NEW
HAVEN, Conn., May 23, 2022
/PRNewswire/ -- Biohaven Pharmaceutical Holding Company Ltd. (NYSE:
BHVN) today announced top-line results from a Phase 3 clinical
trial evaluating the efficacy and safety of its investigational
therapy, troriluzole, in patients with spinocerebellar ataxia
(SCA). The primary endpoint, change from baseline to Week 48 on the
modified functional Scale for the Assessment and Rating of Ataxia
(f-SARA), did not reach statistical significance in the overall SCA
population as there was less than expected disease progression over
the course of the study. In the overall study population (N=213),
the troriluzole and placebo groups each had mean baseline scores of
4.9 on the f-SARA and the two groups showed minimal change at the
48-week endpoint with f-SARA scores of 5.1 and 5.2, respectively
(p=0.76).
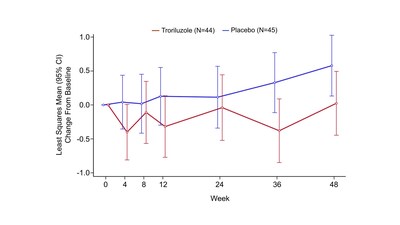
Post hoc analysis of efficacy measures by genotype suggests a
treatment effect in patients with the SCA Type 3 (SCA3) genotype,
which represents the most common form of SCA and accounted for 41
percent of the study population. In the SCA3 subgroup (Figure
1), troriluzole showed a numerical treatment benefit on the change
in f-SARA score from baseline to Week 48 compared to placebo (least
squares [LS] mean change difference -0.55, nominal p-value = 0.053,
95% CI: -1.12, 0.01). SCA patients treated with troriluzole showed
minimal disease progression over the study period. Further,
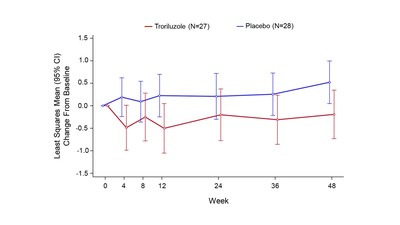
in patients in the SCA3 subgroup who were able to walk without
assistance at baseline (i.e., f-SARA Gait Item score = 1),
troriluzole demonstrated a greater numerical treatment benefit on
the change in f-SARA score from baseline to Week 48 compared to
placebo (LS mean change difference -0.71, nominal p-value = 0.031,
95% CI: -1.36, -0.07). Notably, the f-SARA is a novel, 16-point
scale developed in collaboration with FDA as the primary outcome
measure for this trial; the scale was designed to limit
subjectivity of the scale and focus on functional aspects of the
disease so that significant changes would be considered clinically
meaningful.
Across all genotypes, patients who were able to ambulate at
baseline (i.e., f-SARA Gait Item score = 1) showed a reduction
in the relative risk of falls in troriluzole-treated patients
versus placebo. Patient reported falls, as measured by adverse
events reveal an approximately 58% reduction of fall risk in the
troriluzole group (10% versus 23% AE incidence of falls in the
troriluzole and placebo groups, respectively; nominal p=0.043).
Overall, troriluzole demonstrated a favorable safety and
tolerability profile, consistent with past clinical trial
experience.
Jeremy Schmahmann, M.D.,
Professor of Neurology at Harvard Medical
School and Founding Director of the Ataxia Center at
Massachusetts General Hospital (MGH) commented with regard to the
findings, "The importance of morbidity related to falls in this
patient population cannot be overstated. The reduction of falls in
the troriluzole group, combined with the progression of f-SARA
scores in the untreated SCA3 group compared to SCA3 patients on
troriluzole, demonstrates that SCA3 patients are experiencing a
clinically meaningful improvement in ataxia symptoms on troriluzole
treatment."
Spinocerebellar ataxia is a group of dominantly inherited
disorders characterized by progressive ataxia affecting
coordination of hands, arms and legs as well as balance and speech.
Patients experience significant morbidity, including progression to
a wheelchair, impaired gait leading to falls, inability to
communicate due to speech impairment, difficulty swallowing, and
premature death. While signs and symptoms can appear anytime
from childhood to late adulthood, SCA typically presents in early
adulthood and progresses over a number of years. The range of
symptoms and rate of progression of disease depend on the type of
SCA, age of onset, and other genetic factors. Currently,
there are no FDA-approved treatments and there is no cure for
SCA.
Dr. Susan Perlman, Director of
Ataxia Clinic and Neurogenetics Clinical Trials at the David Geffen
School of Medicine at UCLA stated, "The
f-SARA scale, with a total of 16 points, was specifically designed
to detect significant clinically meaningful change in this patient
population. The f-SARA change in the SCA3 troriluzole treated group
is compelling. Given the excellent safety profile of troriluzole,
and the fact that there are no approved treatment options for
patients with this devastating neurodegenerative disorder, I am
thrilled for my patients that Biohaven is planning to engage with
the FDA to potentially move this program forward."
Vlad Coric, M.D., Chief Executive
Officer of Biohaven stated, "The fact that the overall study
population did not show significant disease progression on the
f-SARA at 1 year, as would have been anticipated, and failed to
meet the study's primary outcome measure, highlight some of the
challenges of studying rare diseases such as SCA. However, the post
hoc analyses by SCA genotype suggest early and sustained
improvements in the f-SARA over 48 weeks for SCA3 patients treated
with troriluzole compared to placebo. The scale improvements
were clinically meaningful, consistent with the role that glutamate
excitotoxicity is thought to play in the underlying pathogenesis of
this disease and was associated with an important risk reduction in
falls in the same SCA3 study population, as well as across all SCA
genotypes. Patients with SCA3 have no approved therapies and suffer
severe disease-related morbidities, including being at high risk
for falling due to incoordination of gait. Given the debilitating
nature of this disease, we look forward to sharing the SCA3
genotype data with regulators and working with the FDA to address
the high unmet need in this patient population."
About Troriluzole
Troriluzole is a third-generation
prodrug and new chemical entity that modulates glutamate, the most
abundant excitatory neurotransmitter in the human body. The primary
mode of action of troriluzole is reducing synaptic levels of
glutamate. Troriluzole increases glutamate uptake from the synapse,
by augmenting the expression and function of excitatory amino acid
transporters located on glial cells that play a key role in
clearing glutamate from the synapse. Troriluzole has the potential
to be developed in a number of other diseases associated with
excessive glutamate. Clinical trials are currently underway to
evaluate the efficacy of troriluzole in Obsessive Compulsive
Disorder (OCD). More information about troriluzole can be found at
the Company's
website: https://www.biohavenpharma.com/science-pipeline/glutamate/troriluzole
About Biohaven
Biohaven is a commercial-stage
biopharmaceutical company with a portfolio of innovative,
best-in-class therapies to improve the lives of patients with
debilitating neurological and neuropsychiatric diseases, including
rare disorders. Biohaven's neuroinnovation portfolio includes
FDA-approved NURTEC ODT (rimegepant) for the acute and preventive
treatment of migraine and a broad pipeline of late-stage product
candidates across three distinct mechanistic platforms: CGRP
receptor antagonism for the acute and preventive treatment of
migraine; glutamate modulation for obsessive-compulsive disorder
and spinocerebellar ataxia; MPO inhibition for multiple amyotrophic
lateral sclerosis; Kv7 ion channel activators for focal
epilepsy and neuronal hyperexcitability, and myostatin inhibition
for neuromuscular diseases. More information about Biohaven is
available at www.biohavenpharma.com.
Forward-Looking Statements
This news release includes
forward-looking statements within the meaning of the Private
Securities Litigation Reform Act of 1995. The use of certain words,
including "believe", "continue", "may", "will", "anticipate",
"expect" and similar expressions, are intended to identify
forward-looking statements. These forward-looking statements
involve substantial risks and uncertainties, including statements
that are based on the current expectations and assumptions of
Biohaven's management about troriluzole as a treatment for patients
with SCA. Factors that could affect these forward-looking
statements include those related to Biohaven's ability to
effectively develop troriluzole. Various important factors could
cause actual results or events to differ materially from those that
may be expressed or implied by forward-looking statements.
Additional important factors to be considered in connection with
forward-looking statements are described in the "Risk Factors"
section of the Company's Annual Report on Form 10-K filed with the
Securities and Exchange Commission on February 10, 2022, and in Biohaven's subsequent
filings with the Securities and Exchange Commission. The
forward-looking statements are made as of the date of this new
release, and Biohaven does not undertake any obligation to update
any forward-looking statements, whether as a result of new
information, future events or otherwise, except as required by
law.
Biohaven Contact:
Jennifer Porcelli
VP, Investor Relations
Jennifer.porcelli@biohavenpharma.com
Media Contact:
Mike Beyer
Sam Brown Inc.
mikebeyer@sambrown.com
312-961-2502
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-provides-update-on-phase-3-clinical-trial-evaluating-troriluzole-for-spinocerebellar-ataxia-sca-301552633.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-provides-update-on-phase-3-clinical-trial-evaluating-troriluzole-for-spinocerebellar-ataxia-sca-301552633.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.