TIDMRENX
RNS Number : 0685G
Renalytix PLC
22 July 2021
Renalytix plc
("Renalytix" or the "Company")
Renalytix Appoints Joseph Hutson Vice President of Global
Quality and Regulatory
Hutson To Build-on Renalytix Quality and Regulatory Strategy for
KidneyIntelX Global Commercialization
NEW YORK and SALT LAKE CITY, July 22, 2021 - Renalytix Plc
(NASDAQ: RNLX) (LSE: RENX ) today announced the appointment of
Joseph Hutson as its Vice President of Quality and Regulatory.
Hutson brings 23 years of experience in the life sciences industry,
including quality and regulatory leadership roles at Cardinal
Health, CareFusion, Becton Dickinson and Abbott Diagnostics. He
will lead teams focused on assuring compliance with United States
and international Quality Management System regulations and
standards and driving efficient approval for a series of products
under the KidneyIntelX brand through review by the appropriate
regulatory authorities including the United States Food and Drug
Administration (FDA) and the European Medicines Agency (EMA), and
other country specific regulatory bodies.
The KidneyIntelX commercial program is being designed to CLIA,
CAP, ISO, FDA, and In Vitro Diagnostics Regulation (IVDR)
regulations and international standards which will support global
market access for current and future KidneyIntelX innovative
products and services. KidneyIntelX is currently under FDA review
for De Novo marketing authorization under Breakthrough Device
designation. Hutson will contribute his knowledge and expertise to
the efficient FDA review of this submission.
"The dynamic quality and regulatory environments in the United
States, Europe and across the world are now a critical part of
building competitive differentiation and gaining access to global
markets. Without a robust Quality Management System and
well-defined regulatory strategies for product development,
approval, and marketing, it is impossible to remain product
competitive," said Hutson. "Particularly in an area as large and
consequential to global health-care systems as kidney disease,
implementing processes that can meet or exceed FDA regulations and
IVDR's standards of quality and safety will be the requirement to
set a new level of care for early-stage prognosis and
treatment."
Most recently, at Abbott Molecular Diagnostics, Hutson led
Quality, Global and Clinical Compliance and he played an integral
role in the rapid development, FDA Emergency Use Authorizations,
manufacturing scale up and global distribution of 50 million
COVID-19 diagnostic tests throughout the world. Previously, he
served as Worldwide Vice-President, Quality and Regulatory at
Becton Dickinson Respiratory Solutions with responsibility for 31
locations in North and South America, Europe, and Asia and as
Global Vice-President, Quality and Regulatory at CareFusion
acquired by Becton Dickinson in 2015.
For further information, please contact:
Renalytix plc www.renalytix.com
James McCullough, CEO Via Walbrook PR
Stifel (Nominated Adviser, Joint Broker) Tel: 020 7710 7600
Alex Price / Nicholas Moore
Investec Bank plc (Joint Broker) Tel: 020 7597 4000
Gary Clarence / Daniel Adams
Walbrook PR Limited Tel: 020 7933 8780 or renalytix@walbrookpr.com
Paul McManus / Lianne Cawthorne Mob: 07980 541 893 / 07584 391 303
About Kidney Disease
Kidney disease is now recognized as a public health epidemic
affecting over 850 million people globally. The Centers for Disease
Control and Prevention (CDC) estimates that 15% of US adults, or 37
million people, currently have chronic kidney disease (CKD).
Further, the CDC reports that 9 out of 10 adults with
CKD do not know they have it and one out of two people with very
low kidney function who are not on dialysis do not know they have
CKD.(1) Kidney disease is referred to as a "silent killer" because
it often has no symptoms and can go undetected until a very
advanced stage. Each year, kidney disease kills more people than
breast and prostate cancer. Every day, 13 patients in the United
States die while waiting for a kidney transplant.
(1) https://www.cdc.gov/kidneydisease/publications-resources/2019-national-facts.html
About KidneyIntelX
KidneyIntelX, is a first-of-kind, bioprognostic(TM) platform
that employs a proprietary artificial intelligence-enabled
algorithm to combine diverse data inputs, including validated
blood-based biomarkers, inherited genetics, and personalized
patient data from electronic health record, or EHR, systems, to
generate a unique patient risk score. This patient risk score
enables prediction of progressive kidney function decline in
chronic kidney disease, or CKD, allowing physicians and healthcare
systems to optimize the allocation of treatments and clinical
resources to patients at highest risk.
About Renalytix
Renalytix (LSE: RENX) (NASDAQ: RNLX) is a developer of
artificial intelligence-enabled clinical in vitro diagnostic
solutions for kidney disease, one of the most common and costly
chronic medical conditions globally. The Company's lead product is
KidneyIntelX, which has been granted Breakthrough Designation by
the U.S. Food and Drug Administration and which is being designed
to help make significant improvements in kidney disease prognosis,
transplant management, clinical care, patient stratification for
drug clinical trials, and drug target discovery (visit
www.kidneyintelx.com ). For more information, visit
www.renalytix.com .
Forward Looking Statements
Statements contained in this press release regarding matters
that are not historical facts are "forward-looking statements"
within the meaning of the Private Securities Litigation Reform Act
of 1995, as amended. Examples of these forward-looking statements
include statements concerning: the potential for KidneyIntelX to
receive regulatory approval from the FDA, our global market access
strategy for KidneyIntelX, the commercial prospects of
KidneyIntelX, if approved, including whether KidneyIntelX will be
successfully distributed and marketed, our expectations regarding
reimbursement decisions, our plans for expansion of our business
and the ability of KidneyIntelX to curtail costs of chronic and
end-stage kidney disease, optimize care delivery and improve
patient outcomes. Words such as "anticipates," "believes,"
"estimates," "expects," "intends," "plans," "seeks," and similar
expressions are intended to identify forward-looking statements. We
may not actually achieve the plans and objectives disclosed in the
forward-looking statements, and you should not place undue reliance
on our forward-looking statements. Any forward-looking statements
are based on management's current views and assumptions and involve
risks and uncertainties that could cause actual results,
performance or events to differ materially from those expressed or
implied in such statements. These risks and uncertainties include,
among others: that KidneyIntelX is based on novel artificial
intelligence technologies that are rapidly evolving and potential
acceptance, utility and clinical practice remains uncertain; we
have only recently commercially launched KidneyIntelX; and risks
relating to the impact on our business of the COVID-19 pandemic or
similar public health crises. These and other risks are described
more fully in our filings with the Securities and Exchange
Commission (SEC), including the "Risk Factors" section of our
annual report on Form 20-F filed with the SEC on October 28, 2020,
and other filings we make with the SEC from time to time. All
information in this press release is as of the date of the release,
and we undertake no obligation to publicly update any
forward-looking statement, whether as a result of new information,
future events, or otherwise, except as required by law.
This information is provided by RNS, the news service of the
London Stock Exchange. RNS is approved by the Financial Conduct
Authority to act as a Primary Information Provider in the United
Kingdom. Terms and conditions relating to the use and distribution
of this information may apply. For further information, please
contact rns@lseg.com or visit www.rns.com.
RNS may use your IP address to confirm compliance with the terms
and conditions, to analyse how you engage with the information
contained in this communication, and to share such analysis on an
anonymised basis with others as part of our commercial services.
For further information about how RNS and the London Stock Exchange
use the personal data you provide us, please see our Privacy
Policy.
END
MSCUAVORAKUBUUR
(END) Dow Jones Newswires
July 22, 2021 02:00 ET (06:00 GMT)
Renalytix (LSE:RENX)
Historical Stock Chart
From Mar 2024 to Apr 2024
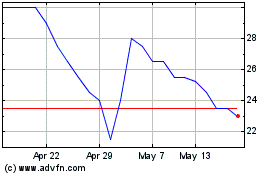
Renalytix (LSE:RENX)
Historical Stock Chart
From Apr 2023 to Apr 2024