da3-40171d898fcf
Investor Contact:
investor.relations@3dsystems.com
Media Contact:
press@3dsystems.com
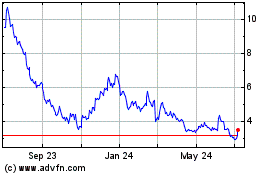
3D Systems (NYSE:DDD)
Historical Stock Chart
From Mar 2024 to Apr 2024
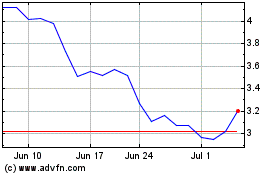
3D Systems (NYSE:DDD)
Historical Stock Chart
From Apr 2023 to Apr 2024