Lead Researcher Behind Genprex’s Diabetes Gene Therapy Featured in New Video Interview
March 05 2020 - 8:05AM
Business Wire
Genprex, Inc. (“Genprex” or the “Company”) (NASDAQ: GNPX), a
clinical-stage gene therapy company developing potentially
life-changing technologies for patients with cancer and other
serious diseases, today announced that Dr. George Gittes, the lead
researcher and Harvard graduate that developed Genprex’s new
potentially curative diabetes gene therapy, was featured in a video
interview discussing the therapy.
Dr. Gittes sat down with one of Proactive’s broadcast
journalists to give an overview of the gene therapy, how it works,
how it may have the potential to help treat type 1 and type 2
diabetes and next steps to move the gene therapy to the clinic.
“We noticed that by gene therapy we could cause some of the
cells in the pancreas that do not normally make insulin, but they
are closely related to insulin cells, to turn into an
insulin-producing cell,” said Dr. Gittes, Co-Scientific Director
and Professor of Surgery at the UPMC Children’s Hospital of
Pittsburgh. “The real excitement came when we realized that when we
did this in a mouse that normally reacts to its own insulin cells
in the same way a juvenile diabetic or Type 1 diabetic does, it
didn’t notice these [new] cells and left them alone for a long time
without us doing anything else. It was just one treatment with a
gene therapy, and they [the mice] were fine for months, which is
exciting.”
To watch the full video interview, please visit:
http://bit.ly/2Ii8C1J.
In February 2020, Genprex signed an exclusive license agreement
with the University of Pittsburgh for the potentially curative gene
therapy for diabetes. According to the American Diabetes
Association, more than 30 million Americans have diabetes, and
approximately 1.5 million Americans are diagnosed with diabetes
every year.
Dr. Gittes and his team at the University of Pittsburgh plan to
continue studying the technology in a pre-clinical phase. As the
research is refined and sufficient animal data is generated, the
Company, in conjunction with Dr. Gittes and his team, plan to move
forward with a Phase I clinical trial in diabetic patients, which
could be the first-ever gene therapy tested in humans for
diabetes.
About Genprex, Inc.
Genprex, Inc. is a clinical-stage gene therapy company
developing potentially life-changing technologies for patients with
cancer and other serious diseases. Genprex’s technologies are
designed to administer disease-fighting genes to provide new
treatment options for large patient populations with cancer and
other serious diseases who currently have limited treatment
options. Genprex works with world-class institutions and
collaborators to in-license and develop drug candidates to further
its pipeline of gene therapies in order to provide novel treatment
approaches for patients with cancer and other serious diseases. The
company’s lead product candidate, Oncoprex™ immunogene therapy for
non-small cell lung cancer (NSCLC), uses the company’s unique,
proprietary platform which delivers cancer-fighting genes by
encapsulating them into nanoscale hollow spheres called
nanovesicles, which are then administered intravenously and taken
up by tumor cells where they express proteins that are missing or
found in low quantities. In January 2020, the FDA granted Fast
Track Designation for Oncoprex in combination with AstraZeneca’s
Tagrisso® for the treatment of NSCLC. For more information, please
visit the company’s website at www.genprex.com or follow Genprex on
Twitter, Facebook and LinkedIn.
Forward-Looking Statements
Statements contained in this press release regarding matters
that are not historical facts are “forward-looking statements”
within the meaning of the Private Securities Litigation Reform Act
of 1995. Because such statements are subject to risks and
uncertainties, actual results may differ materially from those
expressed or implied by such forward-looking statements. Such
statements include, but are not limited to, statements regarding
the effects of the licensed gene therapy on diabetes and the effect
of Genprex’s other product candidates, alone and in combination
with other therapies, on cancer, as well as Genprex’s ongoing and
planned preclinical and clinical studies and potential
partnerships. Risks that contribute to the uncertain nature of the
forward-looking statements include risks relating to the effects of
the safety and effectiveness of the licensed gene therapy and of
Genprex’s other product candidates, alone and in combination with
other therapies, as well as the success of Genprex’s ongoing and
planned preclinical and clinical studies and the success of
Genprex’s efforts in concluding potential partnering arrangements
for product development and commercialization. Other risks and
uncertainties associated with Genprex and its product candidates
are described more fully under the caption “Risk Factors” and
elsewhere in Genprex’s filings and reports with the United States
Securities and Exchange Commission. All forward-looking statements
contained in this press release speak only as of the date on which
they were made. Genprex undertakes no obligation to update such
statements to reflect events that occur or circumstances that exist
after the date on which they were made.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200305005053/en/
Genprex, Inc. (877) 774-GNPX (4679)
Investor Relations GNPX Investor Relations (877) 774-GNPX
(4679) ext. #2 investors@genprex.com
Media Contact Genprex Media Relations Kalyn Dabbs (877)
774-GNPX (4679) ext. #3 media@genprex.com
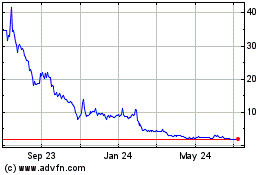
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Mar 2024 to Apr 2024
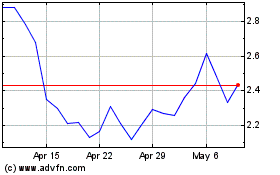
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Apr 2023 to Apr 2024