If any of the securities being registered
on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, check the
following box. x
If this Form is filed to register additional
securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities
Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment
filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment
filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same offering. o
Indicate by check mark whether the registrant is a large accelerated
filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions
of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging
growth company” in Rule 12b-2 of the Exchange Act.
If an emerging growth company, indicate
by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial
accounting standards provided to Section 7(a)(2)(B) of the Securities Act. o
PROSPECTUS SUMMARY
This summary highlights information
contained elsewhere in this prospectus and does not contain all of the information that you should consider in making your investment
decision. Before investing in our securities, you should carefully read this entire prospectus, including the documents to which
we have referred you under the headings “Where You Can Find More Information” and “Incorporation of Documents
by Reference” and the information set forth under the headings “Risk Factors” and “Management’s Discussion
and Analysis of Financial Condition and Results of Operations,” in each case, included elsewhere in this prospectus or incorporated
herein by reference.
Overview
We are a clinical stage biopharmaceutical
company focused on the discovery, development and commercialization of drugs for the treatment of cancer. We are developing proprietary
drugs independently and through research and development collaborations. Our core objective is to leverage our proprietary phospholipid
drug conjugate™ (PDC™) delivery platform to develop PDCs that are designed to specifically target cancer cells, and
deliver improved efficacy and better safety as a result of fewer off-target effects. Our PDC platform possesses the potential for
the discovery and development of the next generation of cancer-targeting treatments, and we plan to develop PDCs both independently
and through research and development collaborations. The COVID-19 pandemic has created uncertainties in the expected timelines
for clinical stage biopharmaceutical companies such as us, and because of such uncertainties, it is difficult for us to accurately
predict expected outcomes at this time. We have continued to enroll patients in our clinical trials. However, COVID-19 may impact
our ability to recruit patients for clinical trials, obtain adequate supply of CLR 131 and obtain additional financing.
CLR 131 and PDC Platform
Our lead PDC therapeutic, CLR 131 is a
small-molecule PDC designed to provide targeted delivery of iodine-131 directly to cancer cells, while limiting exposure to healthy
cells. We believe this profile differentiates CLR 131 from many traditional on-market treatment options. CLR 131 is the company’s
lead product candidate and is currently being evaluated in a Phase 2 study in relapsed/refractory (r/r) malignancies, including
multiple myeloma (MM), chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), lymphoplasmacytic lymphoma/Waldenstrom’s
macroglobulinemia (LPL/WM), marginal zone lymphoma (MZL), mantle cell lymphoma (MCL), and diffuse large B-cell lymphoma (DLBCL).
CLR 131 is also being evaluated in a Phase 1 dose escalation study in pediatric solid tumors and lymphoma. The U.S. Food and Drug
Administration (“FDA”) granted CLR 131 Fast Track Designation for both r/r MM and r/r DLBCL and Orphan Drug Designation
(ODD) of MM, LPL/WM, neuroblastoma, rhabdomyosarcoma, Ewing’s sarcoma and osteosarcoma. CLR 131 was also granted Rare Pediatric
Disease Designation (RPDD) for the treatment of neuroblastoma, rhabdomyosarcoma, Ewing’s sarcoma and osteosarcoma. Most recently,
the European Commission granted an ODD for r/r MM.
Our product pipeline also includes one
preclinical PDC chemotherapeutic program (CLR 1900) and several partnered PDC assets. The CLR 1900 Series is being targeted for
solid tumors with a payload that inhibits mitosis (cell division) a validated pathway for treating cancers.
We have leveraged our PDC platform to establish
four collaborations featuring five unique payloads and mechanisms of action. Through research and development collaborations, our
strategy is to generate near-term capital, supplement internal resources, gain access to novel molecules or payloads, accelerate
product candidate development and broaden our proprietary and partnered product pipelines.
Our PDC platform provides selective delivery
of a diverse range of oncologic payloads to cancerous cells, whether a hematologic cancer or solid tumor, a primary tumor, or a
metastatic tumor and cancer stem cells. The PDC platform’s mechanism of entry does not rely upon specific cell surface epitopes
or antigens as are required by other targeted delivery platforms. Our PDC platform takes advantage of a metabolic pathway utilized
by all tumor cell types in all stages of the tumor cycle. Tumor cells modify specific regions on the cell surface as a result of
the utilization of this metabolic pathway. Our PDCs bind to these regions and directly enter the intracellular compartment. This
mechanism allows the PDC molecules to accumulate over time, which enhances drug efficacy, and to avoid the specialized highly acidic
cellular compartment known as lysosomes, which allows a PDC to deliver molecules that previously could not be delivered. Additionally,
molecules targeting specific cell surface epitopes face challenges in completely eliminating a tumor because the targeted antigens
are limited in the total number on the cell surface, have longer cycling time from internalization to being present on the cell
surface again and available for binding and are not present on all of the tumor cells in any cancer. This means a subpopulation
of tumor cells always exist that cannot be targeted by therapies targeting specific surface epitopes. In addition to the benefits
provided by the mechanism of entry, PDCs offer the ability to conjugate payload molecules in numerous ways, thereby increasing
the types of molecules selectively delivered via the PDC.
The PDC platform features include the capacity
to link with almost any molecule, provide a significant increase in targeted oncologic payload delivery and the ability to target
all types of tumor cells. As a result, we believe that we can generate PDCs to treat a broad range of cancers with the potential
to improve the therapeutic index of oncologic drug payloads, enhance or maintain efficacy while also reducing adverse events by
minimizing drug delivery to healthy cells, and increasing delivery to cancerous cells and cancer stem cells.
We employ a drug discovery and development
approach that allows us to efficiently design, research and advance drug candidates. Our iterative process allows us to rapidly
and systematically produce multiple generations of incrementally improved targeted drug candidates.
A description of our PDC product candidates
follows:
Clinical Pipeline
Our lead PDC therapeutic, CLR 131 is a small-molecule,
PDC designed to provide targeted delivery of iodine-131 directly to cancer cells, while limiting exposure to healthy cells. We
believe this profile differentiates CLR 131 from many traditional on-market treatments and treatments in development. CLR 131 is
currently being evaluated in a Phase 2 study in r/r B-cell lymphomas, and two Phase 1 dose-escalating clinical studies, one in
r/r MM and one in r/r pediatric solid tumors and lymphoma. The initial Investigational New Drug (IND) application was accepted
by the FDA in March 2014 with multiple INDs submitted since that time. Initiated in March 2017, the primary goal of the Phase 2
study is to assess the compound’s efficacy in a broad range of hematologic cancers. The Phase 1 study is designed to assess
the compound’s safety and tolerability in patients with r/r MM (to determine maximum tolerated dose) and was initiated in
April 2015. The FDA previously accepted our IND application for a Phase 1 open-label, dose escalating study to evaluate the safety
and tolerability of a single intravenous administration of CLR 131 in up to 30 children and adolescents with cancers including
neuroblastoma, sarcomas, lymphomas (including Hodgkin’s lymphoma) and malignant brain tumors. This study was initiated during
the first quarter of 2019. These cancer types were selected for clinical, regulatory and commercial rationales, including the radiosensitive
nature and continued unmet medical need in the r/r setting, and the rare disease determinations made by the FDA based upon the
current definition within the Orphan Drug Act.
In December 2014, the FDA granted ODD for CLR 131
for the treatment of MM. Multiple myeloma is an incurable cancer of the plasma cells and is the second most common form of hematologic
cancers. In 2018, the FDA granted ODD and RPDD for CLR 131 for the treatment of neuroblastoma, rhabdomyosarcoma, Ewing’s
sarcoma and osteosarcoma. The FDA may award priority review vouchers to sponsors of rare pediatric disease products that meet its
specified criteria. The key criteria to receiving a priority review voucher is that the disease being treated is life-threatening
and that it primarily effects individuals under the age of 18. Under this program, a sponsor who receives an approval for a drug
or biologic for a rare pediatric disease can receive a priority review voucher that can be redeemed to receive a priority review
of a subsequent marketing application for a different product. Additionally, these priority review vouchers can be exchanged or
sold to other companies for them to use the voucher. In May 2019, the FDA granted Fast Track designation for CLR 131 for the treatment
of multiple myeloma in July 2019 for the treatment of DLBCL, in September, CLR 131 received Orphan Drug Designation from the European
Union for Multiple Myeloma, and in January 2020, CLR 131 the FDA granted Orphan Drug Designation for CLR 131 in lymphoplasmacytic
lymphoma (LPL).
Phase 2 Study in Patients with r/r select B-cell
Malignancies
In February 2020, we announced positive data from
our Phase 2 CLOVER-1 study in patients with relapsed/refractory B-cell lymphomas. Relapsed/Refractory MM and non-Hodgkin lymphoma
(NHL) patients were treated with three different doses (<50mCi, ~50mCi and ~75mCi total body dose (TBD). The <50mCi total
body dose was a deliberately planned sub-therapeutic dose. CLR 131 achieved the primary endpoint for the study. Patients with r/r
MM who received the highest dose of CLR 131 showed a 42.8% overall response rate (ORR). Those who received ~50mCi TBD had a 26.3%
ORR with a combined rate of 34.5% ORR (n=33) while maintaining a well-tolerated safety profile. Patients in the studies were elderly
with a median age of 70, and heavily pre-treated, with a median of five prior lines of treatment (range: 3 to 17), which included
immunomodulatory drugs, proteasome inhibitors and CD38 antibodies for the majority of patients. Additionally, a majority of the
patients (53%) were quad refractory or greater and 44% of all treated multiple myeloma patients were triple class refractory. 100%
of all evaluable patients (n=43) achieved clinical benefit (primary outcome measure) as defined by having stable disease or better.
85.7% of multiple myeloma patients receiving the higher total body dose levels of CLR 131 experienced tumor reduction. The 75mCi
TBD demonstrated positive activity in both high-risk patients and triple class refractory patients with a 50% and 33% ORR, respectively.
Patients with r/r NHL who received ~50mCi TBD and
the ~75mCi TBD had a 42% and 43% ORR, respectively and a combined rate of 42%. These patients were also heavily pre-treated, having
a median of three prior lines of treatment (range, 1 to 9) with the majority of patients being refractory to rituximab and/or ibrutinib.
The patients had a median age of 70 with a range of 51 to 86. All patients had bone marrow involvement with an average of 23%.
In addition to these findings, subtype assessments were completed in the r/r B-cell NHL patients. Patients with DLBCL demonstrated
a 30% ORR with one patient achieving a complete response (CR), which continues at nearly 24 months post-treatment. The ORR for
CLL/SLL/MZL patients was 33%. Current data from our Phase 2 CLOVER-1 clinical study show that four LPL/WM patients demonstrated
100% ORR with one patient achieving a CR which continues at nearly 27 months post-treatment. This may represent an important improvement
in the treatment of relapsed/refractory LPL/WM as we believe no approved or late-stage development treatments for second- and third-line
patients have reported a CR. LPL/WM is a rare, indolent and incurable form of NHL that is comprised of a niche patient population
in need of new and better treatment options.
The most frequently reported adverse events in r/r
MM patients were cytopenias, which followed a predictable course and timeline. The frequency of adverse events have not increased
as doses were increased and the profile of cytopenias remains consistent. Importantly, these cytopenias have had a predictable
pattern to initiation, nadir and recovery and are treatable. The most common grade ≥3 events at the highest dose (75mCi TBD)
were hematologic toxicities including thrombocytopenia (65%), neutropenia (41%), leukopenia (30%), anemia (24%) and lymphopenia
(35%). No patients experienced cardiotoxicities, neurological toxicities, infusion site reactions, peripheral neuropathy, allergic
reactions, cytokine release syndrome, keratopathy, renal toxicities, or changes in liver enzymes. The safety and tolerability profile
in patients with r/r NHL was similar to r/r MM patients except for fewer cytopenias of any grade. Based upon CLR 131 being well
tolerated across all dose groups and the profound observed response rate, especially in difficult to treat patients such as high
risk and triple class refractory or penta-refractory, and corroborating data showing the potential to further improve upon current
ORRs and durability of those responses, the study has been expanded to test a two-cycle dosing optimization regimen of CLR 131.
In July 2016, we were awarded a $2,000,000 National
Cancer Institute (NCI) Fast-Track Small Business Innovation Research grant to further advance the clinical development of CLR 131.
The funds are supporting the Phase 2 study initiated in March 2017 to define the clinical benefits of CLR 131 in r/r MM and other
niche hematologic malignancies with unmet clinical need. These niche hematologic malignancies include Chronic Lymphocytic Leukemia,
Small Lymphocytic Lymphoma, Marginal Zone Lymphoma, Lymphoplasmacytic Lymphoma and DLBCL. The study is being conducted in approximately
10 U.S. cancer centers in patients with orphan-designated relapse or refractory hematologic cancers. The study’s primary
endpoint is CBR, with additional endpoints of ORR, PFS, median Overall Survival (mOS) and other markers of efficacy following a
single 25.0 mCi/m2 dose of CLR 131, with the option for a second 25.0 mCi/m2 dose approximately 75-180 days later. Based on the
performance results from Cohort 5 of our Phase 1 study in patients with r/r MM, reviewed below, we have modified the dosing regimen
of this study to a fractionated dose of 15.625 mCi/m2 administered on day 1 and day 8.
Phase 1 Study in Patients with r/r Multiple Myeloma
In February 2020, we announced the successful completion
of our Phase 1 dose escalation study. Data from the study demonstrated that CLR 131 was safe and tolerated at total body doses
of >80mCi in r/r multiple myeloma (MM), The Phase 1 multicenter, open-label, dose-escalation study was designed to evaluate
the safety and tolerability of CLR 131 administered as a 30-minute I.V. infusion, either as a single bolus dose or as two fractionated
doses. The r/r multiple myeloma patients in this study received doses ranging from ≤25mCi to >80mCi total body dose. To date,
an independent Data Monitoring Committee determined that all doses have been safe and well-tolerated by patients.
CLR 131 in combination with dexamethasone is currently
under investigation in adult patients with r/r MM. Patients must have been refractory to or relapsed from at least one proteasome
inhibitor and at least one immunomodulatory agent. The clinical study is a standard three-plus-three dose escalation safety study
to determine the maximum tolerable dose. Multiple myeloma is an incurable cancer of the plasma cells and is the second most common
form of hematologic cancers. Secondary objectives include the evaluation of therapeutic activity by assessing surrogate efficacy
markers, which include M protein, free light chain FLC, PFS and OS. All patients have been heavily pretreated with an average of
five prior lines of therapy. CLR 131 was deemed by an Independent Data Monitoring Committee (IDMC) to be safe and tolerable up
to its planned maximum single, bolus dose of 31.25 mCi/m2. The four single dose cohorts examined were: 12.5 mCi/m2 (~25mCi TBD),
18.75 mCi/m2 (~37.5mCi TBD), 25 mCi/m2(~50mCi TBD), and 31.25 mCi/m2(~62.5mCi TBD), all in combination with low dose dexamethasone
(40 mg weekly). Of the five patients in the first cohort, four achieved stable disease and one patient progressed at Day 15 after
administration and was taken off the study. Of the five patients admitted to the second cohort, all five achieved stable disease
however one patient progressed at Day 41 after administration and was taken off the study. Four patients were enrolled to the third
cohort and all achieved stable disease. In September 2017, we announced results for cohort 4, showing that a single infusion up
to 30-minutes of 31.25mCi/m2 of CLR 131 was safe and tolerated by the three patients in the cohort. Additionally, all three patients
experienced CBR with one patient achieving a partial response (PR). We use the International Myeloma Working Group (IMWG) definitions
of response, which involve monitoring the surrogate markers of efficacy, M protein and FLC. The IMWG defines a PR as a greater
than or equal to 50% decrease in FLC levels (for patients in whom M protein is unmeasurable) or 50% or greater decrease in M protein.
The patient experiencing a PR had an 82% reduction in FLC. This patient did not produce M protein, had received seven prior lines
of treatment including radiation, stem cell transplantation and multiple triple combination treatments including one with daratumumab
that was not tolerated. One patient experiencing stable disease attained a 44% reduction in M protein. In January 2019, we announced
that the pooled mOS data from the first four cohorts was 22.0 months. In late 2018, we modified this study to evaluate a fractionated
dosing strategy to potentially increase efficacy and decrease adverse events.
Following the determination that all prior dosing
cohorts were safe and tolerated, we initiated a cohort 7 utilizing a 40mCi/m2 fractionated dose administered 20mCi/m2 (~40mCi TBD)
on days 1 and day 8. Cohort 7 was the highest pre-planned dose cohort and subjects have completed the evaluation period. Final
study report and study close-out will be completed later this year.
In May 2019, we announced that the FDA granted Fast
Track Designation for CLR 131 in fourth line or later r/r MM. CLR 131 is our small-molecule radiotherapeutic PDC designed to deliver
cytotoxic radiation directly and selectively to cancer cells and cancer stem cells. It is currently being evaluated in our ongoing
CLOVER-1 Phase 2 clinical study in patients with relapsed or refractory multiple myeloma and other select B-cell lymphomas.
Phase 1 Study in r/r Pediatric Patients with select
Solid tumors, Lymphomas and Malignant Brain Tumors
In December 2017 the Division of Oncology at the FDA
accepted our IND and study design for the Phase 1 study of CLR 131 in children and adolescents with select rare and orphan designated
cancers. This study was initiated during the first quarter of 2019. In December 2017, we filed an IND application for r/r pediatric
patients with select solid tumors, lymphomas and malignant brain tumors. The Phase 1 clinical study of CLR 131 is an open-label,
sequential-group, dose-escalation study evaluating the safety and tolerability of intravenous administration of CLR 131 in up to
30 children and adolescents with cancers including neuroblastoma, sarcomas, lymphomas (including Hodgkin’s lymphoma) and
malignant brain tumors. Secondary objectives of the study are to identify the recommended Phase 2 dose of CLR 131 and to determine
preliminary antitumor activity (treatment response) of CLR 131 in children and adolescents. In 2018, the FDA granted OD and RPDD
for CLR 131 for the treatment of neuroblastoma, rhabdomyosarcoma, Ewing’s sarcoma and osteosarcoma. Should any of these indications
reach approval, the RPDD would enable us to receive a priority review voucher. Priority review vouchers can be used by the sponsor
to receive priority review for a future New Drug Application (“NDA”) or Biologic License Application (“BLA”)
submission, which would reduce the FDA review time from 12 months to six months. Currently, these vouchers can also be transferred
or sold to another entity.
Phase 1 Study in r/r Head and Neck Cancer
In August 2016, the University of Wisconsin Carbone
Cancer Center (“UWCCC”) was awarded a five-year Specialized Programs of Research Excellence (“SPORE”) grant
of $12,000,000 from the National Cancer Institute and the National Institute of Dental and Craniofacial Research to improve treatments
and outcomes for head and neck cancer, HNC, patients. HNC is the sixth most common cancer across the world with approximately 56,000
new patients diagnosed every year in the U.S. As a key component of this grant, the UWCCC researchers completed testing of CLR
131 in various animal HNC models and initiated the first human clinical trial enrolling up to 30 patients combining CLR 131 and
external beam radiation with recurrent HNC in Q4 2019. As of the date of this filing, this clinical trial is suspended due to the
COVID-19 pandemic.
Preclinical Pipeline
We believe our PDC platform has potential to provide
targeted delivery of a diverse range of oncologic payloads, as exemplified by the product candidates listed below, that may result
in improvements upon current standard of care (“SOC”) for the treatment of a broad range of human cancers:
|
|
·
|
CLR 1800 Series was a collaborative PDC program with Pierre Fabre that expired in January 2019. The program has been successful in demonstrating improved tolerability and efficacy in multiple animal models. The newly developed PDCs may provide enhanced therapeutic indices to otherwise highly potent, nontargeted payloads through the targeted delivery of the chemotherapeutic payload to cancer cells via our proprietary phospholipid ether delivery platform. The CLR 1800 Series remains under evaluation by us as a number of PDC molecules have the potential to be progressed toward and into IND enabling studies.
|
|
|
·
|
CLR 1900 Series is an internally developed proprietary PDC program leveraging a novel small molecule cytotoxic compound as the payload. The payload inhibits mitosis (cell division) and targets a key pathway required to inhibit rapidly dividing cells that results in apoptosis. We believe that this program could produce a product candidate targeted to select solid tumors. Currently, the program is in early preclinical development and if we elect to progress any molecules further, we will select preferred candidates.
|
|
|
·
|
CLR 2000 Series is a collaborative PDC program with Avicenna Oncology, or Avicenna, that we entered into in July 2017. Avicenna is a developer of antibody drug conjugates (“ADCs”). The objective of the research collaboration is to design and develop a series of PDCs utilizing Avicenna’s proprietary cytotoxic payload. Although Avicenna is a developer of ADCs, this collaboration was sought as a means to overcome many of the challenges associated with ADCs, including those associated with the targeting of specific cell surface epitopes. The CLR 2000 Series has demonstrated improved safety, efficacy and tissue distribution with the cytotoxic payload in animal models. A candidate molecule and a back-up have been selected for further advancement.
|
|
|
·
|
CLR 2100 and 2200 Series are collaborative PDC programs with Onconova Therapeutics, Inc., or Onconova, that we entered into in September 2017. Onconova is a biotechnology company specializing in the discovery and development of novel small molecule cancer therapies. The collaboration is structured such that we will design and develop a series of PDCs utilizing different small molecules that Onconova was developing as payloads with the intent to show improved targeting and specificity to the tumor. At least one of the molecules was taken into Phase 1 clinical studies previously by Onconova. We would own all new intellectual property associated with the design of the new PDCs, and both companies will have the option to advance compounds.
|
|
|
·
|
CLR 12120 Series is a collaborative PDC program with Orano Med for the development of novel PDCs utilizing Orano Med’s unique alpha emitter, lead 212 conjugated to our phospholipid ether; the companies intend to evaluate the new PDCs in up to three oncology indications. Currently this series has shown efficacy in the first two animal models tested.
|
Our shares are listed on the Nasdaq Capital
Market under the symbol “CLRB.” Before August 15, 2014, our shares were quoted on the OTCQX marketplace, and prior
to February 12, 2014, they were quoted under the symbol “NVLT.”
Key Risks and Uncertainties
We are subject to numerous risks and uncertainties,
including the following:
|
|
·
|
Our operations and financial condition may be adversely impacted by the COVID-19 pandemic.
|
|
|
·
|
We will require additional capital in order to continue our operations and may have difficulty raising additional capital.
|
|
|
·
|
We are a clinical-stage company with a going concern qualification to our financial statements and a history of losses, and
we can provide no assurance as to our future operating results.
|
|
|
·
|
We rely on a collaborative outsourced business model, and disruptions with these third-party collaborators may impede our ability
to gain FDA approval and delay or impair commercialization of any products.
|
|
|
·
|
We will require additional capital in order to continue our operations and may have difficulty raising additional capital.
|
|
|
·
|
We rely on a small number of key personnel who may terminate their employment with us at any time, and our success will depend
on our ability to hire additional qualified personnel.
|
|
|
·
|
We cannot assure the successful development and commercialization of our compounds in development.
|
|
|
·
|
Our proposed products and their potential applications are in an early stage of clinical and manufacturing/process development
and face a variety of risks and uncertainties.
|
|
|
·
|
Failure to complete the development of our technologies, to obtain government approvals, including required FDA approvals,
or comply with ongoing governmental regulations could prevent, delay or limit introduction or sale of proposed products and result
in failure to achieve revenues or maintain our ongoing business.
|
|
|
·
|
Clinical studies involve a lengthy and expensive process with an uncertain outcome, and results of earlier studies and trials
may not be predictive of future trial results.
|
|
|
·
|
We may be required to suspend or discontinue clinical studies due to unexpected side effects or other safety risks that could
preclude approval of our product candidates.
|
|
|
·
|
Controls we or our third-party collaborators have in place to ensure compliance with all applicable laws and regulations may
not be effective.
|
|
|
·
|
We expect to rely on our patents as well as specialized regulatory designations such as orphan drug classification for our
product candidates, but regulatory drug designations may not confer marketing exclusivity or other expected commercial benefits.
|
|
|
·
|
The FDA has granted rare pediatric disease designation, RPDD, to CLR 131 for treatment of neuroblastoma and rhabdomyosarcoma;
however, we may not be able to realize any value from such designation.
|
|
|
·
|
We are exposed to product, clinical and preclinical liability risks that could create a substantial financial burden should
we be sued.
|
|
|
·
|
Acceptance of our products in the marketplace is uncertain and failure to achieve market acceptance will prevent or delay our
ability to generate revenues.
|
|
|
·
|
The market for our proposed products is rapidly changing and competitive, and new therapeutics, drugs and treatments that may
be developed by others could impair our ability to develop our business or become competitive.
|
|
|
·
|
We may face litigation from third parties claiming that our products infringe on their intellectual property rights, particularly
because there is often substantial uncertainty about the validity and breadth of medical patents.
|
|
|
·
|
If we are unable to adequately protect or enforce our rights to intellectual property or to secure rights to third-party patents,
we may lose valuable rights, experience reduced market share, assuming any, or incur costly litigation to protect our intellectual
property rights.
|
|
|
·
|
Conflicts, military actions, terrorist attacks, natural disasters. public health crises, including the occurrence of a contagious
disease or illness, such as the COVID-19 coronavirus, cyber-attacks and general instability could adversely affect our business.
|
|
|
·
|
Confidentiality agreements with employees and others may not adequately prevent disclosure of our trade secrets and other proprietary
information and may not adequately protect our intellectual property, which could limit our ability to compete.
|
|
|
·
|
We may be subject to claims that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
|
|
|
·
|
Due to continued changes in marketing, sales and distribution, we may be unsuccessful in our efforts to sell our proposed products,
develop a direct sales organization, or enter into relationships with third parties.
|
|
|
·
|
If we are unable to convince physicians of the benefits of our intended products, we may incur delays or additional expense
in our attempt to establish market acceptance.
|
|
|
·
|
If users of our products are unable to obtain adequate reimbursement from third-party payors, or if additional healthcare reform
measures are adopted, it could hinder or prevent the commercial success of our product candidates.
|
|
|
·
|
Our business and operations may be materially, adversely affected in the event of computer system failures or security breaches.
|
|
|
·
|
Failure to maintain effective internal controls could adversely affect our ability to meet our reporting requirements.
|
|
|
·
|
We have in the past received notices from Nasdaq of noncompliance with its listing rules, and delisting with Nasdaq could impact
the price of our common stock and our ability to raise funds.
|
|
|
·
|
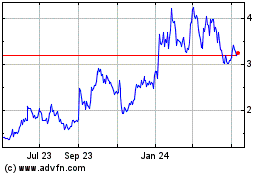
Our stock price has experienced price fluctuations.
|
|
|
·
|
Our common stock could be further diluted as the result of the issuance of additional shares of common stock, convertible securities,
warrants or options.
|
|
|
·
|
Provisions of our certificate of incorporation, by-laws, and Delaware law may make an acquisition of us or a change in our
management more difficult.
|
|
|
·
|
We have not paid dividends in the past and do not expect to pay dividends for the foreseeable future. Any return on investment
may be limited to the value of our common stock.
|
|
|
·
|
Our management team will have immediate and broad discretion over the use of the net proceeds from this offering, and you may
not agree with our use of the net proceeds.
|
|
|
·
|
You will experience immediate and substantial dilution as a result of this offering and may experience additional dilution
in the future.
|
|
|
·
|
You may experience future dilution as a result of future equity offerings.
|
|
|
·
|
The warrants issued in this offering may not have any value.
|
|
|
·
|
A warrant does not entitle the holder to any rights as common stockholders until the holder exercises the warrant for shares
of our common stock.
|
|
|
·
|
There is no public market for the warrants or pre-funded warrants being offered by us in this offering.
|
For more information regarding the material
risks and uncertainties we face, please see “Risk Factors” beginning on page 16 of this prospectus.
Corporate Information
Our principal executive offices are located
at 100 Campus Drive, Florham Park, New Jersey 07932. We maintain a website at www.cellectar.com. The information included or referred
to on, or accessible through, our website does not constitute part of, and is not incorporated by reference into, this prospectus.
The Offering
|
Common stock offered by us:
|
|
shares, assuming the sale of our common stock at an assumed public offering price of $ per share, which is the last reported sale price of our common stock on , 2020, and no sale of any pre-funded warrants.
|
|
Pre-funded warrants offered by us:
|
|
We are also offering to certain purchasers whose purchase of shares of common stock in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock immediately following the consummation of this offering, the opportunity to purchase, if such purchasers so choose, pre-funded warrants, in lieu of shares of common stock that would otherwise result in any such purchaser’s beneficial ownership exceeding 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock. Each pre-funded warrant will be exercisable for one share of our common stock. The purchase price of each pre-funded warrant and the accompanying Series H Warrant will equal the price at which the share of common stock and the accompanying Series H Warrant are being sold to the public in this offering, minus $0.00001, and the exercise price of each pre-funded warrant will be $0.00001 per share. The pre-funded warrants will be exercisable immediately and may be exercised at any time until all of the pre-funded warrants are exercised in full. This offering also relates to the shares of common stock issuable upon exercise of any pre-funded warrants sold in this offering. For each pre-funded warrant we sell, the number of shares of common stock we are offering will be decreased on a one-for-one basis. Because we will issue a Series H warrant for each share of our common stock and for each pre-funded warrant to purchase one share of our common stock sold in this offering, the number of Series H Warrants sold in this offering will not change as a result of a change in the mix of the shares of our common stock and pre-funded warrants sold.
|
|
Description of Series H Warrants:
|
|
Series H Warrants to purchase an aggregate of shares of our common stock. Each share of our common stock and each pre-funded warrant to purchase one share of our common stock is being sold together with a Series H Warrant to purchase one share of our common stock. Each Series H Warrant will have an exercise price of $ per share, will be immediately exercisable and will expire on the fifth anniversary of the original issuance date. The shares of common stock and pre-funded warrants, and the accompanying Series H Warrants, as the case may be, can only be purchased together in this offering but will be issued separately and will be immediately separable upon issuance. This prospectus also relates to the offering of the shares of common stock issuable upon exercise of the Series H Warrants
|
|
Over-allotment Option:
|
|
We have granted the underwriter the option to purchase up to an additional shares of our common stock, solely to cover over-allotments, if any. The underwriter can exercise this option at any time within 30 days after the date of this prospectus
|
|
Shares of common stock outstanding before this offering:
|
|
9,396,015 shares
|
|
Shares of common stock to be outstanding after this offering:
|
|
shares
|
|
Use of proceeds:
|
|
We estimate that the net proceeds to us
from this offering will be approximately $ million,
based on the assumed offering price of $ per share, which is the last reported sale price of our common
stock on the Nasdaq Capital Market on , 2020, and after deducting
the estimated underwriting discounts and commissions and estimated offering expenses payable by us.
We expect to use the net proceeds received from this offering
to fund our research and development activities and for general corporate purposes. For a more complete description of our anticipated
use of proceeds from this offering, see “Use of Proceeds.”
|
|
Risk factors:
|
|
See “Risk Factors” beginning on page 16 and the other information included in this prospectus for a discussion of factors you should carefully consider before deciding whether to purchase our securities.
|
|
Nasdaq symbol for our common stock:
No listing of warrants:
|
|
CLRB
We do not intend to apply for listing of the pre-funded warrants
or Series H Warrants on any national securities exchange or trading system.
|
Unless we specifically state otherwise,
the share information in this prospectus, including the number of shares of common stock outstanding before this offering, is as
of May 5, 2020.
The number of shares of our common stock
outstanding before and after this offering is based on 9,396,015 shares of common stock outstanding as of May 5, 2020 and excludes,
as of that date:
|
|
·
|
an aggregate of 884,464 shares of common stock issuable upon the exercise of outstanding stock options issued to employees,
directors and consultants;
|
|
|
·
|
an aggregate of 537,500 shares of common stock issuable upon the conversion of outstanding shares of Series C preferred stock
|
|
|
·
|
an aggregate of 9,268,352 additional shares of common stock reserved for issuance under outstanding warrants having expiration
dates between October 1, 2020, and October 14, 2024, and exercise prices ranging from $2.40 to $283.00 per share; and
|
|
|
·
|
shares of our common stock that may be issued upon the exercise of pre-funded warrants and Series H warrants issued in this
offering.
|
Unless otherwise noted, the information in this prospectus
reflects and assumes the following:
|
|
·
|
no exercise of outstanding options and warrants; and
|
|
|
·
|
no exercise of the underwriter’s over-allotment option to purchase additional shares of common stock and/or warrants.
|
Summary Historical Financial Information
The following table summarizes our financial
data. We derived the following summary of our statements of operations data for the three months ended March 31, 2020 and 2019
and the summary of our balance sheet data as of March 31, 2020 from our unaudited consolidated financial statements, for the applicable
periods, which have been incorporated by reference in this prospectus. We derived the following summary of our statements
of operations data for the years ended December 31, 2019 and 2018, and the summary of our balance sheet data as of December 31,
2019 and 2018, from our audited consolidated financial statements, for the applicable periods, which have been incorporated by
reference in this prospectus. The summary of our financial data set forth below should be read together with our financial statements
and the related notes to those statements referred to under the heading “Documents Incorporated by Reference.”
|
|
|
Three Months Ended
March 31,
|
|
|
Year Ended
December 31,
|
|
|
|
|
2020
|
|
|
2019
|
|
|
2019
|
|
|
2018
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Statement of Operations Data:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Costs and expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
$
|
2,616,337
|
|
|
$
|
2,308,397
|
|
|
$
|
8,996,058
|
|
|
$
|
6,835,229
|
|
|
General and administrative
|
|
|
1,342,318
|
|
|
|
1,321,415
|
|
|
|
5,182,566
|
|
|
|
4,820,073
|
|
|
Impairment of goodwill
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,675,462
|
|
|
Total costs and expenses
|
|
|
3,958,655
|
|
|
|
3,629,812
|
|
|
|
14,178,624
|
|
|
|
13,330,764
|
|
|
Loss from operations
|
|
|
(3,958,655
|
)
|
|
|
(3,629,812
|
)
|
|
|
(14,178,624
|
)
|
|
|
(13,330,764
|
)
|
|
Other income (expense):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(Loss)/gain on revaluation of derivative warrants
|
|
|
—
|
|
|
|
(4,000
|
)
|
|
|
43,000
|
|
|
|
62,050
|
|
|
Interest income, net
|
|
|
1,047
|
|
|
|
12,171
|
|
|
|
42,712
|
|
|
|
29,687
|
|
|
Total other income, net
|
|
|
1,047
|
|
|
|
8,171
|
|
|
|
85,712
|
|
|
|
91,737
|
|
|
Net loss
|
|
$
|
(3,957,608
|
)
|
|
$
|
(3,621,641
|
)
|
|
$
|
(14,092,912
|
)
|
|
$
|
(13,239,027
|
)
|
|
Deemed dividend on preferred stock
|
|
|
|
|
|
|
|
|
|
|
—
|
|
|
|
(2,241,795
|
)
|
|
Net loss attributable to common stockholders
|
|
|
—
|
|
|
|
—
|
|
|
|
(14,092,912
|
)
|
|
|
(15,480,822
|
)
|
|
Basic and diluted net loss attributable to common stockholders per common share
|
|
$
|
(0.42
|
)
|
|
$
|
(0.76
|
)
|
|
$
|
(1.84
|
)
|
|
$
|
(5.23
|
)
|
|
Shares used in computing basic and diluted net loss attributable to common stockholders per common share
|
|
|
9,389,661
|
|
|
|
4,773,500
|
|
|
|
7,675,092
|
|
|
|
2,961,972
|
|
|
|
|
March 31, 2020
|
|
|
December 31,
|
|
|
|
|
(Unaudited)
|
|
|
2019
|
|
|
2018
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance Sheet Data:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current assets
|
|
$
|
7,863,436
|
|
|
$
|
11,385,673
|
|
|
$
|
13,951,834
|
|
|
Working capital
|
|
|
4,812,784
|
|
|
|
8,615,915
|
|
|
|
12,329,712
|
|
|
Total assets
|
|
|
8,689,549
|
|
|
|
12,250,811
|
|
|
|
15,054,082
|
|
|
Total liabilities
|
|
|
3,443,602
|
|
|
|
3,191,402
|
|
|
|
1,793,121
|
|
|
Total stockholders’ equity
|
|
|
5,245,947
|
|
|
|
9,059,409
|
|
|
|
13,260,961
|
|
RISK FACTORS
An investment in our securities involves
a high degree of risk. You should carefully consider the following risk factors, together with the other information about these
risks contained in this prospectus, as well as the other information contained in this prospectus generally, before deciding to
buy our securities. Any of the risks we describe below could adversely affect our business, financial condition, operating results,
or prospects. The market price for our securities could decline if one or more of these risks and uncertainties develop into actual
events and you could lose all or part of your investment. Additional risks and uncertainties that we do not yet know of, or that
we currently think are immaterial, may also impair our business operations. You should also refer to the other information contained
in this prospectus, including our financial statements and the related notes.
Risks Related to Our Business and Industry
Our operations and financial condition may be adversely
impacted by the COVID-19 pandemic.
In December 2019, a strain of novel coronavirus,
or COVID-19, was first reported in Wuhan, China, resulting in thousands of confirmed cases of the disease in China. By January,
the Chinese government implemented a quarantine protocol for Wuhan and implemented other restrictions for other major Chinese cities,
including mandatory business closures, social distancing measures, and various travel restrictions, all of which have subsequently
been adopted in countries throughout the world. On March 11, 2020, as COVID-19 spread outside of China, the World Health
Organization designated the outbreak as a global pandemic. This pandemic could affect our business, employees, operating results,
ability to obtain additional funding, product development programs, research and development programs, suppliers and third-party
manufacturers.
We anticipate that COVID-19 and
a prolonged public health crisis may negatively impact our financial condition and operating results; however, given the evolving
health, economic, social, and governmental environments, the breadth and duration of the impact remains uncertain. Due to the pandemic,
our clinical trial recruiting and participants and supply chain could also be slowed or delayed, or in a more severe scenario,
our business, financial condition and operating results could be more severely affected. Given the dynamic nature of these circumstances,
the duration of any business disruption or potential impact to our business resulting from the COVID-19 coronavirus is difficult
to predict, but it may increase our costs or expenses.
The potential effects of the COVID-19 pandemic
could impact many of our risk factors, included in Part 1, Item A of our 2019 Form 10-K, However, given the evolving health, economic,
social, and governmental environments, the potential impact that the COVID-19 pandemic could have on our risk factors that are
described in our 2019 Form 10-K remain uncertain.
We will require additional capital in order
to continue our operations and may have difficulty raising additional capital.
We expect that we will continue to generate
significant operating losses for the foreseeable future. At March 31, 2020, our consolidated cash balance was approximately $7.1
million. We believe our cash balance at March 31, 2020, is adequate to fund operations at budgeted levels into the first quarter
of 2021. We will require additional funds to conduct research and development, establish and conduct clinical and preclinical trials,
establish commercial-scale manufacturing arrangements and provide for the marketing and distribution of our products. Our ability
to execute our operating plan depends on our ability to obtain additional funding via the sale of equity and/or debt securities,
a strategic transaction or otherwise. We continue to actively pursue financing alternatives. However, there can be no assurance
that we will obtain the necessary funding in the amounts we seek or that it will be available on a timely basis or upon terms acceptable
to us. If we obtain capital by issuing debt or preferred stock, the holders of such securities would likely obtain rights that
are superior to those of holders of our common stock.
Our capital requirements and our ability
to meet them depend on many factors, including:
|
|
·
|
current and future impacts of the COVID-19 pandemic on all aspects of our business;
|
|
|
·
|
the number of potential products and technologies in development;
|
|
|
·
|
continued progress and cost of our research and development programs;
|
|
|
·
|
progress with preclinical studies and clinical trials;
|
|
|
·
|
the time and costs involved in obtaining regulatory clearance;
|
|
|
·
|
costs involved in preparing, filing, prosecuting, maintaining and enforcing patent claims;
|
|
|
·
|
costs of developing sales, marketing and distribution channels and our ability to sell our drugs;
|
|
|
·
|
costs involved in establishing manufacturing capabilities for clinical trial and commercial quantities of our drugs;
|
|
|
·
|
competing technological and market developments;
|
|
|
·
|
Claims or enforcement actions with respect to our products or operations:
|
|
|
·
|
market acceptance of our products;
|
|
|
·
|
costs for recruiting and retaining management, employees and consultants;
|
|
|
·
|
Our ability to manage computer system failures or security breaches;
|
|
|
·
|
costs for educating physicians regarding the application and use of our products;
|
|
|
·
|
whether we are able to maintain our listing on a national exchange;
|
|
|
·
|
uncertainty and economic instability resulting from conflicts, military actions, terrorist attacks, natural disasters, public
health crises, including the occurrence of a contagious disease or illness, such as the COVID-19 pandemic, cyber-attacks and general
instability; and
|
|
|
·
|
the condition of capital markets and the economy generally, both in the U.S. and globally.
|
We may consume available resources more
rapidly than currently anticipated, resulting in the need for additional funding sooner than expected. We may seek to raise any
necessary additional funds through the issuance of warrants, equity or debt financings or executing collaborative arrangements
with corporate partners or other sources, which may be dilutive to existing stockholders or have a material effect on our current
or future business prospects. In addition, in the event that additional funds are obtained through arrangements with collaborative
partners or other sources, we may have to relinquish economic and/or proprietary rights to some of our technologies or products
under development that we would otherwise seek to develop or commercialize by ourselves. If we cannot secure adequate financing
when needed, we may be required to delay, scale back or eliminate one or more of our research and development programs or to enter
into license or other arrangements with third parties to commercialize products or technologies that we would otherwise seek to
develop ourselves and commercialize ourselves. In such an event, our business, prospects, financial condition, and results of operations
may be adversely affected.
We have incurred net losses and negative
cash flows since inception. We currently have no product revenues, and may not succeed in developing or commercializing any products
that will generate product or licensing revenues. We do not expect to have any products on the market for several years. Our primary
activity to date has been research and development and conducting clinical trials. Development of our product candidates requires
a process of preclinical and clinical testing, during which our product candidates could fail. We may not be able to enter into
agreements with one or more companies experienced in the manufacturing and marketing of therapeutic drugs and, to the extent that
we are unable to do so, we may not be able to market our product candidates. Whether we achieve profitability or not will depend
on our success in developing, manufacturing, and marketing our product candidates. We have experienced net losses and negative
cash flows from operating activities since inception and we expect such losses and negative cash flows to continue for the foreseeable
future. As of March 31, 2020, we had a stockholders’ equity of approximately $5,246,000. The net loss for the three months
ended March 31, 2020 was approximately $3,958,000, and we may never achieve profitability.
We are a clinical stage biopharmaceutical company
with a going concern qualification to our financial statements and a history of losses, and we can provide no assurance as to our
future operating results.
We are a clinical stage biopharmaceutical
company and have experienced net losses and negative cash flows from operating activities since inception, and we expect such losses
and negative cash flows to continue for the foreseeable future. Whether or not we achieve profitability will depend on our success
in developing, manufacturing and marketing our product candidates. Our primary activity to date has been research and development
and conducting clinical studies. Development of our product candidates requires a process of preclinical and clinical testing during
which our product candidates could fail. We do not expect to have any products on the market for several years. We currently have
no product revenues and may not succeed in developing or commercializing any products that will generate product or licensing revenues.
We may not be able to enter into agreements with companies experienced in the manufacturing and marketing of therapeutic drugs
and, to the extent that we are unable to do so, we may not be able to market any product candidates.
As of March 31, 2020, we had working capital
of approximately $4.8 million and stockholders’ equity of approximately $5.2 million. For the period from our inception in
November 2002 until the business combination with Novelos Therapeutics, Inc. on April 8, 2011, and thereafter through March 31,
2020, we incurred aggregate net losses of approximately $115.6 million. The net loss for the year ended December 31, 2019, was
approximately $14.1 million. We may never achieve profitability.
Our financial statements as of December
31, 2019, were prepared under the assumption that we will continue as a going concern. The independent registered public accounting
firm that audited our 2019 financial statements, in its report, included an explanatory paragraph referring to our recurring losses
since inception and expressed substantial doubt in our ability to continue as a going concern. Our financial statements do not
include any adjustments that might result from the outcome of this uncertainty. Our ability to continue as a going concern depends
on our ability to obtain additional equity or debt financing, attain further operating efficiencies, reduce expenditures, and ultimately
generate revenue.
We rely on a collaborative outsourced business
model, and disruptions with our third-party collaborators, including potential disruptions at our sole source supplier of CLR 131,
Centre for Probe Development and Commercialization, CPDC, may impede our ability to gain FDA approval and delay or impair commercialization
of any products.
We are in the preclinical and clinical
study phases of product development and commercialization. We have closed manufacturing operations located at our corporate headquarters,
and have implemented a collaboration outsourcing model to more efficiently manage costs. We rely significantly on contracts with
third parties to use their facilities to conduct our research, development and manufacturing.
We have engaged CPDC, which has been a
validated cGMP manufacturing organization specializing in radiopharmaceuticals, as our exclusive source to supply drug product
for our ongoing research and clinical studies, including our Phase 1 and Phase 2 studies of CLR 131.
In addition, we rely exclusively on contract
research organizations to conduct research and development. Any inability of these organizations to fulfill the requirements of
their agreements with us may delay or impair our ability to gain FDA approval and commercialization of our drug delivery technology
and products.
Our reliance on third-party collaborators
exposes us to risks related to not being able to directly oversee the activities of these parties. Furthermore, these collaborators,
whether foreign or domestic, may experience regulatory compliance difficulties, mechanical shutdowns, employee strikes, or other
unforeseeable acts that may delay fulfillment of their agreements with us. Failure of any of these collaborators to provide the
required services in a timely manner or on commercially reasonable terms could materially delay the development and approval of
our products, increase our expenses, and materially harm our business, prospects, financial condition and results of operations.
We believe that we have a good working
relationship with our third-party collaborators. However, should the situation change, we may be required to relocate these activities
on short notice, and we do not currently have access to alternate facilities to which we could relocate our research, development
and/or manufacturing activities. The cost and time to establish or locate an alternate research, development and/or manufacturing
facility to develop our technology would be substantial and would delay obtaining FDA approval and commercializing our products.
Furthermore, if our products are approved
for commercial sale, we will need to work with our existing third-party collaborators to ensure sufficient capacity, or engage
additional parties with the capacity, to commercially manufacture our products in accordance with FDA and other regulatory requirements.
There can be no assurance that we would be able to successfully establish any such capacity or identify suitable manufacturing
partners on acceptable terms.
We rely on a small number of key personnel
who may terminate their employment with us at any time, and our success will depend on our ability to hire additional qualified
personnel.
Our success depends to a significant degree
on the continued services of our executive officers, including our Chief Executive Officer, James V. Caruso. Our management and
other employees may voluntarily terminate their employment with us at any time, and there can be no assurance that these individuals
will continue to provide services to us. Our success will depend on our ability to attract and retain highly skilled personnel.
We may be unable to recruit such personnel on a timely basis, if at all. The loss of services of key personnel, or the inability
to attract and retain additional qualified personnel, could result in delays in development or approval of our products, loss of
sales and diversion of management resources.
We cannot assure the successful development
and commercialization of our compounds in development.
At present, our success is dependent on
one or more of the following to occur: the successful development of CLR 131 for the treatment of a hematologic or solid tumor
cancer including multiple myeloma and B-Cell lymphomas or the treatment of pediatric solid tumors and lymphomas; the development
of new PDCs, specifically new products developed from our PDC program, and the advancement of our PDC agents through research and
development; and/or commercialization partnerships.
We are a biopharmaceutical company focused
on the discovery, development and commercialization of drugs for the treatment of cancer. We leverage our PDC platform to specifically
target treatments to cancer cells. The PDC platform possesses the potential for the discovery and development of the next generation
of cancer-targeting agents. The PDC platform features include the capacity to link with almost any molecule, the delivery of a
significant increase in targeted oncologic payload, and the ability to target all tumor cells. As a result, we believe that we
can generate PDCs to treat a broad range of cancers with the potential to improve the therapeutic index of oncologic drug payloads,
enhance or maintain efficacy while reducing adverse events by minimizing drug delivery to healthy cells, and increase delivery
to cancerous cells and cancer stem cells.
Our proposed products and their potential
applications are in an early stage of clinical and manufacturing/process development and face a variety of risks and uncertainties,
including the following:
|
|
·
|
Future clinical study results may show that our cancer-targeting and delivery technologies are not well-tolerated by patients at their effective doses or are not efficacious.
|
|
|
·
|
Future clinical study results may be inconsistent with testing results obtained to-date.
|
|
|
·
|
Even if our cancer-targeting and delivery technologies are shown to be safe and effective for their intended purposes, we may face significant or unforeseen difficulties in obtaining or manufacturing sufficient quantities at reasonable prices or at all.
|
|
|
·
|
Our ability to complete the development and commercialization of our cancer-targeting and delivery technologies for their intended use is substantially dependent upon our ability to raise sufficient capital or to obtain and maintain experienced and committed partners to assist us with obtaining clinical and regulatory approvals for, and the manufacturing, marketing and distribution of, our products.
|
|
|
·
|
Even if our cancer-targeting and delivery technologies are successfully developed, approved by all necessary regulatory authorities, and commercially produced, there is no guarantee that there will be market acceptance of our products.
|
|
|
·
|
Our competitors may develop therapeutics or other treatments that are superior or less costly than our own with the result that our product candidates, even if they are successfully developed, manufactured and approved, may not generate sufficient revenues to offset the development and manufacturing costs of our product candidates.
|
If we are unsuccessful in dealing with
any of these risks, or if we are unable to successfully advance the development of our cancer-targeting and delivery technologies
for some other reason, our business, prospects, financial condition and results of operations may be adversely affected.
Failure to complete the development of our
technologies, obtain government approvals, including required FDA approvals, or comply with ongoing governmental regulations could
prevent, delay or limit introduction or sale of proposed products and result in failure to achieve revenues or maintain our ongoing
business.
Our research and development activities
and the manufacture and marketing of our intended products are subject to extensive regulation for safety, efficacy and quality
by numerous government authorities in the U.S. and abroad. Before receiving approval to market our proposed products by the FDA,
we will have to demonstrate that our products are safe and effective for the patient population for the diseases that are to be
treated. Clinical studies, manufacturing and marketing of drugs are subject to the rigorous testing and approval process of the
FDA and equivalent foreign regulatory authorities. The Federal Food, Drug, and Cosmetic Act and other federal, state and foreign
statutes and regulations govern and influence the testing, manufacturing, labeling, advertising, distribution and promotion of
drugs and medical devices. As a result, clinical studies and regulatory approval can take many years to accomplish and require
the expenditure of substantial financial, managerial and other resources.
In addition to the required regulatory
approval described above, in order to be commercially viable, we must successfully research, develop, manufacture, introduce, market
and distribute our technologies. This includes meeting a number of critical developmental milestones, including:
|
|
·
|
demonstrating benefit from delivery of each specific drug for specific medical indications;
|
|
|
·
|
demonstrating through preclinical and clinical studies that each drug is safe and effective; and
|
|
|
·
|
demonstrating that we have established viable FDA cGMPs capable of potential scale-up.
|
The timeframe necessary to achieve these
developmental milestones may be long and uncertain, and we may not successfully complete these milestones for any of our intended
products in development.
In addition to the risks previously discussed,
our technology is subject to developmental risks that include the following:
|
|
·
|
uncertainties arising from the rapidly growing scientific aspects of drug therapies and potential treatments;
|
|
|
·
|
uncertainties arising as a result of the broad array of alternative potential treatments related to cancer and other diseases; and
|
|
|
·
|
expense and time associated with the development and regulatory approval of treatments for cancer and other diseases.
|
In order to conduct the clinical studies
that are necessary to obtain approval by the FDA to market a product, it is necessary to receive clearance from the FDA to conduct
such clinical studies. The FDA can halt clinical studies at any time for safety reasons or because we or our clinical investigators
do not follow the FDA’s requirements for conducting clinical studies. If any of our studies are halted, we will not be able
to obtain FDA approval until and unless we can address the FDA’s concerns. If we are unable to receive clearance to conduct
clinical studies for a product, we will not be able to achieve any revenue from that product in the U.S., as it is illegal to sell
any drug for use in humans in the U.S. without FDA approval.
Even if we do ultimately receive FDA approval
for any of our products, these products will be subject to extensive ongoing regulation, including regulations governing manufacturing,
labeling, packaging, testing, dispensing, prescription and procurement quotas, record keeping, reporting, handling, shipment and
disposal of any such drug. Failure to obtain and maintain required registrations or to comply with any applicable regulations could
further delay or preclude development and commercialization of our drugs and subject us to enforcement action.
Clinical studies involve a lengthy and expensive
process with an uncertain outcome, and results of earlier studies may not be predictive of future study results.
In order to obtain regulatory approval
for the commercialization of our product candidates, we must conduct, at our own expense, extensive clinical studies to demonstrate
safety and efficacy of these product candidates. Clinical testing is expensive, it can take many years to complete, and its outcome
is uncertain. Failure can occur at any time during the clinical study process.
We may experience delays in clinical testing
of our product candidates. We do not know whether planned clinical studies will begin on time, need to be redesigned, or be completed
on schedule, if at all. Clinical studies can be delayed for a variety of reasons, including delays in obtaining regulatory approval
to commence a study, reaching agreement on acceptable clinical study terms with prospective sites, obtaining institutional review
board approval to conduct a study at a prospective site, recruiting patients to participate in a study, or obtaining sufficient
supplies of clinical study materials. Many factors affect patient enrollment, including the size of the patient population, the
proximity of patients to clinical sites, the eligibility criteria for the study, competing clinical studies, and new drugs approved
for the conditions we are investigating. Prescribing physicians will also have to decide to use our product candidates over existing
drugs that have established safety and efficacy profiles or other drugs undergoing development in clinical studies. Any delays
in completing our clinical studies will increase our costs, slow down our product development and approval process, and delay our
ability to generate revenue.
In addition, the results of preclinical
studies and early clinical studies of our product candidates do not necessarily predict the results of later-stage clinical studies.
Product candidates in later stages of clinical studies may fail to show the desired safety and efficacy traits despite having progressed
through initial clinical testing. The data collected from clinical studies of our product candidates may not be sufficient to support
the submission of an NDA or to obtain regulatory approval in the U.S. or elsewhere. Because of the uncertainties associated with
drug development and regulatory approval, we cannot determine if or when we will have an approved product for commercialization
or will achieve sales or profits.
Our clinical studies may not demonstrate
sufficient levels of efficacy necessary to obtain the requisite regulatory approvals for our drugs, and our proposed drugs may
not be approved for marketing.
We may be required to suspend or discontinue
clinical studies due to unexpected side effects or other safety risks that could preclude approval of our product candidates.
Our clinical studies may be suspended at
any time for a number of reasons. For example, we may voluntarily suspend or terminate our clinical studies if at any time we believe
that they present an unacceptable risk to the clinical study patients. In addition, regulatory agencies may order the temporary
or permanent discontinuation of our clinical studies at any time if they believe that the clinical studies are not being conducted
in accordance with applicable regulatory requirements or that they present an unacceptable safety risk to the clinical study patients.
Administering any product candidates to
humans may produce undesirable side effects. These side effects could interrupt, delay or halt clinical studies of our product
candidates and could result in the FDA or other regulatory authorities denying further development or approval of our product candidates
for any or all targeted indications. Ultimately, some or all of our product candidates may prove to be unsafe for human use. Moreover,
we could be subject to significant liability if any volunteer or patient suffers, or appears to suffer, adverse health effects
as a result of participating in our clinical studies.
Controls we or our third-party collaborators
have in place to ensure compliance with all applicable laws and regulations may not be effective.
We and our third-party collaborators are
subject to federal, state and local laws and regulations governing the storage, use and disposal of hazardous materials and waste
products. Current or future regulations may impair our research, development, manufacturing and commercialization efforts. The
inability of our third-party collaborators to maintain the required licenses and permits for any reason will negatively impact
our manufacturing, research and development activities. In addition, we may be required to indemnify third-party collaborators
against certain liabilities arising out of any failure by them to comply with such regulations and/or laws. If we or our third
party collaborators fail to comply with any of these regulations and/or laws, a range of consequences could result, including the
suspension or termination of clinical studies, failure to obtain approval of a product candidate, restrictions on our products
or manufacturing processes, withdrawal of our products from the market, significant fines, exclusion from government healthcare
programs, or other sanctions or litigation.
We expect to rely on our patents as well as
specialized regulatory designations such as orphan drug classification for our product candidates, but regulatory drug designations
may not confer marketing exclusivity or other expected commercial benefits.
We expect to file for ODD or other regulatory
designations (fast track, break-through, priority review, etc.) as appropriate for our product candidates. Orphan drug status confers
seven years of marketing exclusivity under the Federal Food, Drug, and Cosmetic Act in the U.S., and up to ten years of marketing
exclusivity in Europe for a particular product in a specified indication. We have been granted ODD in the U.S. for CLR 131 as a
therapeutic for the treatment of multiple myeloma, neuroblastoma, osteosarcoma, rhabdomyosarcoma, Ewing’s sarcoma and lymphoplasmacytic
lymphoma. While we have been granted this orphan designation, we will not be able to rely on it to exclude other companies from
manufacturing or selling products using the same principal molecular structural features for the same indication beyond these timeframes
without our patent portfolio. For any product candidate for which we have been or will be granted ODD in a particular indication,
it is possible that another company also holding ODD for the same product candidate will receive marketing approval for the same
indication before we do. If that were to happen, our applications for that indication may not be approved until the competing company’s
period of exclusivity expires. Even if we were the first to obtain marketing authorization for an orphan drug indication, there
are circumstances under which a competing product may be approved for the same indication during the seven-year period of marketing
exclusivity, such as if the later product is shown to be clinically superior to the orphan product or deemed a different product
than ours. Further, the seven-year marketing exclusivity would not prevent competitors from obtaining approval of the same product
candidate as ours for indications other than those in which we have been granted ODD, or for other indications if not for our patent
portfolio, or for the use of other types of products in the same indications as our orphan product. Furthermore, although the ODD
and exclusivity are in effect right now, the FDA has the authority to modify this assessment at any time.
The FDA has granted rare pediatric disease
designation, RPDD, to CLR 131 for treatment of neuroblastoma and rhabdomyosarcoma; however, we may not be able to realize any value
from such designation.
Our CLR 131 compound has received RPDD
designation from the FDA for the treatment of neuroblastoma, rhabdomyosarcoma, osteosarcoma and Ewing’s sarcoma. The FDA
defines a “rare pediatric disease” as a disease that affects fewer than 200,000 individuals in the U.S. primarily under
the age of 18 years old. Under the FDA’s Rare Pediatric Disease Priority Review Voucher program, upon the approval of an
NDA or a BLA for the treatment of a rare pediatric disease, the sponsor of such application would be eligible for a Rare Pediatric
Disease Priority Review Voucher that can be used to obtain priority review for a subsequent NDA or BLA. There is no assurance we
will receive a Rare Pediatric Disease Priority Review Voucher or that it will result in a faster development process, review or
approval for a subsequent marketing application. Further, this program has been subject to criticism, including by the FDA, and
it is possible that even if we obtain approval for CLR 131 and qualify for such a Priority Review Voucher, the program may no longer
be in effect at the time of approval. Also, although Priority Review Vouchers may be sold or transferred to third parties, there
is no guaranty that we will be able to realize any value if we were to sell a Priority Review Voucher.
We are exposed to product, clinical and preclinical
liability risks that could create a substantial financial burden should we be sued.
Our business exposes us to potential product
liability and other liability risks that are inherent in the testing, manufacturing and marketing of pharmaceutical products. In
addition, the use in our clinical studies of pharmaceutical products that we, or our current or potential collaborators, may develop
and then subsequently sell, may cause us to bear a portion of, or all, product liability risks. While we carry an insurance policy
covering up to $5,000,000 per occurrence and $5,000,000 in the aggregate for liability incurred in connection with such claims
should they arise, there can be no assurance that our insurance will be adequate to cover all situations. Moreover, there can be
no assurance that such insurance, or additional insurance if required, will be available or, if available, will be available on
commercially reasonable terms. Furthermore, our current and potential partners with whom we have collaborative agreements, or our
future licensees, may not be willing to indemnify us against these types of liabilities and may not themselves be sufficiently
insured or have a net worth sufficient to satisfy any product liability claims. A successful product liability claim or series
of claims brought against us could have a material adverse effect on our business, prospects, financial condition and results of
operations.
Acceptance of our products in the marketplace
is uncertain and failure to achieve market acceptance will prevent or delay our ability to generate revenues.
Our future financial performance will depend,
at least in part, on the introduction and customer acceptance of our proposed products. Even if approved for marketing by the necessary
regulatory authorities, our products may not achieve market acceptance. The degree of market acceptance will depend on a number
of factors including:
|
|
·
|
receiving regulatory clearance of marketing claims for the uses that we are developing;
|
|
|
·
|
establishing and demonstrating the advantages, safety and efficacy of our technologies;
|
|
|
·
|
pricing and reimbursement policies of government and third-party payors such as insurance companies, health maintenance organizations and other health plan administrators;
|
|
|
·
|
attracting corporate partners, including pharmaceutical companies, to assist in commercializing our intended products; and
|
|
|
·
|
marketing our products.
|
Physicians, patients, payors or the medical
community, in general, may be unwilling to accept, use or recommend any of our products. If we are unable to obtain regulatory
approval or commercialize and market our proposed products as planned, we may not achieve any market acceptance or generate revenue.
The market for our proposed products is rapidly
changing and competitive, and new therapeutics, drugs and treatments that may be developed by others could impair our ability to
develop our business or become competitive.
The pharmaceutical and biotechnology industries
are subject to rapid and substantial technological change. Developments by others may render our technologies and proposed products
noncompetitive or obsolete, or we may be unable to keep pace with technological developments or other market factors. Technological
competition from pharmaceutical and biotechnology companies, universities, governmental entities and others diversifying into the
field is intense and expected to increase. Most of these entities have significantly greater research and development capabilities
and budgets than we do, as well as substantially more marketing, manufacturing, financial and managerial resources. These entities
represent significant competition for us. Acquisitions of, or investments in, competing pharmaceutical or biotechnology companies
by large corporations could increase our competitors’ financial, marketing, manufacturing and other resources.
Our resources are limited, and we may experience
management, operational or technical challenges inherent in our activities and novel technologies. Competitors have developed,
or are in the process of developing, technologies that are, or in the future may be, the basis for competition. Some of these technologies
may accomplish therapeutic effects similar to those of our technology, but through different means. Our competitors may develop
drugs and drug delivery technologies that are more effective than our intended products and, therefore, present a serious competitive
threat to us.
The potential widespread acceptance of
therapies that are alternatives to ours may limit market acceptance of our products even if they are commercialized. Many of our
targeted diseases and conditions can also be treated by other medication or drug delivery technologies. These treatments may be
widely accepted in medical communities and have a longer history of use. The established use of these competitive drugs may limit
the potential for widespread acceptance of our technologies and products if commercialized.
We may face litigation from third parties claiming
our products infringe on their intellectual property rights, particularly because there is often substantial uncertainty about
the validity and breadth of medical patents.
We may be exposed to future litigation
by third parties based on claims that our technologies, products or activities infringe on the intellectual property rights of
others or that we have misappropriated the trade secrets of others. This risk is exacerbated by the fact that the validity and
breadth of claims covered in medical technology patents, and the breadth and scope of trade-secret protection, involve complex
legal and factual questions for which important legal principles are unresolved. Any litigation or claims against us, whether valid
or not, could result in substantial costs, place a significant strain on our financial and managerial resources, and harm our reputation.
License agreements that we may enter into in the future would likely require that we pay the costs associated with defending this
type of litigation. In addition, intellectual property litigation or claims could force us to do one or more of the following:
|
|
·
|
cease selling, incorporating or using any of our technologies and/or products that incorporate the challenged intellectual property, which would adversely affect our ability to generate revenue;
|
|
|
·
|
obtain a license from the holder of the infringed intellectual property right, which license may be costly or may not be available on reasonable terms, if at all; or
|
|
|
·
|
redesign our products, which would be costly and time-consuming.
|
If we are unable to adequately protect or enforce
our rights to intellectual property or to secure rights to third-party patents, we may lose valuable rights, experience reduced
market share, assuming any, or incur costly litigation to protect our intellectual property rights.
Our ability to obtain licenses to patents,
maintain trade-secret protection, and operate without infringing the proprietary rights of others will be important to commercializing
any products under development. Therefore, any disruption in access to the technology could substantially delay the development
of our technology.
The patent positions of biotechnology and
pharmaceutical companies, such as ours, for products that involve licensing agreements are frequently uncertain and involve complex
legal and factual questions. In addition, the coverage claimed in a patent application can be significantly reduced before the
patent is issued or in subsequent legal proceedings. Consequently, our patent applications and any issued and licensed patents
may not provide protection against competitive technologies or may be held invalid if challenged or circumvented. To the extent
we license patents from third parties, the early termination of any such license agreement would result in the loss of our rights
to use the covered patents, which could severely delay, inhibit or eliminate our ability to develop and commercialize compounds
based on the licensed patents. Our competitors may also independently develop products similar to ours or design around or otherwise
circumvent patents issued or licensed to us. In addition, the laws of some foreign countries may not protect our proprietary rights
to the same extent as U.S. law.
We also rely on trade secrets, technical
know-how and continuing technological innovation to develop and maintain our competitive position. Although we generally require
our employees, consultants, advisors and collaborators to execute appropriate confidentiality and assignment-of-inventions agreements,
our competitors may independently develop substantially equivalent proprietary information and techniques, reverse engineer our
information and techniques, or otherwise gain access to our proprietary technology. We may be unable to meaningfully protect our
rights in trade secrets, technical know-how and other nonpatented technology.
We may have to resort to litigation to
protect our rights for certain intellectual property or to determine the scope, validity or enforceability of our intellectual
property rights. Enforcing or defending our rights would be expensive, could cause diversion of our resources, and may not prove
successful. Any failure to enforce or protect our rights could cause us to lose the ability to exclude others from using our technology
to develop or sell competing products.
Conflicts, military actions, terrorist attacks,
natural disasters, public health crises, including the occurrence of a contagious disease or illness, such as the COVID-19 coronavirus,
cyber-attacks and general instability could adversely affect our business.
Conflicts, military actions, terrorist
attacks, natural disasters and public health crises have precipitated economic instability and turmoil in financial markets. Instability
and turmoil may result in raw material cost increases. The uncertainty and economic disruption resulting from hostilities, military
action, acts of terrorism, public health crises or cyber-attacks may impact our operations or those of our suppliers. Accordingly,
any conflict, military action, terrorist attack, public health crises or cyber-attack that impacts us or any of our suppliers,
could have a material adverse effect on our business, liquidity, prospects, financial condition and results of operations.
Confidentiality agreements with employees and
others may not adequately prevent disclosure of our trade secrets and other proprietary information and may not adequately protect
our intellectual property, which could limit our ability to compete.
We operate in the highly technical field
of research and development of small-molecule drugs and rely, in part, on trade-secret protection in order to protect our proprietary
trade secrets and unpatented know-how. However, trade secrets are difficult to protect, and we cannot be certain that our competitors
will not develop the same or similar technologies on their own. We have taken steps, including entering into confidentiality agreements
with our employees, consultants, outside scientific collaborators, sponsored researchers and other advisors, to protect our trade
secrets and unpatented know-how. These agreements generally require that the other party keep confidential and not disclose to
third parties all confidential information developed by the party or made known to the party by us during the course of the party’s
relationship with us. Also, we typically obtain agreements from these parties that inventions conceived by them in the course of
rendering services to us will be our exclusive property. However, these agreements may not be honored and may not effectively assign
intellectual property rights to us. Enforcing a claim that a party has illegally obtained, and is using our trade secrets or know-how,
is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, courts outside the U.S. may be less
willing to protect trade secrets or know-how. The failure to obtain or maintain trade-secret protection could adversely affect
our competitive position.
We may be subject to claims that our employees
have wrongfully used or disclosed alleged trade secrets of their current or former employers.
As is common in the biotechnology and pharmaceutical
industry, we engage individuals who were previously employed at other biotechnology or pharmaceutical companies, including our
competitors or potential competitors or who are employed by academic research institutions. Although no claims against us are currently
pending, we may be subject to claims that we, or these employees, have used or disclosed trade secrets or other proprietary information
of their current or former employers, either inadvertently or otherwise. Litigation may be necessary to defend against these claims.
Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction
to management.
Due to continued changes in marketing, sales
and distribution, we may be unsuccessful in our efforts to sell our proposed products, develop a direct sales organization, or
enter into relationships with third parties.
We have not established marketing, sales
or distribution capabilities for our proposed products. Until such time as our proposed products are further along in the development
process, we will not devote any meaningful time and resources to this effort. At the appropriate time, we will determine whether
we will develop our own sales and marketing capabilities or enter into agreements with third parties to sell our products.
We have limited experience in developing,
training or managing a sales force. If we choose to establish a direct sales force, we may incur substantial additional expenses
in developing, training and managing such an organization. We may be unable to build a sales force on a cost-effective basis or
at all. In addition, we will compete with many other companies that currently have extensive marketing and sales operations. Our
marketing and sales efforts may be unable to compete against these other companies. We may be unable to establish a sufficient
sales and marketing organization on a cost-effective or timely basis, if at all.
If we choose to enter into agreements with
third parties to sell our proposed products, we may be unable to establish or maintain third-party relationships on a commercially
reasonable basis, if at all. In addition, these third parties may have similar or more established relationships with our competitors.
We may be unable to engage qualified distributors.
Even if engaged, these distributors may:
|
|
·
|
fail to adequately market our products;
|
|
|
·
|
fail to satisfy financial or contractual obligations to us;
|
|
|
·
|
offer, design, manufacture or promote competing products; or
|
|
|
·
|
cease operations with little or no notice.
|
If we fail to develop sales, marketing
and distribution channels, we would experience delays in product sales and incur increased costs, which would have a material adverse
effect on our business, prospects, financial condition and results of operation.
If we are unable to convince physicians of
the benefits of our intended products, we may incur delays or additional expense in our attempt to establish market acceptance.
Achieving use of our products in the target
market of cancer diagnosis and treatment may require physicians to be informed regarding these products and their intended benefits.
The time and cost of such an educational process may be substantial. Inability to successfully carry out this physician education
process may adversely affect market acceptance of our proposed products. We may be unable to educate physicians, in sufficient
numbers, in a timely manner regarding our intended proposed products to achieve our marketing plans and product acceptance. Any
delay in physician education may materially delay or reduce demand for our proposed products. In addition, we may expend significant
funds towards physician education before any acceptance or demand for our proposed products is created, if at all.
If users of our products are unable to obtain
adequate reimbursement from third-party payors, or if additional healthcare reform measures are adopted, it could hinder or prevent
the commercial success of our product candidates.
The continuing efforts of government and
insurance companies, health maintenance organizations and other payors of healthcare costs to contain or reduce costs of healthcare
may adversely affect our ability to generate future revenues and achieve profitability, including by limiting the future revenues
and profitability of our potential customers, suppliers and collaborative partners. For example, in certain foreign markets pricing
or profitability of prescription pharmaceuticals are subject to government control. The U.S. government is implementing, and other
governments have shown significant interest in pursuing, healthcare reform. Any government-adopted reform measures could adversely
affect the pricing of healthcare products and services in the U.S. or internationally and the amount of reimbursement available
from governmental agencies or other third-party payors. The continuing efforts of the U.S. and foreign governments, insurance companies,
managed care organizations, and other payors of healthcare services to contain or reduce healthcare costs may adversely affect
our ability to set prices for our products, should we be successful in commercializing them, and this would negatively affect our
ability to generate revenues and achieve and maintain profitability.
New laws, regulations and judicial decisions,
or new interpretations of existing laws, regulations and decisions, that relate to healthcare availability, methods of delivery
or payment for healthcare products and services, or sales, marketing or pricing of healthcare products and services may also limit
our potential revenue and may require us to revise our research and development programs. The pricing and reimbursement environment
may change in the future and become more challenging for several reasons, including policies advanced by the current or future
executive administrations in the U.S., new healthcare legislation, or fiscal challenges faced by government health administration
authorities. Specifically, in both the U.S. and some foreign jurisdictions, there have been a number of legislative and regulatory
proposals to change the healthcare system in ways that could affect our ability to sell our products profitably. In the U.S., changes
in the federal healthcare policy were enacted in 2010 and are being implemented. Some reforms could result in reduced reimbursement
rates for our product candidates, which would adversely affect our business strategy, operations and financial results. Our ability
to commercialize our products will depend in part on the extent to which appropriate reimbursement levels for the cost of our products
and related treatment are obtained by governmental authorities, private health insurers, and other organizations such as health
maintenance organizations (“HMOs”). Third-party payors are increasingly challenging the prices charged for medical
drugs and services. Also, the trend toward managed healthcare in the U.S. and the concurrent growth of organizations such as HMOs
that could control or significantly influence the purchase of healthcare services and drugs, as well as legislative proposals to
reform healthcare or change government insurance programs, may all result in lower prices for or rejection of our drugs. The cost
containment measures that healthcare payors and providers are instituting, and the effect of any healthcare reform, could materially
harm our ability to operate profitably.
Our business and operations may be materially
adversely affected in the event of computer system failures or security breaches.
Despite
the implementation of security measures, our internal computer systems, and those of our third-party manufacturers, contract research
organizations and other third parties on which we rely, are vulnerable to damage from computer viruses, unauthorized access, cyber-attacks,
natural disasters, fire, terrorism, war and telecommunication and electrical failures. If such an event were to occur and interrupt
our operations, it could result in a material disruption in our business. For example, the loss of clinical study data from ongoing
or planned clinical studies could result in delays in our regulatory approval efforts and significantly increase our costs to recover
or reproduce the data. To the extent that any disruption or security breach results in a loss of or damage to our data or applications,
loss of trade secrets, inappropriate disclosure of confidential or proprietary information, including protected health information
or personal data of employees or former employees, lack of access to our clinical data, or disruption of the manufacturing process,
we could incur liability and the further development of our drug candidates could be delayed. We may also be vulnerable to cyber-attacks
or other malfeasance by hackers. This type of breach of our cybersecurity may compromise our confidential and financial information,
adversely affect our business, or result in legal proceedings. Further, these cybersecurity breaches may inflict reputational harm
upon us that may result in decreased market value and erode public trust.
Failure to maintain effective internal controls
could adversely affect our ability to meet our reporting requirements.
We are required to establish and maintain
appropriate internal controls over financial reporting. Rules adopted by the SEC pursuant to Section 404 of the Sarbanes-Oxley
Act of 2002 require an annual assessment of internal controls over financial reporting and for certain issuers an attestation of
this assessment by the issuer’s independent registered public accounting firm. The standards to assess that our internal
controls over financial reporting are effective are evolving and complex, require significant documentation and testing, and may
require remediation if they are not met. We expect to incur significant expenses and to devote resources to Section 404 compliance
on an ongoing basis. It is difficult for us to predict how long it will take or costly it will be to complete the assessment of
the effectiveness of our internal control over financial reporting for each year and to remediate any deficiencies in our internal
control over financial reporting. As a result, we may not be able to complete the assessment and remediation process on a timely
basis. In addition, although attestation requirements by our independent registered public accounting firm are not presently applicable
to us, we could become subject to these requirements in the future, and we may encounter problems or delays in completing the implementation
of any resulting changes to internal controls over financial reporting.
Effective internal controls are necessary
for us to provide reasonable assurance with respect to our financial reports and to effectively prevent fraud. Failure to maintain
effective internal controls could adversely affect our public disclosures regarding our business, prospects, financial condition
or results of operations. In addition, management’s assessment of internal controls over financial reporting may identify
weaknesses and conditions that need to be addressed in our internal controls over financial reporting or other matters that may
raise concerns for investors. Any actual or perceived weaknesses and conditions that need to be addressed in our internal control
over financial reporting or disclosure of management’s assessment of our internal controls over financial reporting our business
and results of operations could be harmed, we could fail to meet our reporting obligations, and there could be a material adverse
effect on our common stock price.
Risks Related to Our Equity Securities
We have in the past received notices from Nasdaq
of noncompliance with its listing rules, and delisting with Nasdaq could impact the price of our common stock and our ability to
raise funds.
The failure to meet continuing compliance
standards subjects our common stock to delisting. We have not received any other notices of noncompliance with Nasdaq listing rules,
but we have received such notices as recently as 2016. Any future failure to comply with Nasdaq’s listing rules and any resulting
delisting from the Nasdaq would reduce the visibility, liquidity and price of our common
stock and could limit our ability to raise funds in the future.
Our stock price has experienced price fluctuations.
There can be no assurance that the market
price for our common stock will remain at its current level, and a decrease in the market price could result in substantial losses
for investors. The market price of our common stock may be significantly affected by one or more of the following factors:
|
|
·
|
announcements or press releases relating to the biopharmaceutical sector or to our own business or prospects;
|
|
|
·
|
regulatory, legislative or other developments affecting us or the healthcare industry generally;
|
|
|
·
|
sales by holders of restricted securities pursuant to effective registration statements or exemptions from registration;
|
|
|
·
|
market conditions specific to biopharmaceutical companies, the healthcare industry and the stock market generally; and
|
|
|
·
|
our ability to maintain our listing on the Nasdaq exchange.
|
Our common stock could be further diluted as
the result of the issuance of additional shares of common stock, convertible securities, warrants or options.
In the past, we have issued common stock,
convertible securities (such as convertible preferred stock and notes) and warrants in order to raise capital. We have also issued
equity as compensation for services and incentive compensation for our employees and directors. We have shares of common stock
reserved for issuance upon the exercise of certain of these securities and may increase the shares reserved for these purposes
in the future. Our issuance of additional common stock, convertible securities, options and warrants could dilute our common stock,
affect the rights of our stockholders, reduce the market price of our common stock, result in adjustments to exercise prices of
outstanding warrants (resulting in these securities becoming exercisable for, as the case may be, a greater number of shares of
our common stock), or obligate us to issue additional shares of common stock to certain of our stockholders.
Provisions of our certificate of incorporation,
by-laws, and Delaware law may make an acquisition of us or a change in our management more difficult.
Certain provisions of our certificate of
incorporation and by-laws could discourage, delay or prevent a merger, acquisition or other change in control that stockholders
may consider favorable, including transactions in which an investor might otherwise receive a premium for its shares. These provisions
also could limit the price that investors might be willing to pay in the future for shares of our common stock or warrants, thereby
depressing the market price of our common stock. Stockholders who wish to participate in these transactions may not have the opportunity
to do so.
Furthermore, these provisions could prevent
or frustrate attempts by our stockholders to replace or remove our management. These provisions:
|
|
·
|
provide for the division of the Board into three classes as nearly equal in size as possible with staggered three-year terms and further limit the removal of directors and the filling of vacancies;
|
|
|
·
|
authorize our Board to issue without stockholder approval blank-check preferred stock that, if issued, could operate as a “poison pill” to dilute the stock ownership of a potential hostile acquirer to prevent an acquisition that is not approved by our Board;
|
|
|
·
|
require that stockholder actions must be effected at a duly called stockholder meeting and prohibit stockholder action by written consent;
|
|
|
·
|
establish advance notice requirements for stockholder nominations to our Board or for stockholder proposals that can be acted on at stockholder meetings;
|
|
|
·
|
limit who may call stockholder meetings; and
|
|
|
·
|
require the approval of the holders of 75% of the outstanding shares of our capital stock entitled to vote in order to amend certain provisions of our certificate of incorporation and by-laws.
|
In addition, because we are incorporated
in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which may, unless certain
criteria are met, prohibit large stockholders, in particular those owning 15% or more of our outstanding voting stock, from merging
or combining with us for a prescribed period of time.
We have not paid dividends in the past and
do not expect to pay dividends for the foreseeable future. Any return on investment may be limited to the value of our common stock.
No cash dividends have been paid on our
common stock. We do not expect to pay cash dividends in the near future. Payment of dividends would depend upon our profitability
at the time, cash available for those dividends, and other factors as our Board may consider relevant. If we do not pay dividends,
our common stock may be less valuable because a return on an investor’s investment will occur only if our stock price appreciates.
Risks Related to this Offering
Our management team will have immediate and
broad discretion over the use of the net proceeds from this offering, and you may not agree with our use of the net proceeds.
The net proceeds from this offering will
be immediately available to our management to use at its discretion. We currently intend to use the net proceeds from this offering
to fund our research and development activities, general corporate purposes, and possibly for acquisitions of other companies,
products or technologies, although no such acquisitions are currently contemplated. See “Use of Proceeds.” We have
not allocated specific amounts of the net proceeds from this offering for any of the foregoing purposes. Accordingly, our management
will have significant discretion and flexibility in applying the net proceeds of this offering. You will be relying on the judgment
of our management with regard to the use of these net proceeds, and you will not have the opportunity, as part of your investment
decision, to assess whether the proceeds are being used appropriately. It is possible that the net proceeds will be invested in
a way that does not yield a favorable, or any, return for us or our stockholders. The failure of our management to use such funds
effectively could have a material adverse effect on our business, prospects, financial condition and results of operation.
You will experience immediate and substantial
dilution as a result of this offering and may experience additional dilution in the future.
You will incur immediate and substantial
dilution as a result of this offering. After giving effect to the sale by of the securities offered in this offering, at an assumed
public offering price of $ per share and warrant, and after deducting the
underwriter’s discounts and commissions and other estimated offering expenses payable by us, investors in this offering can
expect an immediate dilution of $ per share, or
%, at the assumed public offering price of per share and warrant, assuming
no exercise of the warrants. In addition, in the past, we issued options and warrants to acquire shares of common stock. To the
extent these options are ultimately exercised, you will sustain future dilution.
You may experience future dilution as a result
of future equity offerings.
In order to raise additional capital, in
the future we may offer additional shares of our common stock or other securities convertible into or exchangeable for our common
stock at prices that may not be the same as the price in this offering. We may sell shares or other securities in any other offering
at a price that is less than the price paid by investors in this offering, and investors purchasing shares or other securities
in the future could have rights superior to existing stockholders. The price at which we sell additional shares of our common stock,
or securities convertible or exchangeable into common stock, in future transactions may be higher or lower than the price paid
by investors in this offering.
The warrants issued in this offering
may not have any value.
Each warrant will have an exercise price
equal to $ and will expire on the five year anniversary of the date they first become
exercisable. In the event our common stock price does not exceed the exercise price of the warrants during the period when the
warrants are exercisable, the warrants may not have any value.
A warrant does not entitle the holder
to any rights as common stockholders until the holder exercises the warrant for shares of our common stock.
Until you acquire shares of our common
stock upon exercise of your warrants, the warrants will not provide you any rights as a common stockholder. Upon exercise of your
warrants, you will be entitled to exercise the rights of a common stockholder only as to matters for which the record date occurs
on or after the exercise date.
There is no public market for the
warrants or the pre-funded warrants being offered by us in this offering.
There is no established public trading
market for the warrants or the pre-funded warrants being offered in this offering, and we do not expect a market to develop. In
addition, we do not intend to apply to list the warrants or the pre-funded warrants on any national securities exchange or other
nationally recognized trading system, including The Nasdaq Capital Market. Without an active market, the liquidity of the warrants
and the pre-funded warrants will be limited.
USE OF PROCEEDS
Based on an assumed public offering price
of $ per share of common stock and warrant, which was the last reported sale price of our common
stock on the Nasdaq Capital Market on , 2020 we estimate that the net proceeds to us from the
sale of the securities that we are offering, assuming gross proceeds of $ million and no exercise
of the over-allotment option, will be approximately $ million, after deducting underwriting
discounts and commissions and estimated offering expenses. In addition, if all of the warrants offered pursuant to this prospectus
are exercised in full for cash, we will receive approximately an additional $ million in cash.
We expect to use any proceeds received
from this offering as follows:
|
|
·
|
research and development activities, including the further development of CLR 131, and the research advancement of our PDC
platform, including product candidates, CLR 1700, CLR 1800, CLR 1900, CLR 2000, CLR 2100, CLR 2200 series and CLR 12120.
|
|
|
·
|
general corporate purposes, such as human resource acquisition to support organizational priorities, general and administrative
expenses, capital expenditures, working capital, repayment of debt, prosecution and maintenance of our intellectual property, and
the potential investment in technologies, products or collaborations that complement our business.
|
Even if we sell all of the securities subject
to this offering, we will still need to obtain additional financing in the future in order to fully fund these product candidates
through the regulatory approval process. We may seek such additional financing through public or private equity or debt offerings
or other sources, including collaborative or other arrangements with corporate partners, and through government grants and contracts.
There can be no assurance we will be able to obtain additional financing. Although we currently anticipate that we will use the
net proceeds of this offering as described above, there may be circumstances when a reallocation of funds is necessary. The amounts
and timing of our actual expenditures will depend upon numerous factors, including the progress of our development and commercialization
efforts, the progress of our clinical studies, whether or not we enter into strategic collaborations or partnerships, and our operating
costs and expenditures. Accordingly, our management will have significant flexibility in applying the net proceeds of this offering.
The costs and timing of drug development
and regulatory approval, particularly conducting clinical studies, are highly uncertain, subject to substantial risks, and can
often change. Accordingly, we may change the allocation of use of these proceeds as a result of contingencies such as the progress
and results of our clinical studies and other development activities, the establishment of collaborations, our manufacturing requirements,
and regulatory or competitive developments.
Pending the application of the net proceeds
as described above or otherwise, we may invest the proceeds in short-term, investment-grade, interest-bearing securities or guaranteed
obligations of the U.S. government or other securities.
CAPITALIZATION
The following table sets forth our cash
and cash equivalents and capitalization, each as of March 31, 2020:
|
|
·
|
on an actual basis; and
|
|
|
·
|
on a pro forma basis, as adjusted to give effect to the issuance of the securities offered hereby.
|
You should consider this table in conjunction
with our financial statements and the notes to those financial statements incorporated by reference in this prospectus. The pro
forma as adjusted information set forth in the table below is illustrative only and will be adjusted based on the actual public
offering price and other terms of this offering determined at pricing.
|
|
|
As of March 31, 2020
|
|
|
|
|
(Unaudited)
|
|
|
|
|
|
|
|
Pro Forma,
|
|
|
|
|
Actual
|
|
|
As Adjusted
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
7,092,099
|
|
|
$
|
|
|
|
Current portion of lease liability
|
|
|
109,257
|
|
|
|
|
|
|
Total debt obligations
|
|
|
109,257
|
|
|
|
|
|
|
Stockholders’ equity:
|
|
|
|
|
|
|
|
|
|
Preferred stock, par value $0.00001 per share:
|
|
|
|
|
|
|
|
|
|
7,000 shares authorized; 215 actual; pro forma
|
|
|
1,148,204
|
|
|
|
|
|
|
Common stock, par value $0.00001 per share:
|
|
|
|
|
|
|
|
|
|
80,000,000 shares authorized; 9,396,015 actual; pro forma
|
|
|
94
|
|
|
|
|
|
|
Additional paid-in capital
|
|
|
119,736,512
|
|
|
|
|
|
|
Accumulated deficit
|
|
|
(115,638,863
|
)
|
|
|
|
|
|
Total stockholders’ equity
|
|
|
5,245,947
|
|
|
|
|
|
|
Total capitalization
|
|
$
|
5,355,204
|
|
|
$
|
|
|
The information set forth above is based
on 9,396,015 shares of common stock outstanding as of May 5, 2020, assumes no exercise by the underwriter of its over-allotment
option, and excludes, as of that date:
|
|
·
|
an aggregate of 884,464 shares of common stock issuable upon the exercise of outstanding stock options issued to employees,
directors and consultants;
|
|
|
·
|
an aggregate of 537,500 shares of common stock issuable upon the conversion of outstanding shares of Series C preferred stock
|
|
|
·
|
an aggregate of 9,268,352 additional shares of common stock reserved for issuance under outstanding warrants having expiration
dates between October 1, 2020, and October 14, 2024, and exercise prices ranging from $2.40 to $283.00 per share; and
|
|
|
·
|
shares of our common stock that may be issued upon the exercise of pre-funded warrants and Series H warrants issued in this
offering.
|
MARKET FOR COMMON EQUITY
Our common stock is quoted under the CLRB
ticker symbol on the Nasdaq Capital Market.
On May 5, 2020, there were 267 holders
of record of our common stock. This number does not include stockholders for whom shares were held in a “nominee” or
“street” name.
We have not declared or paid any cash dividends
on our common stock and do not anticipate declaring or paying any cash dividends in the foreseeable future. We currently expect
to retain future earnings, if any, for the continued development of our business.
Our transfer agent and registrar is American
Stock Transfer and Trust Company, 6201 15th Avenue, Brooklyn, NY 11219.
DILUTION
Our net tangible book value as of March
31, 2020, was approximately $5.2 million, or $0.56 per share of common stock, based upon 9,396,015 shares outstanding. Net tangible
book value per share is determined by dividing such number of outstanding shares of common stock into our net tangible book value,
which is our total tangible assets, less total liabilities.
After
giving effect to the sale of shares
of our common stock in this offering at the assumed offering price of $ per share, the last
reported sale price of our common stock on the Nasdaq Capital Market on
, 2020, and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us,
and assuming no exercise of the underwriter’s over-allotment option in full, our as adjusted net tangible book value as of
March 31, 2020 would have been approximately $ million, or $
per share. This represents an immediate decrease in net tangible book value of $ per
share to existing stockholders. Investors purchasing our common stock in this offering will have paid $ less
than the as adjusted net tangible book value per share after this offering. This represents an immediate increase in net
tangible book value of approximately $ per share to our existing stockholders, and an immediate
dilution of $ per share to investors purchasing securities in the offering.
The following table illustrates the per
share dilution to investors purchasing securities in the offering:
|
Assumed public offering price per share of common stock,
|
|
|
|
|
|
|
|
together with a warrant to purchase shares of common stock
|
|
|
|
|
$
|
|
|
Net tangible book value per share as of March 31, 2020
|
|
$
|
0.56
|
|
|
|
|
Decrease per share attributable to sale of securities to investors
|
|
$
|
|
|
|
|
|
Adjusted net tangible book value per share after the offering
|
|
|
|
|
$
|
|
|
Dilution per share to investors
|
|
|
|
|
$
|
|
The foregoing illustration does not reflect
potential dilution from the exercise of outstanding options or warrants to purchase shares of our common stock. The foregoing illustration
also does not reflect the dilution that would result from the exercise of the warrants sold in the offering. The dilution information
set forth in the table above is illustrative only and will be adjusted based on the actual public offering price and other terms
of this offering determined at pricing.
The information set forth above is based
on 9,396,015 shares of common stock outstanding as of May 5, 2020 and excludes, as of that date:
|
|
·
|
an aggregate of 884,464 shares of common stock issuable upon the exercise of outstanding stock options issued to employees,
directors and consultants;
|
|
|
·
|
an aggregate of 537,500 shares of common stock issuable upon the conversion of outstanding shares of Series C preferred stock
|
|
|
·
|
an aggregate of 9,268,352 additional shares of common stock reserved for issuance under outstanding warrants having expiration
dates between October 1, 2020, and October 14, 2024, and exercise prices ranging from $2.40 to $283.00 per share; and
|
|
|
·
|
shares of our common stock that may be issued upon the exercise of pre-funded warrants and Series H warrants issued in this
offering.
|
BUSINESS
Business Overview
We are a clinical stage biopharmaceutical
company focused on the discovery, development and commercialization of drugs for the treatment of cancer. We are developing proprietary
drugs independently and through research and development collaborations. Our core objective is to leverage our proprietary phospholipid
drug conjugate™ (PDC™) delivery platform to develop PDCs that are designed to specifically target cancer cells, and
deliver improved efficacy and better safety as a result of fewer off-target effects. Our PDC platform possesses the potential for
the discovery and development of the next generation of cancer-targeting treatments, and we plan to develop PDCs both independently
and through research and development collaborations. The COVID-19 pandemic has created uncertainties in the expected timelines
for clinical stage biopharmaceutical companies such as us, and because of such uncertainties, it is difficult for us to accurately
predict expected outcomes at this time. We have continued to enroll patients in our clinical trials. However, In addition, COVID-19
may impact our ability to recruit patients for clinical trials, obtain adequate supply of CLR 131 and obtain additional financing.
Our lead PDC therapeutic, CLR 131 is a
small-molecule PDC designed to provide targeted delivery of iodine-131 directly to cancer cells, while limiting exposure to healthy
cells. We believe this profile differentiates CLR 131 from many traditional on-market treatment options. CLR 131 is the company’s
lead product candidate and is currently being evaluated in a Phase 2 study in relapsed/refractory (r/r) malignancies, including
multiple myeloma (MM), chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), lymphoplasmacytic lymphoma/Waldenstrom’s
macroglobulinemia (LPL/WM), marginal zone lymphoma (MZL), mantle cell lymphoma (MCL), and diffuse large B-cell lymphoma (DLBCL).
CLR 131 is also being evaluated in two Phase 1 dose escalation studies, one in r/r MM and one in pediatric solid tumors and lymphoma.
The U.S. Food and Drug Administration (“FDA”) granted CLR 131 Fast Track Designation for both r/r MM and r/r DLBCL
and Orphan Drug Designation (ODD) of MM, LPL/WM, neuroblastoma, rhabdomyosarcoma, Ewing’s sarcoma and osteosarcoma. CLR 131
was also granted Rare Pediatric Disease Designation (RPDD) for the treatment of neuroblastoma, rhabdomyosarcoma, Ewing’s
sarcoma and osteosarcoma. Most recently, the European Commission granted an ODD for r/r MM.
Our product pipeline also includes one
preclinical PDC chemotherapeutic program (CLR 1900) and several partnered PDC assets. The CLR 1900 Series is being targeted for
solid tumors with a payload that inhibits mitosis (cell division) a validated pathway for treating cancers.
We have leveraged our PDC platform to establish
four collaborations featuring five unique payloads and mechanisms of action. Through research and development collaborations, our
strategy is to generate near-term capital, supplement internal resources, gain access to novel molecules or payloads, accelerate
product candidate development and broaden our proprietary and partnered product pipelines.
Our PDC platform provides selective delivery
of a diverse range of oncologic payloads to cancerous cells, whether a hematologic cancer or solid tumor, a primary tumor, or a
metastatic tumor and cancer stem cells. The PDC platform’s mechanism of entry does not rely upon specific cell surface epitopes
or antigens as are required by other targeted delivery platforms. Our PDC platform takes advantage of a metabolic pathway utilized
by all tumor cell types in all stages of the tumor cycle. Tumor cells modify specific regions on the cell surface as a result of
the utilization of this metabolic pathway. Our PDCs bind to these regions and directly enter the intracellular compartment. This
mechanism allows the PDC molecules to accumulate over time, which enhances drug efficacy, and to avoid the specialized highly acidic
cellular compartment known as lysosomes, which allows a PDC to deliver molecules that previously could not be delivered. Additionally,
molecules targeting specific cell surface epitopes face challenges in completely eliminating a tumor because the targeted antigens
are expressed in limited in the total numbers on the cell surface, have longer cycling time from internalization to being present
on the cell surface again upon binding and are not present on all tumor cells of a particular cancer type. This means a subpopulation
of tumor cells always exist that cannot be targeted by therapies targeting specific surface epitopes. In addition to the benefits
provided by the mechanism of entry, PDCs offer the ability to conjugate payload molecules in numerous ways, thereby increasing
the types of molecules selectively delivered via the PDC.
The PDC platform features include the capacity
to link with almost any molecule, provide a significant increase in targeted oncologic payload delivery and the ability to target
all types of tumor cells. As a result, we believe that we can generate PDCs to treat a broad range of cancers with the potential
to improve the therapeutic index of oncologic drug payloads, enhance or maintain efficacy while also reducing adverse events by
minimizing drug delivery to healthy cells, and increasing delivery to cancerous cells and cancer stem cells.
We employ a drug discovery and development
approach that allows us to efficiently design, research and advance drug candidates. Our iterative process allows us to rapidly
and systematically produce multiple generations of incrementally improved targeted drug candidates.
A description of our PDC product candidates
follows:
Clinical Pipeline
Our lead PDC therapeutic, CLR 131 is a
small-molecule, PDC designed to provide targeted delivery of iodine-131 directly to cancer cells, while limiting exposure to healthy
cells. We believe this profile differentiates CLR 131 from many traditional on-market treatments and treatments in development.
CLR 131 is currently being evaluated in a Phase 2 study in r/r B-cell lymphomas, and two Phase 1 dose-escalating clinical studies,
one in r/r MM and one in r/r pediatric solid tumors and lymphoma. The initial Investigational New Drug (IND) application was accepted
by the FDA in March 2014 with multiple INDs submitted since that time. Initiated in March 2017, the primary goal of the Phase 2
study is to assess the compound’s efficacy in a broad range of hematologic cancers. The Phase 1 study is designed to assess
the compound’s safety and tolerability in patients with r/r MM (to determine maximum tolerated dose) and was initiated in
April 2015. The FDA previously accepted our IND application for a Phase 1 open-label, dose escalating study to evaluate the safety
and tolerability of a single intravenous administration of CLR 131 in up to 30 children and adolescents with cancers including
neuroblastoma, sarcomas, lymphomas (including Hodgkin’s lymphoma) and malignant brain tumors. This study was initiated during
the first quarter of 2019. These cancer types were selected for clinical, regulatory and commercial rationales, including the radiosensitive
nature and continued unmet medical need in the r/r setting, and the rare disease determinations made by the FDA based upon the
current definition within the Orphan Drug Act.
In December 2014, the FDA granted ODD for
CLR 131 for the treatment of MM. Multiple myeloma is an incurable cancer of the plasma cells and is the second most common form
of hematologic cancers. In 2018, the FDA granted ODD and RPDD for CLR 131 for the treatment of neuroblastoma, rhabdomyosarcoma,
Ewing’s sarcoma and osteosarcoma. The FDA may award priority review vouchers to sponsors of rare pediatric disease products
that meet its specified criteria. The key criteria to receiving a priority review voucher is that the disease being treated is
life-threatening and that it primarily effects individuals under the age of 18. Under this program, a sponsor who receives an approval
for a drug or biologic for a rare pediatric disease can receive a priority review voucher that can be redeemed to receive a priority
review of a subsequent marketing application for a different product. Additionally, these priority review vouchers can be exchanged
or sold to other companies for them to use the voucher. In May 2019, the FDA granted Fast Track designation for CLR 131 for the
treatment of multiple myeloma in July 2019 for the treatment of DLBCL, in September, CLR 131 received Orphan Drug Designation from
the European Union for Multiple Myeloma, and in January 2020, CLR 131 the FDA granted Orphan Drug Designation for CLR 131 in lymphoplasmacytic
lymphoma (LPL).
Phase 2 Study in Patients
with r/r select B-Cell Malignancies
In February 2020,
we announced positive data from our Phase 2 CLOVER-1 study in patients with relapsed/refractory B-cell lymphomas. Relapsed/Refractory
MM and non-Hodgkin lymphoma (NHL) patients were treated with three different doses (<50mCi, ~50mCi and ~75mCi
total body dose (TBD). The <50mCi total body dose was a deliberately planned sub-therapeutic dose. CLR 131 achieved the primary
endpoint for the study. Patients with r/r MM who received the highest dose of CLR 131 showed a 42.8% overall response rate (ORR).
Those who received ~50mCi TBD had a 26.3% ORR with a combined rate of 34.5% ORR (n=33) while maintaining a well-tolerated safety
profile. Patients in the studies were elderly with a median age of 70, and heavily pre-treated, with a median of five prior lines
of treatment (range: 3 to 17), which included immunomodulatory drugs, proteasome inhibitors and CD38 antibodies for the majority
of patients. Additionally, a majority of the patients (53%) were quad refractory or greater and 44% of all treated multiple myeloma
patients were triple class refractory. 100% of all evaluable patients (n=43) achieved clinical benefit (primary outcome measure)
as defined by having stable disease or better. 85.7% of multiple myeloma patients receiving the higher total body dose levels of
CLR 131 experienced tumor reduction. The 75mCi TBD demonstrated positive activity in both high-risk patients and triple class refractory
patients with a 50% and 33% ORR, respectively.
Patients with
r/r NHL who received ~50mCi TBD and the ~75mCi TBD had a 42% and 43% ORR, respectively and a combined rate of 42%. These patients
were also heavily pre-treated, having a median of three prior lines of treatment (range, 1 to 9) with the majority of patients
being refractory to rituximab and/or ibrutinib. The patients had a median age of 70 with a range of 51 to 86. All patients had
bone marrow involvement with an average of 23%. In addition to these findings, subtype assessments were completed in the r/r B-cell
NHL patients. Patients with DLBCL demonstrated a 30% ORR with one patient achieving a complete response (CR), which continues at
nearly 24 months post-treatment. The ORR for CLL/SLL/MZL patients was 33%. Current data from our Phase 2 CLOVER-1 clinical study
show that four LPL/WM patients demonstrated 100% ORR with one patient achieving a CR which continues at nearly 27 months post-treatment.
This may represent an important improvement in the treatment of relapsed/refractory LPL/WM as no approved or as we believe late-stage
development treatments for second- and third-line patients have reported a CR. LPL/WM is a rare, indolent and incurable form of
NHL that is comprised of a niche patient population in need of new and better treatment options.
The most frequently
reported adverse events in r/r MM patients were cytopenias, which followed a predictable course and timeline. The frequency of
adverse events have not increased as doses were increased and the profile of cytopenias remains
consistent. Importantly, these cytopenias have had a predictable pattern to initiation, nadir and recovery and are treatable. The
most common grade ≥3 events at the highest dose (75mCi TBD) were hematologic toxicities including thrombocytopenia (65%), neutropenia
(41%), leukopenia (30%), anemia (24%) and lymphopenia (35%). No patients experienced cardiotoxicities, neurological toxicities,
infusion site reactions, peripheral neuropathy, allergic reactions, cytokine release syndrome, keratopathy, renal toxicities, or
changes in liver enzymes. The safety and tolerability profile in patients with r/r NHL was similar to
r/r MM patients except for fewer cytopenias of any grade. Based upon CLR 131 being well tolerated across all dose groups and the
observed response rate, especially in difficult to treat patients such as high risk and triple class refractory or penta-refractory,
and corroborating data showing the potential to further improve upon current ORRs and durability of those responses, the
study has been expanded to test a two-cycle dosing optimization regimen of CLR 131.
In July 2018, we announced that after a
single 25mCi/m2 IV administration of CLR 131, patients with relapsed/refractory aggressive DLBCL were assessed for response.
These interim data show a 33% ORR and a 50% clinical benefit response (CBR). In addition, the observed responses to date show overall
tumor reduction ranged from 60% to greater than 90%. As a result of these favorable outcomes, we have expanded this cohort to include
up to 30 additional patients. We also announced that a patient in the lymphoplasmacytic lymphoma (LPL) arm with advanced Waldenstrom
macroglobulinema showed a 94% reduction in tumor burden and complete resolution in four of five targeted masses after two doses
of CLR 131 separated by 123 days.
In July 2016, we were awarded a $2,000,000
National Cancer Institute (NCI) Fast-Track Small Business Innovation Research grant to further advance the clinical development
of CLR 131. The funds are supporting the Phase 2 study initiated in March 2017 to define the clinical benefits of CLR 131 in r/r
MM and other niche hematologic malignancies with unmet clinical need. These niche hematologic malignancies include Chronic Lymphocytic
Leukemia, Small Lymphocytic Lymphoma, Marginal Zone Lymphoma, Lymphoplasmacytic Lymphoma and DLBCL. The study is being conducted
in approximately 10 U.S. cancer centers in patients with orphan-designated relapse or refractory hematologic cancers. The study’s
primary endpoint is CBR, with additional endpoints of ORR, PFS, median Overall Survival (mOS) and other markers of efficacy following
a single 25.0 mCi/m2 dose of CLR 131, with the option for a second 25.0 mCi/m2 dose approximately 75-180
days later. Based on the performance results from Cohort 5 of our Phase 1 study in patients with r/r MM, reviewed below, we have
modified the dosing regimen of this study to a fractionated dose of 15.625 mCi/m2 administered on day 1 and day 8.
Phase 1 Study in Patients with r/r Multiple
Myeloma
In February 2020, we announced the successful
completion of our Phase 1 dose escalation study. Data from the study demonstrated that CLR 131 was safe and tolerated at total
body doses of >80mCi in r/r multiple myeloma (MM), The Phase 1 multicenter, open-label, dose-escalation study was designed to
evaluate the safety and tolerability of CLR 131 administered as a 30-minute I.V. infusion, either as a single bolus dose or as
two fractionated doses. The r/r multiple myeloma patients in this study received doses ranging from ≤25mCi to >80mCi total
body dose. To date, an independent Data Monitoring Committee determined that all doses have been safe and well-tolerated by patients.
CLR 131 in combination with dexamethasone
is currently under investigation in adult patients with r/r MM. Patients must have been refractory to or relapsed from at least
one proteasome inhibitor and at least one immunomodulatory agent. The clinical study is a standard three-plus-three dose escalation
safety study to determine the maximum tolerable dose. Multiple myeloma is an incurable cancer of the plasma cells and is the second
most common form of hematologic cancers. Secondary objectives include the evaluation of therapeutic activity by assessing surrogate
efficacy markers, which include M protein, free light chain FLC, PFS and OS. All patients have been heavily pretreated with an
average of five prior lines of therapy. CLR 131 was deemed by an Independent Data Monitoring Committee (IDMC) to be safe and tolerable
up to its planned maximum single, bolus dose of 31.25 mCi/m2. The four single dose cohorts examined were: 12.5 mCi/m2
(~25mCi TBD), 18.75 mCi/m2 (~37.5mCi TBD), 25 mCi/m2(~50mCi TBD), and 31.25 mCi/m2(~62.5mCi TBD),
all in combination with low dose dexamethasone (40 mg weekly). Of the five patients in the first cohort, four achieved stable disease
and one patient progressed at Day 15 after administration and was taken off the study. Of the five patients admitted to the second
cohort, four achieved stable disease and one patient progressed at Day 41 after administration and was taken off the study. Four
patients were enrolled to the third cohort and all achieved stable disease. In September 2017, we announced results for cohort
4, showing that a single infusion up to 30-minutes of 31.25mCi/m2 of CLR 131 was safe and tolerated by the three patients
in the cohort. Additionally, all three patients experienced CBR with one patient achieving a partial response (PR). We use the
International Myeloma Working Group (IMWG) definitions of response, which involve monitoring the surrogate markers of efficacy,
M protein and FLC. The IMWG defines a PR as a greater than or equal to 50% decrease in FLC levels (for patients in whom M protein
is unmeasurable) or 50% or greater decrease in M protein. The patient experiencing a PR had an 82% reduction in FLC. This patient
did not produce M protein, had received seven prior lines of treatment including radiation, stem cell transplantation and multiple
triple combination treatments including one with daratumumab that was not tolerated. One patient experiencing stable disease attained
a 44% reduction in M protein. In January 2019, we announced that the pooled mOS data from the first four cohorts was 22.0 months.
In late 2018, we modified this study to evaluate a fractionated dosing strategy to potentially increase efficacy and decrease adverse
events.
Following the determination that all prior
dosing cohorts were safe and tolerated, we initiated a cohort 7 utilizing a 40mCi/m2 fractionated dose administered 20mCi/m2 (~40mCi
TBD) on days 1 and day 8. Cohort 7 was the highest pre-planned dose cohort and subjects have completed the evaluation period. Final
study report and study close-out will be completed later this year.
In May 2019, we announced that the FDA
granted Fast Track Designation for CLR 131 in fourth line or later r/r MM. CLR 131 is our small-molecule radiotherapeutic PDC designed
to deliver cytotoxic radiation directly and selectively to cancer cells and cancer stem cells. It is currently being evaluated
in our ongoing CLOVER-1 Phase 2 clinical study in patients with relapsed or refractory multiple myeloma and other select B-cell
lymphomas.
Phase 1 Study in
r/r Pediatric Patients with select Solid tumors, Lymphomas and Malignant Brain Tumors.
In December 2017 the Division of Oncology
at the FDA accepted our IND and study design for the Phase 1 study of CLR 131 in children and adolescents with select rare and
orphan designated cancers. This study was initiated during the first quarter of 2019. In December 2017, we filed an IND application
for r/r pediatric patients with select solid tumors, lymphomas and malignant brain tumors. The Phase 1 clinical study of CLR 131
is an open-label, sequential-group, dose-escalation study evaluating the safety and tolerability of intravenous administration
of CLR 131 in up to 30 children and adolescents with cancers including neuroblastoma, sarcomas, lymphomas (including Hodgkin’s
lymphoma) and malignant brain tumors. Secondary objectives of the study are to identify the recommended Phase 2 dose of CLR 131
and to determine preliminary antitumor activity (treatment response) of CLR 131 in children and adolescents. In 2018, the FDA granted
OD and RPDD for CLR 131 for the treatment of neuroblastoma, rhabdomyosarcoma, Ewing’s sarcoma and osteosarcoma. Should any
of these indications reach approval, the RPDD would enable us to receive a priority review voucher. Priority review vouchers can
be used by the sponsor to receive priority review for a future New Drug Application (“NDA”) or Biologic License Application
(“BLA”) submission, which would reduce the FDA review time from 12 months to six months. Currently, these vouchers
can also be transferred or sold to another entity.
Phase 1 Study in r/r Head and Neck Cancer
In August 2016, the University
of Wisconsin Carbone Cancer Center (“UWCCC”) was awarded a five-year Specialized Programs of Research Excellence (“SPORE”)
grant of $12,000,000 from the National Cancer Institute and the National Institute of Dental and Craniofacial Research to improve
treatments and outcomes for head and neck cancer, HNC, patients. HNC is the sixth most common cancer across the world with approximately
56,000 new patients diagnosed every year in the U.S. As a key component of this grant, the UWCCC researchers completed testing
of CLR 131 in various animal HNC models and initiated the first human clinical trial enrolling up to 30 patients combining CLR
131 and external beam radiation with recurrent HNC in Q4 2019. As of the date of this filing, this clinical trial is suspended
due to the COVID-19 pandemic.
Preclinical Pipeline
We believe our PDC platform has potential
to provide targeted delivery of a diverse range of oncologic payloads, as exemplified by the product candidates listed below, that
may result in improvements upon current standard of care (“SOC”) for the treatment of a broad range of human cancers:
|
|
·
|
CLR 1800 Series was a collaborative PDC program with Pierre Fabre that expired in January 2019. The program has been successful in demonstrating improved tolerability and efficacy in multiple animal models. The newly developed PDCs may provide enhanced therapeutic indices to otherwise highly potent, nontargeted payloads through the targeted delivery of the chemotherapeutic payload to cancer cells via our proprietary phospholipid ether delivery platform. The CLR 1800 Series remains under evaluation by us as a number of PDC molecules have the potential to be progressed toward and into IND enabling studies.
|
|
|
·
|
CLR 1900 Series is an internally developed proprietary PDC program leveraging a novel small molecule cytotoxic compound as the payload. The payload inhibits mitosis (cell division) and targets a key pathway required to inhibit rapidly dividing cells that results in apoptosis. We believe that this program could produce a product candidate targeted to select solid tumors. Currently, the program is in early preclinical development and if we elect to progress any molecules further, we will select preferred candidates.
|
|
|
·
|
CLR 2000 Series is a collaborative PDC program with Avicenna Oncology, or Avicenna, that we entered into in July 2017. Avicenna is a developer of antibody drug conjugates (“ADCs”). The objective of the research collaboration is to design and develop a series of PDCs utilizing Avicenna’s proprietary cytotoxic payload. Although Avicenna is a developer of ADCs, this collaboration was sought as a means to overcome many of the challenges associated with ADCs, including those associated with the targeting of specific cell surface epitopes. The CLR 2000 Series has demonstrated improved safety, efficacy and tissue distribution with the cytotoxic payload in animal models. A candidate molecule and a back-up have been selected for further advancement.
|
|
|
·
|
CLR 2100 and 2200 Series are collaborative PDC programs with Onconova Therapeutics, Inc., or Onconova, that we entered into in September 2017. Onconova is a biotechnology company specializing in the discovery and development of novel small molecule cancer therapies. The collaboration is structured such that we will design and develop a series of PDCs utilizing different small molecules that Onconova was developing as payloads with the intent to show improved targeting and specificity to the tumor. At least one of the molecules was taken into Phase 1 clinical studies previously by Onconova. We would own all new intellectual property associated with the design of the new PDCs, and both companies will have the option to advance compounds.
|
|
|
·
|
CLR 12120 Series is a collaborative PDC program with Orano Med for the development of novel PDCs utilizing Orano Med’s unique alpha emitter, lead 212 conjugated to our phospholipid ether; the companies intend to evaluate the new PDCs in up to three oncology indications. Currently this series has shown efficacy in the first two animal models tested.
|
Technology Overview
Our product candidates are based on a cancer-targeting
delivery platform of optimized phospholipid ether (PLE) analogs (phospholipid ether proprietary delivery vehicle) that interact
with lipid rafts. Lipid rafts are specialized regions of a cell’s membrane phospholipid bilayer that contain high concentrations
of cholesterol and sphingolipids and serve to organize cell surface and intracellular signaling molecules. As a result of enrichment
and stabilization of lipid rafts in cancer cells, including cancer stem cells, our product candidates provide selective targeting
preferentially to cancer cells over normal healthy cells. The cancer-targeting PLE delivery vehicle was deliberately designed to
be combined with therapeutic, diagnostic and imaging molecules. For example, the cytotoxic radioisotope, iodine-131 can be attached
via a stable covalent bond to the PLE resulting in our lead PDC, CLR 131. Non-radioactive molecules, including many classes of
small molecule chemotherapeutic compounds, peptides and other molecules can also be attached to the delivery vehicle.
We are focused on exploring the creation
of additional PDCs ranging from newly discovered to well-characterized chemotherapeutic payloads. The objective is to develop PDC
chemotherapeutics through conjugation of our delivery vehicle and non-targeted anti-cancer agents to improve therapeutic indices
and expand potential indications through the targeted delivery of chemotherapeutic payloads. Initial PDC product candidates include
our CLR 1800, 1900, 2000, 2100, 2200, and 12120 series of conjugated compounds currently being researched independently and through
partnerships. Other than CLR 12120, all are small-molecule, cancer-targeting chemotherapeutics in pre-clinical research. To date,
multiple cancer-targeting product profiles have been generated from a single chemical core structure that is the foundation of
our technology platform. We also believe that additional cytotoxic PDCs may be developed possessing enhanced therapeutic indices
versus the original, non-targeted cytotoxic payload as a monotherapy.
Malignant tumor targeting, including targeting
of cancer stem cells, has been demonstrated in vivo in animal models as well as in clinical studies. Mice without intact
immune systems and inoculated with Panc-1 (pancreatic carcinoma) cells, were injected with CLR 1502, 24 or 96 hours prior to imaging.
In vivo optical imaging showed pronounced accumulation of CLR 1502 in tumors versus non-target organs and tissues. Similarly,
positron emission tomography (PET) imaging of tumor-bearing animals (colon, glioma, triple negative breast and pancreatic tumor
xenograft models) administered the imaging agent CLR 124 clearly shows selective uptake and retention by both primary tumors and
metastases, including cancer stem cells. PET/CT analysis following co-injection of CLR 131 (for therapy) and CLR 124 (for imaging)
revealed time-dependent tumor responses and disappearance over nine days in a cancer xenograft model. We believe that the capability
of our technology to target and be selectively retained by cancer stem cells in vivo, was demonstrated by treating glioma
stem cell-derived orthotopic tumor-bearing mice with another fluorescent-labeled PDC (CLR 1501), and then removing the tumor and
isolating cancer stem cells, which continued to display CLR 1501 labeling even after three weeks in cell culture.
The basis for selective tumor targeting
of our compounds lies in differences between the plasma membranes of cancer cells as compared to those of most normal cells. Data
suggests that lipid rafts serve as portals of entry for PDCs such as CLR 131 and our multiple series of drug conjugates. The marked
selectivity of our compounds for cancer cells versus non-cancer cells likely results from cancer cells maintenance of an overabundance
of lipid rafts and the stabilization of these microdomains within the plasma membrane as compared to normal cells. Following cell
entry via lipid rafts, CLR 131 is transported into the cytoplasm, where it traffics along the Golgi apparatus and is distributed
to various peri-nuclear organelles (including mitochondria and the endoplasmic reticulum). The pivotal role played by lipid rafts
is underscored by the fact that disruption of lipid raft architecture significantly eliminates uptake of our PDC delivery vehicle
into cancer cells.
Products in Development
CLR 131
CLR 131 is a small-molecule, cancer-targeting
molecular radiotherapeutic PDC that we believe has the potential to be the first radiotherapeutic agent to use PLEs to target cancer
cells. CLR 131 is comprised of our proprietary PLE, 18-(p-[I-131]iodophenyl) octadacyl phosphocholine, acting as a cancer-targeting
delivery and retention vehicle, covalently labeled with iodine-131, a cytotoxic (cell-killing) radioisotope with a half-life of
eight days that is already in common use to treat thyroid, pediatric tumors and other cancer types including NHL. It is this “intracellular
radiation” mechanism of cancer cell killing, coupled with delivery to a wide range of malignant tumor types that we believe
provides CLR 131 with anti-cancer activity. Selective uptake and retention have been demonstrated in cancer stem cells compared
with normal cells, offering the prospect of longer lasting anti-cancer activity.
Pre-clinical experiments in tumor models
have demonstrated selective killing of cancer cells along with a safe and tolerable product profile. CLR 131’s anti-tumor/survival-prolonging
activities have been demonstrated in more than a dozen models including breast, prostate, lung, brain, pancreatic, ovarian, uterine,
renal, and colorectal cancers as well as, melanoma and multiple myeloma. In all but two models, a single administration of a well-tolerated
dose of CLR 131 was sufficient to demonstrate efficacy. Moreover, efficacy was also seen in a model employing human uterine sarcoma
cells that have known resistance to many standard chemotherapeutic drugs. CLR 131 was also tested in combination with a standard
efficacious dose of gemcitabine in a pancreatic cancer model. Single doses of CLR 131 or gemcitabine given alone were equally efficacious,
while the combination therapy was significantly more efficacious than either treatment alone (additive). While single doses of
CLR 131 have been effective and tolerated in multiple preclinical animal models, CLR 131 has been shown to provide a statistically
significant improvement in efficacy and survival when provided in a multi-dose format and remains tolerated. In each study, the
dose of CLR 131 was ~100 µCi, which is approximately 50-fold less than the maximum tolerated dose (MTD) of CLR 131 determined
in a six-month rat radiotoxicity study.
Extensive IND-enabling, Good Laboratory
Practices (GLP) in vivo and in vitro pre-clinical pharmacokinetic/ distribution, toxicology and drug safety studies
were successfully completed in 2007 through 2009 using non-pharmacological concentrations/doses of PLE consistent with its role
as a delivery/retention vehicle in CLR 131. Tissue distribution studies supported prediction of acceptable human organ exposures
and body clearance for CLR 131. Importantly, and in sharp distinction from biological products labeled with I-131, the small-molecule
CLR 131 showed very minimal variation in excretion kinetics and tissue distribution among individuals within species or across
a 500-fold variation in dose. Single and repeat-dose animal toxicology studies indicated very high margins of safety with our PLE
delivery and retention vehicle even when administered at 80-200x over the amount required to deliver the anticipated maximum human
therapy dose of CLR 131.
In 2009, we filed an IND with the FDA to
study CLR 131 in humans. In February 2010, we completed a Phase 1 dosimetry study with a single intravenous dose of 10 mCi/m2
CLR 131 in eight patients with relapsed/refractory advanced solid tumors. Single doses of CLR 131 were tolerated and the reported
adverse events were all considered minimal, manageable and either not dose limiting or not related to CLR 131. There were no serious
adverse events reported. Analysis of total body imaging and blood and urine samples collected over 42 days following injection
indicated that doses of CLR 131 expected to be therapeutically effective could be administered without harming vital organs. Two
subjects (one with colorectal cancer metastasized to lung and another with prostate cancer) had tumors that were imaged with 3D
nuclear scanning (SPECT/CT) on day 6 after administration of CLR 131. Uptake of CLR 131 into tumor tissue (but not adjacent normal
tissue or bone marrow) was clearly demonstrated in both subjects. Confirming animal studies, pharmacokinetic analyses demonstrated
a prolonged half-life of radioactivity in the plasma after CLR 131 administration (approximately 200 hours) and that there was
no significant variation in excretion or radiation dosimetry among subjects. The study established an initial dose of 12.5 mCi/m2,
for the Phase 1b escalating dose study that commenced in January 2012.
The primary objective of the multicenter
Phase 1b dose-escalation study in patients with a range of advanced solid tumors was to define the MTD of CLR 131. In addition
to determining the MTD, the Phase 1b study was intended to evaluate overall tumor response (using standard RESIST 1.1 criteria)
and safety. In September 2012, we announced that we had successfully completed the second cohort in this Phase 1b dose-escalation
study. Dose escalation in four cohorts subsequently occurred with refractory cancer patients receiving single doses of 25 mCi/m2,
31.25 mCi/m2 or 37.5 mCi/m2.
Tumor treatment with radioactive isotopes
has been used as a fundamental cancer therapeutic for decades. The goals of targeted cancer therapy — selective delivery
of effective doses of isotopes that destroy tumor tissue, sparing of surrounding normal tissue, and non-accumulation in vital organs
such as the liver and kidneys — remain goals of new therapies as well. We believe our isotope delivery technology has the
potential to achieve these goals. To date, CLR 131 has been shown in animal models to reliably and near-universally accumulate
in cancer cells, including cancer stem cells, and because the therapeutic properties of iodine-131 are well known, we believe the
risk of non-efficacy in human clinical studies is less than that of other cancer therapies at this stage of development, although
no assurance can be given.
In view of CLR 131’s selective uptake
and retention in a wide range of solid tumors and in cancer stem cells, its single-agent efficacy in animal models and its non-specific
mechanism of cancer-killing (radiation), we are initially developing CLR 131 as a monotherapy for cancer indications with significant
unmet medical need. While a number of indications were evaluated as the initial target treatment, multiple myeloma was selected
principally because it is an incurable hematologic disease that is highly radiosensitive, with significant unmet medical need in
the relapse or refractory clinical setting and is designated as an orphan disease. As a result, this may provide an accelerated
regulatory pathway due to CLR 131’s unique benefits such as a novel mechanism of action, ease of administration, and positive
benefit/risk profile potential in various high unmet cancer populations. The IND application for multiple myeloma was accepted
by the FDA in September 2014. In December 2014, the FDA granted ODD for CLR 131 for the treatment of multiple myeloma. We initiated
our Phase 1 Study of CLR 131 for the treatment of r/r MM in April 2015 and have provided periodic clinical updates. CLR 131 is
being evaluated as a monotherapy and will subsequently be explored as a combination therapy with chemotherapeutic agents, immunomodulatory
agents and in combination with external beam radiotherapy.
In September 2017, we announced results
for Cohort 4 showing that a single 30-minute infusion of 31.25mCi/m2 of CLR 131 was safe and tolerated by the three
patients in the cohort. Additionally, all three patients experienced clinical benefit with one patient achieving a partial response
(“PR”). We are monitoring response rates via surrogate markers of efficacy including M protein and FLC. The IMWG defines
a PR as a greater than or equal to 50% decrease in FLC levels (for patients in whom M protein is unmeasurable) or 50% decrease
in M protein. The patient experiencing a PR had an 82% reduction in FLC. This patient did not produce M protein, received seven
prior lines of treatment including radiation, stem cell transplantation and multiple triple combination treatments including one
with daratumumab that was not tolerated. One patient experiencing stable disease attained a 44% reduction in M protein. For all
of the patients receiving the single dose, CLR 131 was the third line of treatment or later. We have recently converted the Phase
1a clinical data (single CLR 131 dose) to pooled data for presentation of the total performance of the results to date. On January
7, 2019, we announced that the pooled mOS data from the first four cohorts was 22.0 months. Based on the safety observed to date
as well as various efficacy signals, including reductions in M protein and FLC and a pooled mOS that has not yet been reached,
the study protocol was modified for cohort 5 to introduce a fractionated dose of 15.625 mCi/m2 administered on day 1
and day 8 to further determine the optimal dose-range for CLR 131. Results from Cohort 5 indicate enhanced tolerability and safety
in comparison to Cohort 4 despite an 18% increase in total average dose from 55.29 mCi to 65.15 mCi of CLR 131. Patients in Cohort
5 required less supportive care such as transfusions of platelets or packed red blood cells than seen in previous cohorts. Furthermore,
a review of surrogate efficacy markers demonstrated that patients in Cohort 5 monitored by M-protein showed a nearly 50% further
reduction in M-Protein than seen in Cohort 4. Based on the results and an IDMC, on December 4, 2018 we initiated a sixth cohort
using a fractionated two dose regimen of 18.75 mCi/m2 administered one week apart.
CLR 131 is also being evaluated in a Phase
2 clinical study examining r/r MM patients as well as selected other B-cell hematological malignancies. Patients will receive a
25 mCi/m2 dose infused over approximately 30 minutes with the option of a second 25 mCi/m2 dose 75-180 days
later based on physician assessment. Based on the performance results from Cohort 5 of our Phase 1 study in patients with r/r MM,
reviewed below, we modified the dosing regimen of this study to a fractionated dose of 15.625 mCi/m2 administered on
day 1 and day 8. This study is partially funded through a $2,000,000 Fast Track NCI SBIR award which was granted in July 2016.
In February 2019, we announced that a single,
25mCi/m2, 30-minute intravenous infusion of CLR 131 in the first 10 patients with r/r MM were assessed. These interim
data show a 30% response rate in a patient population which received an average of five prior lines of systemic therapy (including
daratumumab), at least one stem cell transplantation with the average age being 70. The observed responses to date show overall
reductions in surrogate markers of disease (M protein or free light chains, depending upon which is being used to monitor the patient’s
disease) between 70% and over 90%. In addition to these patients, 100% of patients achieved stable disease with 2 patients experiencing
a minimal response or a minimum reduction of a 25% in the surrogate marker being used to monitor the patient’s disease. Historically,
patients receiving 4th line chemotherapy treatment show a 15% response rate, and patients receiving 5th line chemotherapy show
an 8% response rate, whether dosed as mono-therapy or in combination. The multiple myeloma average treatment response rate (RR)
provided by line of therapy was obtained through a global information and technology vendor specializing in healthcare data analysis
utilizing over 12.5 billion U.S. insurance claims and 90 million electronic medical records. As a result of these outcomes, we
have expanded this cohort to include up to 30 additional patients.
In July 2018, we announced that after a
single 25mCi/m2 IV administration of CLR 131, patients with relapsed/refractory aggressive DLBCL were assessed for response.
These interim data show a 33% ORR and a 50% CBR. In addition, the observed responses to date show overall tumor reduction ranged
from 60% to greater than 90%. As a result of these favorable outcomes, we have expanded this cohort to include up to 30 additional
patients. We also announced that a patient in the lymphoplasmacytic lymphoma (LPL) arm with advanced Waldenstrom macroglobulinema
showed a 94% reduction in tumor burden and complete resolution in four of five targeted masses after two doses of CLR 131 separated
by 123 days.
In December 2017, we filed an IND application
with the Division of Oncology at the FDA for a proposed Phase 1 study of CLR 131 in children and adolescents with select rare and
orphan designated cancers. The Phase 1 clinical study of CLR 131 is an open-label, sequential-group, dose-escalation study to evaluate
the safety and tolerability of a single intravenous administration of CLR 131 in up to 30 children and adolescents with cancers
including neuroblastoma, sarcomas, lymphomas (including Hodgkin’s lymphoma) and malignant brain tumors. Secondary objectives
of the study are to identify the recommended Phase 2 dose of CLR 131 and to determine preliminary antitumor activity (treatment
response) of CLR 131 in children and adolescents. In 2018, the FDA a granted ODD and RPDD for CLR 131 for the treatment of neuroblastoma,
rhabdomyosarcoma, Ewing’s sarcoma and osteosarcoma. Should any of these indications reach approval, the RPDD may enable us
to receive a priority review voucher. Priority review vouchers can be used by the sponsor to receive Priority Review for a future
NDA or BLA submission, which would reduce the FDA review time from 12 months to six months. Currently, these vouchers can also
be transferred or sold to another entity. We plan to initiate this Phase 1 study in 2019, at 3-5 pediatric cancer centers within
and possibly outside the US.
Market Overview
Our target market is broad and represents
the market for the treatment of cancer. The American Cancer Society estimated that approximately 1.76 million new cancer cases
were expected to be diagnosed in the U.S. in 2019 and approximately 606,880 cancer deaths in the U.S. The global market for cancer
drugs reached $107 billion in annual sales (June 2015), and could reach $150 billion by 2020, according to a report dated June
2016 by the IMS Institute for Healthcare Informatics, a unit of drug data provider IQVIA. This growth will be driven by emerging
targeted therapies, which are expected to change the cancer treatment landscape (Cowen Report), and an increased use of cancer
drug combination regimens.
Multiple Myeloma
According to the National Cancer Institute
SEER database, multiple myeloma is the second most common hematologic cancer with a U.S. incidence rate and a relapse or refractory
patient population of 10,000 to 15,000. The Decision Resources Group in 2016 estimated the multiple myeloma dollar market size
to be over $17B in 2018 and is forecasted to increase to nearly $27B in 2023. The increase in drug sales over this period will
be mainly driven by the increasing incidence of multiple myeloma in each of the seven key markets with the U.S. market remaining
the largest potential market. It is believed the largest growth will occur in patients receiving at least three lines of treatment
due to the expanding elderly population, increases in treatment population and increasing rates of survival from earlier lines
of treatment. According to data obtained from Decision Resource Group, over 40% of patients in later lines of therapy while eligible,
refuse treatment due to higher treatment failure, severity of adverse events and difficulty of treatment dosing regimen. The average
response rates for patients receiving their fourth- and fifth-line treatment are 15% and 8% response rates respectively. Additionally,
the mOS for these patients also decreases by line of therapy and is less than 9 months post third-line treatment.
Based on the CLR 131 Phase 1 and Phase
2 product profile demonstrated in fifth-line patients to date with a single dose, we believe CLR 131 may meet the unmet medical
need in the heavily pre-treated patient population described above.
B-Cell Lymphoma
B-cell
lymphoma represents cancers of the lymphatic system. The lymphoma may be indolent or aggressive and circulates in the blood or
form tumors in lymph nodes. According to the National Cancer Institute SEER data base the estimated 2018 incidence of B-Cell Lymphoma
was 163,000 cases. Types of B-Cell Lymphomas include Chronic Lymphocytic Leukemia, Small Lymphocytic Leukemia, Mantel Cell Lymphoma,
Marginal Zone Lymphoma, and the most common lymphoma, DLBCL.
We
believe there is a significant unmet medical need in B-Cell Lymphoma due to continued high mortality and poor response rates remain
in second- and third- line treatments compounded by the limited durability of responses.
Based on the CLR 131 Phase 2 product profile
demonstrated in DLBCL patients to date with a single dose, we believe CLR 131 may meet the unmet medical need in the patient population
described above as well.
Neuroblastoma
Neuroblastoma, a
neoplasm of the sympathetic nervous system, is the most common extracranial solid tumor of childhood, accounting for approximately
7.8% of childhood cancers in the U.S. The National Cancer Institute states the incidence is about 10.54 cases per 1 million per
year in children younger than 15 years and 90% are younger than 5 years at diagnosis. Over 650 new cases are diagnosed each year
in North America. Approximately 50% of patients present with metastatic disease requiring systemic treatment. Clinical consequences
include abdominal distension, proptosis, bone pain, pancytopenia, fever and paralysis. Although the prognosis is favorable in children
under one year of age with an 86 to 95% 5-year survival, in children aged one to 14 years the 5-year survival ranges from 34 to
68%.
Sarcomas
Sarcomas represent a heterogeneous disease
group. Sarcomas grow in connective tissue, or cells that connect or support other kinds of tissue in the body. These tumors are
most common in the bones, muscles, tendons, cartilage, nerves and blood vessels. Sarcomas represent 15% of all pediatric tumors
and 21% of pediatric solid tumors. The National Cancer Institute SEER data base estimates
that there were 2,060 incidences in 2019. The median age at diagnosis was 3, the median age of death was 5.
We
are focused on 3 subsets of Sarcomas:
|
|
·
|
Osteosarcoma: The tumor develops in growing bone tissues, accounts for 28% of all bone sarcomas and is the most common pediatric sarcoma (56%).
|
|
|
·
|
Ewing’s Sarcoma: The tumor develops in immature tissues in bone marrow
|
|
|
·
|
Rhabdomyosarcoma: Tumors develop in the muscles predominately skeletal muscle.
|
Based on information from Market Insights,
Epidemiology, and Market Forecast, the global market value of the Pediatric Sarcoma Market is expected to nearly double from $324
million in 2018 to $635 million in 2025. This growth is expected to be driven by the high rate of recurrence in pediatrics, increased
incidence in select markets and new high priced therapies coming to the market.
Manufacturing
CLR 131 drug product is made via a five-step
synthetic scheme. The release specifications for the drug product have been established and validated. Through process improvements,
we have been able to achieve a longer expiry dating for the compound extending finished product shelf-life to further facilitate
ex-U.S. distribution from North America.
The drug substance base molecule is a dry
powder produced via a six-step synthetic scheme. The release specifications for the drug substance have been established
and validated. We have successfully executed large scale production of the drug substance via a contract manufacturing organization
that has been inspected and approved by the FDA and the European Medicines Agency. We have also demonstrated 60-month stability
for the drug substance in desiccated and refrigerated forms at small scale and are replicating this at large scale.
In January 2018, we initiated the planned
shutdown of our radiopharmaceutical manufacturing facility in Madison, Wisconsin. This facility was designed to provide pilot and
small-scale production of our lead clinical program CLR 131. In December 2017, we transferred the manufacturing of CLR 131 to Centre
for Probe Development and Commercialization (“CPDC”), a validated Current Good Manufacturing Practices (“cGMPs”)
manufacturing organization specializing in radiopharmaceuticals, as our exclusive source to supply drug product for our ongoing
research and clinical studies, including our Phase 1 and Phase 2 studies of CLR 131. We believe that CPDC and our other third-party
manufactures have the ability to supply large scale clinical and commercial scale material.
Sales and Marketing
We plan to pursue and evaluate all available
options to develop, launch and commercialize our compounds. These options presently include but are not limited to: entering into
an agreement for a contract sales organization (CSO) or partnering arrangement with one or more biotechnology or pharmaceutical
company with strong product development and commercialization expertise and distribution infrastructure in the U.S., Europe and/or
Japan. While we currently do not plan to build our own commercial organization for the launch and commercialization of our compounds,
we may reconsider that in the future.
Competition for Our Clinical-Stage Compounds
Currently, several classes of approved
products with various mechanisms of action exist, including: immune-modulating agents, proteasome inhibitors, histone deacetylase
inhibitors, monoclonal antibodies, corticosteroids, and traditional chemotherapeutics for the treatment of liquid and solid tumors.
While a number of indications were evaluated as the initial target treatment for CLR 131, multiple myeloma and hematologic cancers
were selected for initial clinical development principally because of its highly radio-sensitive nature, single or multi-dose treatment,
and novel mechanism of action relative to all existing classes of approved drugs. As a result, we believe CLR 131 is a therapeutic
option in the relapse or refractory setting either as a monotherapy or in combination with currently approved agents, some of which
are radio-sensitizing and maintain a differential adverse event profile from that of CLR 131.
Intellectual Property
Our core technology platform is based on
research conducted at the University of Michigan in 1994, where phospholipid ether analogs were initially designed, synthesized,
radiolabeled, and evaluated. This research was transferred to the University of Wisconsin - Madison between 1998 and the subsequent
founding of Cellectar in 2002 to further develop and commercialize the technology. We obtained exclusive rights to the related
technology patents owned by University of Michigan in 2003 and continued development of the PDC platform while obtaining ownership
of numerous additional patents and patent applications (with various expiry until 2034 without extensions). We have established
a broad U.S. and international intellectual property rights portfolio around our proprietary cancer-targeting PLE technology platform
including CLR 131 and our PDC Programs.
PDC chemotherapeutic Programs
In November 2015, we converted our previously
filed provisional patent application for Phospholipid-Ether Analogs as Cancer Targeting Drug Vehicles to non-provisional US and
International (PCT) patent applications and were published by the U.S. Patent & Trade Office (USPTO) in May of 2016. These
patent applications further protect composition of matter and method of use for PDCs developed with our proprietary phospholipid-ether
delivery vehicle conjugated with any existing or future cytotoxic agents, including chemotherapeutics for targeted delivery to
cancer cells and cancer stem cells. Additional cytotoxic PDC compounds are covered by pending patent applications directed to the
composition of matter and method of use for cancer therapy provide intellectual property protection in the U.S. and up to 148 additional
countries. These applications, if granted, offer protection extending through at least 2035 in the U.S. and key international markets.
CLR 131
We have taken a broad approach to creating
market exclusivity for CLR 131 both within the U.S., and globally, including all major markets. This approach includes numerous
patents, patent applications and regulatory filings to provide maximum market exclusivity. Our patent portfolio for CLR 131 includes
all of the typical filings as well as unique methods of use, methods of manufacturing, use in combinations, use to treat cancer
stem cells, novel formulations, etc. In addition, to our patents, we were granted orphan designation for CLR 131 for the treatment
of multiple myeloma by the FDA in December 2014 and expect to file additional orphan designations for other rare diseases. We continue
to evaluate CLR 131 in additional hematologic and solid tumor orphan designated indications. Our patents have a variety expected
expiry with some potentially being extended on a country-by-country basis. In 2018, the FDA a granted orphan drug and a Rare Pediatric
Disease Designation (RPDD) for CLR 131 for the treatment of neuroblastoma, rhabdomyosarcoma, Ewing’s sarcoma and osteosarcoma.
We initiated a Phase 1 study in 2019.
We expect to continue to file patent applications
and acquire licenses to other patents covering methods of use, composition of matter, formulation, method of manufacture and other
patentable claims related to CLR 131 and new PDCs. These patent applications will be filed in key commercial markets worldwide.
The issued patents will generally expire between 2025 and 2035, unless extended, most likely under clinical development extensions.
In addition to the above noted patents/applications
directed to CLR 131 and our PDC pipeline portfolio, we own other patents/applications directed to different forms of phospholipid
ethers, methods of use and methods of manufacturing of phospholipid ethers.
Separate from any patent protection and
following product approval by regulatory authorities, data exclusivity may be available for various compounds for up to 10 years
on a country-by-country basis (e.g., up to five years in the U.S. and up to ten years in Europe).
Licenses / Collaborations
In August 2018, we entered into a collaboration
with Orano Med for the development of novel PDCs utilizing Orano Med’s alpha emitter lead-212 conjugated to our phospholipid
ether; the companies intend to evaluate the new PDCs in up to three oncology indications.
In September 2017, we entered into an arrangement
with Onconova Therapeutics, Inc. (Onconova). Under this arrangement, Onconova will provide us a selection of its proprietary compounds.
We will use our proprietary technology to perform research studies on such compounds with the goal of developing new conjugates.
We agreed to perform the studies within 24 months. We granted Onconova an exclusive option to acquire a royalty-bearing license
with respect to each conjugate developed. In the event an executed license agreement for a particular conjugate is not obtained,
then Onconova’s exclusive option shall terminate with respect to such conjugate.
In July 2017, we entered into an arrangement
with Avicenna Oncology GMBH (Avicenna). Under this arrangement, Avicenna will provide us a selection of its proprietary toxins.
We will use our proprietary conjugation capabilities to proceed with the conjugation in order to obtain PDCs. We will process various
in vitro and in cellulo screening against such PDCs to develop new conjugates. We granted Avicenna an exclusive option
to acquire an exclusive license to our intellectual property with respect to each conjugate developed. In the event the parties
cannot reach agreement on the terms of a definitive agreement despite good faith negotiations, Avicenna’s exclusive option
terminates as to such conjugate. Avicenna also granted to us an exclusive option to acquire an exclusive license to its intellectual
property with respect to the material provided. In the event the parties do not reach agreement on the terms of a definitive agreement,
our exclusive option terminates as to the material of Avicenna.
In December 2015, we entered into an arrangement
with Institut de Recherche Pierre Fabre (IRPF). Pierre Fabre is the third largest French pharmaceutical company with an extensive
oncology research and development infrastructure. The objective of the collaboration is to leverage our expertise in conjugation,
linker chemistry and phospholipid ether chemistry to codesign a library of PDCs employing Pierre Fabre’s chemotherapeutics.
The newly developed PDCs may provide enhanced therapeutic indices to otherwise highly potent, nontargeted payloads through the
targeted delivery to cancer cells provided by our proprietary phospholipid ether delivery platform. Research progress has been
achieved, including the demonstration of improved tolerability in animal models. Our agreement with Pierre Fabre expired in January
2019, however, we are still evaluating the program as a number of PDC molecules are eligible for further development and potentially
to be progressed to IND enabling studies.
Research and Development
Our primary activity to date has been research
and development. The research had historically been conducted at our facility in Madison, Wisconsin and through third-party laboratories
and academic universities. Starting in 2018, we no longer used the facility in Madison, Wisconsin for these activities. The clinical
development has been completed primarily through contract research organizations at hospitals and academic centers. We have established
a collaboration outsourcing model to leverage third-party expertise, accelerate project timelines, improve productivity and limit
spend and fixed costs. Our research and development expenses were approximately $8,996,000 and $6,835,000 for 2019 and 2018, respectively.
Regulation
The production, distribution, and marketing
of products employing our technology, and our development activities, are subject to extensive governmental regulation in the U.S.
and in other countries. In the U.S., we are subject to the Federal Food, Drug, and Cosmetic Act, as amended, and the regulations
of the FDA, as well as to other federal, state, and local statutes and regulations, including the federal, state and local laws
and regulations governing the storage, use and disposal of hazardous materials, including radioactive isotopes. These laws,
and similar laws outside the U.S., govern the clinical and pre-clinical testing, manufacture, safety, effectiveness, approval,
labeling, distribution, sale, import, export, storage, record-keeping, reporting, advertising, and promotion of drugs. Product
development and approval within this regulatory framework, if successful, will take many years and involve the expenditure of substantial
resources. Violations of regulatory requirements at any stage may result in various adverse consequences, including the delay
in approving or refusal to approve a product by the FDA or other health authorities. Violations of regulatory requirements
also may result in enforcement actions, which include civil money penalties, injunctions, seizure of regulated product, and civil
and criminal charges. The following paragraphs provide further information on certain legal and regulatory issues with a particular
potential to affect our operations or future marketing of products employing our technology.
Research, Development, and Product Approval
Process
The research, development, and approval
process in the U.S. and elsewhere is intensive and rigorous and generally takes many years to complete. The typical process
required by the FDA before a therapeutic drug may be marketed in the U.S. includes:
|
|
•
|
pre-clinical laboratory and animal tests performed under the FDA’s Good Laboratory Practices regulations, referred to herein as GLP;
|
|
|
•
|
submission to the FDA of an IND application, which must become effective before human clinical studies may commence;
|
|
|
•
|
human clinical studies performed under the FDA’s Good Clinical Practices regulations, to evaluate the drug’s safety and effectiveness for its intended uses;
|
|
|
•
|
FDA review of whether the facility in which the drug is manufactured, processed, packed, or held meets standards designed to assure the product’s continued quality; and
|
|
|
•
|
submission of a marketing application to the FDA, and approval of the application by the FDA.
|
Pre-Clinical Testing
During pre-clinical testing, studies are
performed with respect to the chemical and physical properties of candidate formulations. These studies are subject to GLP requirements.
Biological testing is typically done in animal models to demonstrate the activity of the compound against the targeted disease
or condition and to assess the apparent effects of the new product candidate on various organ systems, as well as its relative
therapeutic effectiveness and safety.
Submission of IND
An IND must be submitted to the FDA and
become effective before studies in humans may commence. The IND must include a sufficient amount of data and other information
concerning the safety and effectiveness of the compound from laboratory, animal, and human clinical testing, as well as data and
information on manufacturing, product quality and stability, and proposed product labeling.
Clinical Studies
Clinical study programs in humans generally
follow a three-phase process. Typically, Phase 1 studies are conducted in small numbers of healthy volunteers or, on occasion,
in patients afflicted with the target disease. Phase 1 studies are conducted to determine the metabolic and pharmacological action
of the product candidate in humans and the side effects associated with increasing doses, and, if possible, to gain early evidence
of effectiveness. In Phase 2, studies are generally conducted in larger groups of patients having the target disease or condition
in order to validate clinical endpoints, and to obtain preliminary data on the effectiveness of the product candidate and optimal
dosing. This phase also helps determine further the safety profile of the product candidate. In Phase 3, large-scale clinical studies
are generally conducted in patients having the target disease or condition to provide sufficient data for the statistical proof
of effectiveness and safety of the product candidate as required by U.S. regulatory agencies.
In the case of products for certain serious
or life-threatening diseases, the initial human testing may be done in patients with the disease rather than in healthy volunteers.
Because these patients are already afflicted with the target disease or condition, it is possible that such studies will also provide
results traditionally obtained in Phase 2 studies. These studies are often referred to as “Phase 1/2” studies. However,
even if patients participate in initial human testing and a Phase 1/2 study is carried out, the sponsor is still responsible for
obtaining all the data usually obtained in both Phase 1 and Phase 2 studies.
U.S. law requires that studies conducted
to support approval for product marketing be “adequate and well controlled.” In general, this means that either a placebo
or a product already approved for the treatment of the disease or condition under study must be used as a reference control. Studies
must also be conducted in compliance with good clinical practice requirements, and informed consent must be obtained from all study
subjects. The clinical study process for a new compound can take ten years or more to complete. The FDA may prevent clinical studies
from beginning or may place clinical studies on hold at any point in this process if, among other reasons, it concludes that study
subjects are being exposed to an unacceptable health risk. Studies may also be prevented from beginning or may be terminated by
institutional review boards, which must review and approve all research involving human subjects. Side effects or adverse events
that are reported during clinical studies can delay, impede, or prevent marketing authorization. Similarly, adverse events that
are reported after marketing authorization can result in additional limitations being placed on a product’s use and, potentially,
withdrawal of the product from the market.
Submission of NDA
Following the completion of clinical studies,
the data is analyzed to determine whether the studies successfully demonstrated safety and effectiveness and whether a product
approval application may be submitted. In the U.S., if the product is regulated as a drug, an NDA must be submitted and approved
before commercial marketing may begin. The NDA must include a substantial amount of data and other information concerning the safety
and effectiveness of the compound from laboratory, animal, and human clinical testing, as well as data and information on manufacturing,
product quality and stability, and proposed product labeling.
Each domestic and foreign manufacturing
establishment, including any contract manufacturers we may decide to use, must be listed in the NDA and must be registered with
the FDA. The application generally will not be approved until the FDA conducts a manufacturing inspection, approves the applicable
manufacturing process and determines that the facility is in compliance with cGMP requirements.
Under the Prescription Drug User Fee Act,
as amended, the FDA receives fees for reviewing an NDA and supplements thereto, as well as annual fees for commercial manufacturing
establishments and for approved products. These fees can be significant. For fiscal year 2019, the NDA review fee alone is $2,588,478,
although certain limited deferral, waivers, and reductions may be available.
Each NDA submitted for FDA approval is
usually reviewed for administrative completeness and reviewability within 45 to 60 days following submission of the application.
If deemed complete, the FDA will “file” the NDA, thereby triggering substantive review of the application. The FDA
can refuse to file any NDA that it deems incomplete or not properly reviewable. The FDA has established performance goals for the
review of NDAs— six months for priority applications and ten months for standard applications. However, the FDA is not legally
required to complete its review within these periods, and these performance goals may change over time.
Moreover, the outcome of the review, even
if generally favorable, typically is not an actual approval but an “action letter” that describes additional work that
must be done before the application can be approved. The FDA’s review of an application may involve review and recommendations
by an independent FDA advisory committee. Even if the FDA approves a product, it may limit the approved therapeutic uses for the
product as described in the product labeling, require that warning statements be included in the product labeling, require that
additional studies be conducted following approval as a condition of the approval, impose restrictions and conditions on product
distribution, prescribing, or dispensing in the form of a risk management plan, or otherwise limit the scope of any approval.
Post NDA Regulation
Significant legal and regulatory requirements
also apply after FDA approval to market under an NDA. These include, among other things, requirements related to adverse event
and other reporting, product advertising and promotion, and ongoing adherence to cGMP requirements, as well as the need to submit
appropriate new or supplemental applications and obtain FDA approval for certain changes to the approved product labeling, or manufacturing
process. The FDA also enforces the requirements of the Prescription Drug Marketing Act which, among other things, imposes various
requirements in connection with the distribution of product samples to physicians.
The regulatory framework applicable to
the production, distribution, marketing and/or sale of our product pipeline may change significantly from the current descriptions
provided herein in the time that it may take for any of our products to reach a point at which an NDA is approved.
Overall research, development, and approval
times depend on a number of factors, including the period of review at FDA, the number of questions posed by the FDA during review,
how long it takes to respond to the FDA’s questions, the severity or life-threatening nature of the disease in question,
the availability of alternative treatments, the availability of clinical investigators and eligible patients, the rate of enrollment
of patients in clinical studies, and the risks and benefits demonstrated in the clinical studies.
Other U.S. Regulatory Requirements
In the U.S., the research, manufacturing,
distribution, sale, and promotion of drug and biological products are potentially subject to regulation by various federal, state,
and local authorities in addition to the FDA, including the Centers for Medicare and Medicaid Services, other divisions of the
U.S. Department of Health and Human Services (e.g., the Office of Inspector General), the U.S. Department of Justice and individual
U.S. Attorney offices within the Department of Justice, and state and local governments. For example, sales, marketing, and scientific/educational
grant programs must comply with the anti-fraud and abuse provisions of the Social Security Act, the False Claims Act, the privacy
provision of the Health Insurance Portability and Accountability Act, and similar state laws, each as amended. Pricing and rebate
programs must comply with the Medicaid rebate requirements of the Omnibus Budget Reconciliation Act of 1990 and the Veterans Health
Care Act of 1992, each as amended. If products are made available to authorized users of the Federal Supply Schedule of the General
Services Administration, additional laws and requirements apply. All of these activities are also potentially subject to federal
and state consumer protection, unfair competition, and other laws.
Our research and development, manufacturing,
and administration of our drugs involve the controlled use of hazardous materials, including chemicals and radioactive materials,
such as radioactive isotopes. Therefore, we are subject to federal, state and local laws and regulations governing the storage,
use and disposal of these materials and some waste products and are required to maintain both a manufacturer’s license and
a radioactive materials license with State of Wisconsin agencies.
Moreover, we are now, and may become subject
to, additional federal, state, and local laws, regulations, and policies relating to safe working conditions, laboratory practices,
the experimental use of animals, and/or the use, storage, handling, transportation, and disposal of human tissue, waste, and hazardous
substances, including radioactive and toxic materials and infectious disease agents used in conjunction with our research work.
Foreign Regulatory Requirements
We, and any future collaborative partners,
may be subject to widely varying foreign regulations that may be quite different from those of the FDA governing clinical studies,
manufacture, product registration and approval, and pharmaceutical sales. Whether or not FDA approval has been obtained, we or
any future collaboration partners must obtain a separate approval for a product by the comparable regulatory authorities of foreign
countries prior to the commencement of product marketing in these countries. In certain countries, regulatory authorities also
establish pricing and reimbursement criteria. The approval process varies from country to country, and the time may be longer or
shorter than that required for FDA approval. In addition, under current U.S. law, there are restrictions on the export of products
not approved by the FDA, depending on the country involved and the status of the product in that country.
Reimbursement and Pricing Controls
In many of the markets where we, or any
future collaborative partners, would commercialize a product following regulatory approval, the prices of pharmaceutical products
are subject to direct price controls by law and to drug reimbursement programs with varying price control mechanisms. Public and
private health care payors control costs and influence drug pricing through a variety of mechanisms, including through negotiating
discounts with the manufacturers and through the use of tiered formularies and other mechanisms that provide preferential access
to certain drugs over others within a therapeutic class. Payors also set other criteria to govern the uses of a drug that will
be deemed medically appropriate and therefore reimbursed or otherwise covered. In particular, many public and private health care
payors limit reimbursement and coverage to the uses of a drug that are either approved by the FDA or that are supported by other
appropriate evidence (for example, published medical literature) and appear in a recognized drug compendium. Drug compendia are
publications that summarize the available medical evidence for particular drug products and identify which uses of a drug are supported
or not supported by the available evidence, whether or not such uses have been approved by the FDA. For example, in the case of
Medicare coverage for physician-administered oncology drugs, the Omnibus Budget Reconciliation Act of 1993, with certain exceptions,
prohibits Medicare carriers from refusing to cover unapproved uses of an FDA-approved drug if the unapproved use is supported by
one or more citations in the American Hospital Formulary Service Drug Information, the American Medical Association Drug Evaluations,
or the U.S. Pharmacopoeia Drug Information. Another commonly cited compendium, for example under Medicaid, is the DRUGDEX Information
System.
Employees
As of May 7, 2020, we had 11 employees,
all of whom are full-time employees.
Legal Proceedings
From time to time, we may become engaged
in litigation or other legal proceedings as part of our ordinary course of business but are not currently party to any litigation
or legal proceedings that, in the opinion of management, are likely to have a material adverse effect on our business.
Corporate Information
Cellectar Biosciences, Inc., formerly known
as Novelos Therapeutics, Inc., was incorporated in Delaware in June 1996. On April 8, 2011, we entered into a business combination
with Cellectar, Inc., a privately held Wisconsin corporation that designed and developed products to detect, treat and monitor
a wide variety of human cancers. On February 11, 2014, we changed our name to Cellectar Biosciences, Inc. Our common stock is listed
on the Nasdaq Capital Market under the symbol CLRB.
Our principal executive offices are located
at 100 Campus Drive, Florham Park, New Jersey 07932 and our telephone number is (608) 441-8120. Our corporate website address is www.cellectar.com.
Information contained on or accessible through our website is not a part of this prospectus.
MANAGEMENT
Our executive officers and directors are
as follows:
|
Name
|
|
Age
|
|
Position
|
|
James V. Caruso
|
|
61
|
|
President, Chief Executive Officer and Director
|
|
Dov Elefant
|
|
52
|
|
Vice President and Chief Financial Officer
|
|
Jarrod Longcor
|
|
47
|
|
Chief Business Officer
|
|
Igor Grachev, M.D., Ph.D.
|
|
58
|
|
Chief Medical Officer
|
|
Douglas J. Swirsky(1)(2)
|
|
50
|
|
Chairman of the Board and Director
|
|
Stephen A. Hill, B.M. B.Ch., M.A., F.R.C.S. (1)(2)
|
|
62
|
|
Director
|
|
John Neis(1)(3)
|
|
64
|
|
Director
|
|
Stefan D. Loren, PhD(2)(3)
|
|
56
|
|
Director
|
|
Frederick W. Driscoll(3)
|
|
69
|
|
Director
|
|
|
(1)
|
Member of the Compensation
Committee.
|
|
|
(2)
|
Member of the Nominating
and Corporate Governance Committee.
|
|
|
(3)
|
Member of the Audit Committee.
|
The following biographical descriptions
are based on information furnished by the respective individual.
James V. Caruso. Mr. Caruso was
appointed our President and Chief Executive Officer and a director in June 2015. He came to Cellectar from Hip Innovation Technology,
a medical device company where he was a founder and served as Executive Vice President and Chief Operating Officer from August
2010 to June 2015, and he currently serves on their board. Prior to his time at Hip Innovation Technology, he was Executive Vice
President and Chief Commercial Officer of Allos Therapeutics, Inc., an oncology company acquired by Spectrum Pharmaceuticals, from
June 2006 to August 2010. He was also Senior Vice President, Sales and Marketing, from June 2002 to May 2005, at Bone Care International,
Inc., a specialty pharmaceutical company that was acquired by Genzyme Corporation. In addition, Mr. Caruso has held key positions
at several well-known pharmaceutical companies, including Novartis, where he was Vice President of Neuroscience Specialty Sales,
BASF Pharmaceuticals-Knoll, where he was Vice President, Sales, and Bristol-Myers Squibb Company in several senior roles. Mr. Caruso
earned a Bachelor of Science degree in Finance from the University of Nevada. Mr. Caruso’s extensive experience
in the biotechnology industry and his recent experience as our Chief Executive Officer make him a highly qualified member of our
Board of Directors.
Jarrod Longcor. Mr. Longcor was
appointed Chief Business Officer of Cellectar in September 2017. He previously served as Senior Vice President of Corporate Development
and Operations since July 2016. Mr. Longcor brings years of pharmaceutical and biotech experience to Cellectar and was previously
the Chief Business Officer for Avillion LLP. In this role, he was responsible for executing the company’s unique co-development
partnership strategy. Prior to Avillion, Jarrod was the Vice President of Corporate Development for Rib-X Pharmaceuticals, Inc.
(now Melinta Therapeutics) where he was responsible for identifying and concluding several critical collaborations for the company,
including a major discovery collaboration with Sanofi Aventis valued over $700M. Prior to Rib-X, Mr. Longcor held key positions
in several small to midsized biotech companies where he was responsible for business development, strategic planning and operations.
Jarrod holds a B.S. from Dickinson College, a M.S. from Boston University School of Medicine and an M.B.A. from Saint Joseph’s
University’s Haub School of Business.
Dov Elefant. Mr. Elefant was
appointed our Vice President and Chief Financial Officer in September 2019. Mr. Elefant has more than 20 years of industry experience
at both public and private biopharmaceutical companies. Most recently, he served as Chief Financial Officer of Akari Therapeutics
PLC, a publicly-traded biopharmaceutical company specializing in treatments for autoimmune and inflammatory diseases, from September
2015 to August 2019. Prior to his service at Akari Therapeutics, PLC, Mr. Elefant served as Chief Financial Officer of Celsus Therapeutics,
Inc., a publicly-traded biopharmaceutical company, from January 2012 to September 2015. Mr. Elefant holds a B.S. in accounting
from Yeshiva University.
Igor Grachev, M.D., Ph.D. Dr.
Grachev was appointed Chief Medical Officer of Cellectar in January 2020. Dr. Grachev brings nearly 20 years of industry experience
to Cellectar having led clinical development programs at both multinational pharmaceutical and biotech organizations. He served
as Global Development Leader and Head of Innovative Clinical Trials Initiative, R&D for TEVA Branded Specialty Pharmaceuticals,
where he was responsible for the development, execution and management of clinical programs worldwide across all phases of clinical
research, achieving regulatory approvals in multiple countries. Dr. Grachev also had clinical development and medical affairs leadership
roles at GE Healthcare, Novartis, GSK, Merck, Schering Plough, Sanofi-Aventis, and BioClinica. Dr. Grachev is a former Assistant
Professor of Radiology at SUNY Upstate Medical University and as a Fellow in Radiology at Massachusetts General Hospital, Harvard
Medical School. Dr. Grachev is well-published in the field of radiopharmaceuticals, drug development and innovative clinical trials.
He also possesses extensive oncology radiopharmaceutical and diagnostic development experience with GE Healthcare where he had
global responsibility for Phase 2-4 clinical trials. Dr. Grachev earned his M.D. with highest honor in General Medicine from Bogomolets
National Medical University, and his Ph.D. in Medical Sciences from the Shupyk National Medical Academy of Postgraduate Education.
Dr. Grachev went on to complete a Fellowship Program in Radiology (including Nuclear Medicine) at Massachusetts General Hospital,
Harvard Medical School jointly with the National Cancer Institute, and also completed his post-doctoral training at Massachusetts
General Hospital Harvard.
Douglas J. Swirsky. Mr. Swirsky
was appointed as a director of Cellectar in April 2017 and Chairman of our Board in August 2017. Since November 2018, Mr. Swirsky
has served as President, Chief Executive Officer, and a director of Rexahn Pharmaceuticals; having previously served as Rexahn’s
President and Chief Financial Officer from January 2018 until his appointment as CEO. Prior to Rexahn, Mr. Swirsky served as President
and Chief Executive Officer of GenVec, Inc., a clinical-stage biopharmaceutical company, from 2014 to June 2017. From 2006 through
2014, Mr. Swirsky served as Senior Vice President, Chief Financial Officer, Treasurer and Corporate Secretary of GenVec. Prior
to joining GenVec in September 2006, Mr. Swirsky worked at Stifel Nicolaus where he served as a Managing Director and the Head
of Life Sciences Investment Banking. Mr. Swirsky previously held investment banking positions at UBS, PaineWebber, Morgan Stanley,
and Legg Mason. His experience also includes positions in public accounting and consulting. Mr. Swirsky received his undergraduate
degree in business administration from Boston University and his M.B.A. from the Kellogg School of Management at Northwestern University.
Mr. Swirsky is a Certified Public Accountant and a CFA® charterholder. Within the past five years, Mr. Swirsky has also served
on the board Fibrocell Science, Inc., Pernix Therapeutics Holdings, Inc. and GenVec, Inc. Our Board concluded that Mr. Swirsky
should serve as a director and member of Compensation Committee along with Nominating and Corporate Governance Committee because
of his distinguished career in financial services and corporate management, including his investment banking experience and his
experience serving as a principal executive officer and principal financial officer.
Stephen A. Hill. Stephen A. Hill.
Dr. Hill has been a member of the Board of Directors since 2007, and served as its Chairman from 2007 until 2015. Dr. Hill was
appointed Chief Executive Officer of Faraday Pharmaceuticals, Inc. in September 2015. Dr. Hill was the President and CEO of Targacept
Inc. from December 2012 until the company merged with Catalyst Biosciences, Inc. in August 2015. Dr. Hill was the President and
CEO of 21CB, a nonprofit initiative of UPMC designed to provide the United States government with a domestic solution for its biodefense
and infectious disease biologics portfolio, from March 2011 until December 2011. Dr. Hill served as the President and Chief Executive
Officer of Solvay Pharmaceuticals, Inc. from April 2008 until its acquisition by Abbott Laboratories in 2010. Prior to joining
Solvay, Dr. Hill had served as ArQule’s President and Chief Executive Officer since April 1999. Prior to his tenure at ArQule,
Dr. Hill was the Head of Global Drug Development at F. Hoffmann-La Roche Ltd. from 1997 to 1999. Dr. Hill joined Roche in 1989
as Medical Adviser to Roche Products in the United Kingdom. He held several senior positions at Roche, including Medical Director
where he was responsible for clinical trials of compounds across a broad range of therapeutic areas, including CNS, HIV, cardiovascular,
metabolic and oncology products. Subsequently, he served as Head of International Drug Regulatory Affairs at Roche headquarters
in Basel, Switzerland, where he led the regulatory submissions for seven major new chemical entities. Dr. Hill also was a member
of Roche’s Portfolio Management, Research, Development and Pharmaceutical Division Executive Boards. Prior to Roche, Dr.
Hill served seven years with the National Health Service in the United Kingdom in General and Orthopedic Surgery. Dr. Hill has
served on the Board of Directors of Lipocine, Inc. since January 2014. Dr. Hill is a Fellow of the Royal College of Surgeons of
England and holds his scientific and medical degrees from St. Catherine’s College at Oxford University. Dr. Hill chairs the
Compensation Committee. Dr. Hill’s extensive experience in a broad range of senior management positions with companies in
the life sciences sector makes him a highly qualified member of our Board of Directors along with member of Nominating and Corporate
Governance Committee.
John Neis. Mr. Neis been a member
of the Board of Directors since 2008. Mr. Neis is a Managing Director of Venture Investors LLC, where he leads the firm and heads
the firm’s Health Care practice. He also serves on the Board of Directors of privately held Prevacept Infection Control,
Inc., Delphinus Medical Technologies, Inc., TAI Diagnostics, Inc. and Health Scholars, Inc. He serves on the Board of Directors
of the Wisconsin Technology Council, the science and technology advisor to Wisconsin’s Governor and Legislature, and the
Wisconsin Venture Capital Association. He serves on the Board of Trustees at the Morgridge Institute for Research. He also serves
on the Weinert Applied Ventures Program Advisory Board in the School of Business and chairs the Tandem Press Advisory Board in
the School of Education at the University of Wisconsin – Madison. He holds a B.S. in finance from the University of Utah,
and received a M.S. in Marketing and Finance from the University of Wisconsin – Madison. He is a Chartered Financial Analyst.
Mr. Neis’ extensive experience leading emerging companies and his financial experience makes him a highly qualified member
of our Board of Directors as well as member of the Audit and Compensation Committees.
Stefan D. Loren, Ph.D. Dr. Loren
began serving as director of Cellectar in June 2015. Dr. Loren is currently a managing director with Oppenheimer and Company’s
healthcare investment banking group. Prior, he was the founder and managing member of Loren Capital Strategy (LCS), a strategic
consulting and investment firm focused on life science companies since February 2014. Prior to LCS, he headed the life science
practice of Westwicke Partners, a healthcare-focused consulting firm from July 2008 to February 2014. Prior to joining Westwicke,
he worked as an Analyst/Portfolio Manager with Perceptive Advisors, a health care hedge fund, and MTB Investment Advisors, a long-term
oriented family of equity funds. His focus areas included biotechnology, specialty pharmaceuticals, life science tools, and health
care service companies. Prior to moving to the buy side, Dr. Loren was Managing Director, Health Care Specialist/Desk Analyst for
Legg Mason where he discovered, evaluated, and communicated investment opportunities in the health care area to select clients.
In addition, he assisted both advising management teams on strategic options. He started his Wall Street career as a sell side
analyst at Legg Mason covering biotechnology, specialty pharmaceuticals, life science tools, pharmaceuticals, and chemistry outsourcing
companies. In his research career, Dr. Loren was an early member of Abbott Laboratories Advanced Technologies Division, analyzing
and integrating new technological advances in Abbott’s pharmaceutical research. Before industry, he was a researcher at The
Scripps Research Institute, working with Nobel Laureate K. Barry Sharpless on novel synthetic routes to chiral drugs. Dr. Loren
received a doctorate in Organic Chemistry from the University of California at Berkeley and an undergraduate degree in Chemistry
from UCSD. His scientific work has been featured in Scientific American, Time, Newsweek, and Discover, as well as other periodicals
and journals. Dr. Loren is Chair of the Nominating and Corporate Governance Committee and member of the Audit Committee. Dr. Loren’s
extensive experience in the biotechnology and financial industries make him a highly qualified member of our Board of Directors.
Frederick W. Driscoll. Mr. Driscoll
was appointed as a director of Cellectar in April 2017. Mr. Driscoll served as Chief Financial Officer at Flexion Therapeutics
from 2013 to 2017, spearheading an initial public offering in 2014. Prior to joining Flexion, he was Chief Financial Officer at
Novavax, Inc., a publicly traded biopharmaceutical company from 2009 to 2013. From 2008 to 2009, Mr. Driscoll served as Chief Executive
Officer of Genelabs Technologies, Inc., a publicly traded biopharmaceutical and diagnostics company later acquired by GlaxoSmithKline.
He previously served as Genelabs’ Chief Financial Officer from 2007 to 2008. From 2000 to 2006, Mr. Driscoll served as Chief
Executive Officer at OXiGENE, Inc., a biopharmaceutical company. Mr. Driscoll has also served as Chairman of the Board and Audit
Committee Chair at OXiGENE and as a member of the Audit Committee for Cynapsus, which was sold to Sunovion Pharmaceuticals in 2016.
Mr. Driscoll earned a Bachelor’s degree in accounting and finance from Bentley University. Mr. Driscoll is a member of the
board of directors of Cue Biopharma, MEI Pharma and NantKwest. Mr. Driscoll chairs the Audit Committee and our Board concluded
that Mr. Driscoll should serve as a director because of his significant corporate management and board experience at multiple biotechnology
companies as well as his strong financial background.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL
OWNERS AND MANAGEMENT
At the close of business on May 5, 2020,
there were 9,396,015 shares of our common stock outstanding. The following table provides information regarding beneficial ownership
of our common stock as of May 5, 2020:
|
|
·
|
each person known by us to be the beneficial owner of more than 5% of our common stock;
|
|
|
·
|
each executive officer named in the summary compensation table; and
|
|
|
·
|
all of our current directors and executive officers as a group.
|
The address of each executive officer and
director is c/o Cellectar Biosciences, Inc., 100 Campus Drive, Florham Park, New Jersey 07932, except as otherwise indicated. The
persons named in this table have sole voting and investment power for the shares listed, except as otherwise indicated. In these
cases, the information respecting voting and investment power has been provided to us by the security holder. The identification
of natural persons having voting or investment power over securities held by a beneficial owner listed in the table below does
not constitute an admission of beneficial ownership of any such natural person. Shares included in the “Right to Acquire”
column consist of shares that may be purchased through the exercise of options or warrants that are exercisable within 60 days
of May 5, 2020.
|
Name and Address of Beneficial Owner
|
|
Outstanding
|
|
|
Right to Acquire
|
|
|
Total
|
|
|
Percentage
|
|
|
Boxer Capital, LLC (1)
|
|
|
931,764
|
|
|
|
—
|
|
|
|
931,764
|
|
|
|
9.92
|
%
|
|
North Sound Management, Inc. (2)
|
|
|
925,000
|
|
|
|
—
|
|
|
|
925,000
|
|
|
|
9.84
|
%
|
|
James V. Caruso
|
|
|
22,524
|
|
|
|
159,763
|
|
|
|
182,287
|
|
|
|
1.94
|
%
|
|
Dov Elefant
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
*
|
|
|
Jarrod Longcor
|
|
|
7,300
|
|
|
|
66,221
|
|
|
|
73,521
|
|
|
|
*
|
|
|
Igor Grachev, M.D., Ph.D.
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
*
|
|
|
Frederick W. Driscoll
|
|
|
—
|
|
|
|
20,500
|
|
|
|
20,500
|
|
|
|
*
|
|
|
Stephen A. Hill
|
|
|
—
|
|
|
|
23,041
|
|
|
|
23,041
|
|
|
|
*
|
|
|
Stefan Loren, Ph.D.
|
|
|
—
|
|
|
|
22,850
|
|
|
|
22,850
|
|
|
|
*
|
|
|
John Neis (3)
|
|
|
62,610
|
|
|
|
79,783
|
|
|
|
142,393
|
|
|
|
1.52
|
%
|
|
Douglas Swirsky
|
|
|
—
|
|
|
|
30,250
|
|
|
|
30,250
|
|
|
|
*
|
|
All directors and officers as a group
(9 persons)
|
|
|
92,434
|
|
|
|
402,408
|
|
|
|
494,842
|
|
|
|
5.27
|
%
|
|
(1)
|
As reported in Schedule 13G/A filed with the SEC on February 7, 2020. Based on such 13G/A filing, North Sound Management, Inc. has sole voting power over 925,000 shares, shared voting power over 0 shares, sole dispositive power over 925,000 shares and shared dispositive power over 0 shares. Brad Miller has sole voting power over 925,000 shares, shared voting power over 0 shares, sole dispositive power over 925,000 shares and shared dispositive power over 0 shares. North Sound Trading, LP has sole voting power over 925,000 shares, shared voting power over 0 shares, sole dispositive power over 925,000 shares and shared dispositive power over 0 shares. The address of North Sound Management, Inc. is 115 East Putnam Avenue, Greenwich, Connecticut 06830.
|
|
(2)
|
As reported in Schedule 13G filed with the SEC on May 28, 2019. Based on such 13G filing, Boxer Capital, LLC has sole voting power over 0 shares, shared voting power over 931,764 shares, sole dispositive power over 0 shares and shared dispositive power over 931,764 shares. Boxer Asset Management Inc. has sole voting power over 0 shares, shared voting power over 931,764 shares, sole dispositive power over 0 shares and shared dispositive power over 931,764 shares. Joe Lewis has sole voting power over 0 shares, shared voting power over 931,764 shares, sole dispositive power over 0 shares and shared dispositive power over 931,764 shares. The address of Boxer Capital, LLC is 115 11682 El Camino Real, Suite 320, San Diego, CA 92130.
|
|
(3)
|
Consists of shares of common stock held by Venture Investors Early Stage Fund IV Limited Partnership and Advantage Capital Wisconsin Partners I, Limited Partnership. VIESF IV GP LLC is the general partner of Venture Investors Early Stage Fund IV Limited Partnership and Venture Investors LLC is the submanager and special limited partner of Advantage Capital Wisconsin Partners I, Limited Partnership. The investment decisions of VIESF IV GP LLC and Venture Investors LLC are made collectively by five managers, including Mr. Neis. Each such manager and Mr. Neis disclaim such beneficial ownership except to the extent of his pecuniary interest therein. The address of Mr. Neis is c/o Venture Investors LLC, 505 South Rosa Road, #201, Madison, Wisconsin 53719. Shares in the “Right to Acquire” column consist of 56,808 shares of common stock issuable upon the exercise of warrants held by Venture Investors Early Stage Fund IV Limited and Advantage Capital Wisconsin Partners I, Limited Partnership and common stock issuable upon options to purchase 9,291 shares of common stock issued to Mr. Neis in his capacity as director. Shares in the “Right to Acquire” column consist of shares of common stock issuable upon the exercise of warrants at exercise prices ranging from $15.00 to $283.00 per share expiring between August 20, 2019 and November 29, 2021.
|
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
We do not have a written policy for the
review, approval or ratification of transactions with related parties or conflicted transactions. When such transactions arise,
they are referred to the Audit Committee or the Board of Directors for consideration. During 2019, there were no related party
transactions.
DESCRIPTION OF SECURITIES
The following summary description of
our common stock is based on the provisions of our Second Amended and Restated Certificate of Incorporation, as amended, which
we refer to as our certificate of incorporation or charter, our by-laws, and the applicable provisions of the Delaware General
Corporation Law, which we refer to as the DGCL. This description may not contain all of the information that is important to you
and is subject to, and is qualified in its entirety by reference to, our certificate of incorporation, our by-laws and the applicable
provisions of the DGCL. For information on how to obtain copies of our certificate of incorporation and by-laws, see “Where
You Can Find More Information.”
Authorized and Outstanding Capital Stock
Our authorized capital stock consists of
80,000,000 shares of common stock, $0.00001 par value per share and 7,000 shares of preferred stock, $0.00001 par value per share.
Our certificate of incorporation, as amended, authorizes us to issue shares of our preferred stock from time to time in one or
more series without stockholder approval, each such series to have rights and preferences, including voting rights, dividend rights,
conversion rights, redemption privileges and liquidation preferences, as our Board may determine. The rights of the holders of
common stock will be subject to, and may be adversely affected by, the rights of holders of any preferred stock that we may issue
in the future. The issuance of preferred stock, while providing desirable flexibility in connection with possible acquisitions
and other corporate purposes, could have the effect of making it more difficult for others to acquire, or of discouraging others
from attempting to acquire, a majority of our outstanding voting stock.
As of May 5, 2020, there were 9,396,015
shares of common stock outstanding and 215 shares of preferred stock outstanding. All outstanding shares of our common stock and
preferred stock are duly authorized, validly issued, fully paid and nonassessable.
Common Stock
Voting. Holders
of our common stock are entitled to one vote per share held of record on all matters to be voted upon by our stockholders. Our
common stock does not have cumulative voting rights. Persons who hold a majority of the outstanding common stock entitled to vote
on the election of directors can elect all of the directors who are eligible for election.
Dividends. Subject
to preferences that may be applicable to the holders of any outstanding shares of our preferred stock, the holders of our common
stock are entitled to receive such lawful dividends as may be declared by our Board.
Liquidation and Dissolution. In
the event of our liquidation, dissolution or winding up, and subject to the rights of the holders of any outstanding shares of
our preferred stock, the holders of shares of our common stock will be entitled to receive pro rata all of our remaining assets
available for distribution to our stockholders.
Other Rights and Restrictions. Our
charter prohibits us from granting preemptive rights to any of our stockholders.
Preferred Stock
Series A Preferred Stock, Series B Preferred
Stock and Series C Preferred Stock
Our Board has designated 68 shares of our
preferred stock as Series A Convertible Preferred Stock (“Series A Preferred Stock”), none of which are currently outstanding.
The preferences and rights of the Series A Preferred Stock are set forth in a Certificate of Designation of Preferences, Rights
and Limitations of Series A Convertible Preferred Stock.
Our Board has designated 42 shares of our
preferred stock as Series B Convertible Preferred Stock (“Series B Preferred Stock”), none of which are currently issued
and outstanding. The preferences and rights of the Series B Preferred Stock are set forth in a Certificate of Designation of Preferences,
Rights and Limitations of Series B Convertible Preferred Stock.
Our Board has designated 1,114 shares of
our preferred stock as Series C Convertible Preferred Stock (“Series C Preferred Stock”), 215 of which are currently
issued and outstanding. The preferences and rights of the Series C Preferred Stock are set forth in a Certificate of Designation
of Preferences, Rights and Limitations of Series C Convertible Preferred Stock.
Pre-Funded Warrants to be Issued as Part of this Offering
Duration and Exercise Price. Each
pre-funded warrant offered hereby will have an initial exercise price per share equal to $0.00001. The pre-funded warrants will
be immediately exercisable and may be exercised at any time until the pre-funded warrants are exercised in full. The exercise price
and number of shares of common stock issuable upon exercise is subject to appropriate adjustment in the event of stock dividends,
stock splits, reorganizations or similar events affecting our common stock and the exercise price. The pre-funded warrants will
be issued separately from the accompanying common warrants, and may be transferred separately immediately thereafter.
Exercisability. The
pre-funded warrants will be exercisable, at the option of each holder, in whole or in part, by delivering to us a duly executed
exercise notice accompanied by payment in full for the number of shares of our common stock purchased upon such exercise (except
in the case of a cashless exercise as discussed below). Purchasers of the pre-funded warrants in this offering may elect to deliver
their exercise notice following the pricing of the offering and prior to the issuance of the pre-funded warrants at closing to
have their pre-funded warrants exercised immediately upon issuance and receive shares of common stock underlying the pre-funded
warrants upon closing of this offering. A holder (together with its affiliates) may not exercise any portion of the pre-funded
warrant to the extent that the holder would own more than 4.99% of the outstanding common stock immediately after exercise, except
that upon at least 61 days’ prior notice from the holder to us, the holder may increase the amount of ownership of outstanding
stock after exercising the holder’s pre-funded warrants up to 9.99% of the number of shares of our common stock outstanding
immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of the
pre-funded warrants. Purchasers of pre-funded warrants in this offering may also elect prior to the issuance of the pre-funded
warrants to have the initial exercise limitation set at 9.99% of our outstanding common stock. No fractional shares of common stock
will be issued in connection with the exercise of a pre-funded warrant. In lieu of fractional shares, we will round down to the
next whole share.
Cashless Exercise. If,
at the time a holder exercises its pre-funded warrants, a registration statement registering the issuance of the shares of common
stock underlying the pre-funded warrants under the Securities Act is not then effective or available, then in lieu of making the
cash payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the holder
may elect instead to receive upon such exercise (either in whole or in part) the net number of shares of common stock determined
according to a formula set forth in the pre-funded warrants.
Transferability. Subject
to applicable laws, a pre-funded warrant may be transferred at the option of the holder upon surrender of the pre-funded warrant
to us together with the appropriate instruments of transfer.
Exchange Listing. There
is no trading market available for the pre-funded warrants on any securities exchange or nationally recognized trading system.
We do not intend to list the pre-funded warrants on any securities exchange or nationally recognized trading system.
Right as a Stockholder. Except
as otherwise provided in the pre-funded warrants or by virtue of such holder’s ownership of shares of our common stock, the
holders of the pre-funded warrants do not have the rights or privileges of holders of our common stock, including any voting rights,
until they exercise their pre-funded warrants.
Fundamental Transaction. In
the event of a fundamental transaction, as described in the pre-funded warrants and generally including any reorganization, recapitalization
or reclassification of our common stock, the sale, transfer or other disposition of all or substantially all of our properties
or assets, our consolidation or merger with or into another person, the acquisition of more than 50% of our outstanding common
stock, or any person or group becoming the beneficial owner of 50% of the voting power represented by our outstanding common stock,
the holders of the pre-funded warrants will be entitled to receive upon exercise of the pre-funded warrants the kind and amount
of securities, cash or other property that the holders would have received had they exercised the pre-funded warrants immediately
prior to such fundamental transaction.
Series H Warrants to be Issued as Part of this Offering
General. The
warrants offered in this offering will be issued in the form of Series H Warrant filed as an exhibit to the registration statement
of which this prospectus is a part. You should review a copy of the form of warrant for a complete description of the terms and
conditions applicable to the warrants. The following is a brief summary of the Series H Warrant and is subject in all respects
to the provisions contained in the form of warrant. Pursuant to a warrant agency agreement between us and American Stock Transfer
and Trust Company, as warrant agent, the Series H Warrant will be issued in book-entry form and initially will be represented by
one or more global certificates deposited with The Depository Trust Company, or DTC, and registered in the name of Cede & Co.,
a nominee of DTC, or as otherwise directed by DTC. We do not plan to list the Series H Warrants on the Nasdaq Capital Market, any
other national securities exchange or any other nationally recognized trading system.
Fractional Shares; Rights as a Stockholder.
No fractional shares of common stock will be issued in connection with the exercise of a Series H Warrant. Warrant amounts will
be rounded either up to the next whole share. A Series H Warrant may be transferred by a holder, upon surrender of the warrant,
properly endorsed (by the holder executing an assignment in the form attached to the warrant). Prior to the exercise of any warrants
to purchase common stock, holders of the warrants will not have any of the rights of holders of the common stock purchasable upon
exercise, including the right to vote, except as set forth therein.
Duration and Exercise Price.
Each Series H Warrant represents the right to purchase shares of common stock
at an exercise price equal to $ , subject to adjustment as described below. Each Series H Warrant may be exercised
on or after the closing date of this offering through and including the close of business on the fifth anniversary of the date
of issuance. Each Series H Warrant will have a cashless exercise right in the event that the shares of common stock underlying
such warrants are not covered by an effective registration statement at the time of such exercise.
Adjustments; Fundamental Transaction. The
exercise price and the number of shares underlying the Series H Warrants are subject to appropriate adjustment in the event of
stock splits, stock dividends on our common stock, stock combinations or similar events affecting our common stock. In addition,
in the event we consummate a merger or consolidation with or into another person or other reorganization event in which our common
shares are converted or exchange for securities, cash or other property, or we sell, lease, license, assign, transfer, convey or
otherwise dispose of all or substantially all of our assets or we or another person acquire 50% or more of our outstanding common
shares, then following such event, the holders of the warrants will be entitled to receive upon exercise of the warrants the same
kind and amount of securities, cash or property which the holders would have received had they exercised the warrants immediately
prior to such fundamental transaction. Any successor to us or surviving entity will assume the obligations under the warrants.
Amendment and Waiver. Amendments
and waivers of the terms of the Series H Warrants require the written consent of the holder of such warrant and us.
Anti-Takeover Effect of Certain Charter and By-Law Provisions
Provisions of our charter and by-laws could
make it more difficult to acquire us by means of a merger, tender offer, proxy contest, open market purchases, removal of incumbent
directors and otherwise. These provisions, which are summarized below, are expected to discourage types of coercive takeover practices
and inadequate takeover bids and to encourage persons seeking to acquire control of us to first negotiate with us. We believe that
the benefits of increased protection of our potential ability to negotiate with the proponent of an unfriendly or unsolicited proposal
to acquire or restructure us outweigh the disadvantages of discouraging takeover or acquisition proposals because negotiation of
these proposals could result in an improvement of their terms.
Authorized but Unissued Stock.
We have shares of common stock and preferred stock available for future issuance, in some cases, without stockholder approval.
We may issue these additional shares for a variety of corporate purposes, including public offerings to raise additional capital,
corporate acquisitions, stock dividends on our capital stock or equity compensation plans. The existence of unissued and unreserved
common stock and preferred stock may enable our Board to issue shares to persons friendly to current management or to issue preferred
stock with terms that could render more difficult or discourage a third-party attempt to obtain control of us, thereby protecting
the continuity of our management. In addition, if we issue preferred stock, the issuance could adversely affect the voting power
of holders of common stock and the likelihood that such holders will receive dividend payments and payments upon liquidation.
Amendments to By-laws. Our
certificate of incorporation and by-laws authorize the Board to amend, repeal, alter or rescind the by-laws at any time without
stockholder approval. Allowing the Board to amend our by-laws without stockholder approval enhances Board control over our by-laws.
Classification of Board; Removal
of Directors; Vacancies. Our certificate of incorporation provides for the division of the Board into three
classes as nearly equal in size as possible with staggered three-year terms; that directors may be removed only for cause by the
affirmative vote of the holders of two-thirds of our shares of capital stock entitled to vote; and that any vacancy on the Board,
however occurring, including a vacancy resulting from an enlargement of the Board, may be filled only by the vote of a majority
of the directors then in office. The limitations on the removal of directors and the filling of vacancies could have the effect
of making it more difficult for a third party to acquire, or of discouraging a third party from acquiring, control of us. Our certificate
of incorporation requires the affirmative vote of the holders of at least 75% of our shares of capital stock issued and outstanding
and entitled to vote to amend or repeal any of these provisions.
Notice Periods for Stockholder Meetings.
Our by-laws provide that for business to be brought by a stockholder before an annual meeting of stockholders, the stockholder
must give written notice to the corporation not less than 90 nor more than 120 days prior to the one-year anniversary of the date
of the annual meeting of stockholders of the previous year; provided, however, that in the event that the annual meeting of stockholders
is called for a date that is not within 30 days before or after such anniversary date, notice by the stockholder must be received
not later than the close of business on the tenth day following the day on which the corporation’s notice of the date of
the meeting is first given or made to the stockholders or disclosed to the general public, whichever occurs first.
Stockholder Action; Special Meetings.
Our certificate of incorporation provides that stockholder action may not be taken by written action in lieu of a meeting and provides
special meetings of the stockholders may be called only by our president or Board. These provisions could have the effect of delaying
until the next stockholders’ meeting stockholder actions that are favored by the holders of a majority of our outstanding
voting securities. These provisions may also discourage another person or entity from making a tender offer for our common stock,
because that person or entity, even if it acquired a majority of our outstanding voting securities, would be able to take action
as a stockholder only at a duly called stockholders’ meeting, and not by written consent. Our certificate of incorporation
requires the affirmative vote of the holders of at least 75% of our shares of capital stock issued and outstanding and entitled
to vote to amend or repeal the provisions relating to prohibition on action by written consent and the calling of a special meeting
of stockholders.
Nominations. Our by-laws provide
that nominations for election of directors may be made only by: (i) the Board or a committee appointed by the Board; or (ii) a
stockholder entitled to vote on director election, if the stockholder provides notice to the Secretary of the corporation presented
not less than 90 days nor more than 120 days prior to the anniversary of the last annual meeting (subject to the limited exceptions
set forth in the by-laws). These provisions may deter takeovers by requiring that any stockholder wishing to conduct a proxy contest
have its position solidified well in advance of the meeting at which directors are to be elected and by providing the incumbent
Board with sufficient notice to allow it to put an election strategy in place.
Nasdaq Capital Market Listing
Our common stock is listed for trading
and quotation on the Nasdaq Capital Market under the trading symbol “CLRB.” Certain warrants to purchase shares of our common stock expiring on November 29, 2021, are listed on the Nasdaq Capital Market
under the trading symbol “CLRBZ.”
Transfer Agent and Registrar
The transfer agent and registrar for our
common stock is American Stock Transfer and Trust Company.
UNDERWRITING
We entered into
an underwriting agreement with Oppenheimer & Co. Inc. on , 2020. Oppenheimer &
Co. Inc. is acting as the sole book-running manager for this offering. The underwriting agreement provides for the purchase of
a specific number of shares of common stock and/or pre-funded warrants and accompanying Series H Warrants to purchase shares of
common stock by the underwriter. Subject to the terms and conditions of the underwriting agreement, the underwriter has agreed
to purchase the number of shares set forth below:
|
Underwriter
|
|
Number of
Shares of Common Stock
|
|
|
Number of
Pre-Funded Warrants
|
|
|
Number of
Series H Warrants
|
|
|
Oppenheimer & Co. Inc.
|
|
|
|
|
|
|
|
|
|
|
|
|
The underwriter
has agreed to purchase all of the shares of common stock and/or pre-funded warrants and accompanying Series H Warrants offered
by this prospectus, if any are purchased.
The shares of
common stock and/or pre-funded warrants and accompanying Series H Warrants offered hereby are expected to be ready for delivery
on or about , 2020 against payment in immediately available funds.
The underwriter
is offering the shares of common stock and/or pre-funded warrants and accompanying Series H Warrants subject to various conditions
and may reject all or part of any order. The underwriter proposes initially to offer the shares of common stock and/or pre-funded
warrants and accompanying Series H Warrants to purchase shares of common stock to the public at the public offering price set forth
on the cover page of this prospectus and to dealers at a price less a concession not in excess of $
per share and accompanying Series H Warrant or $ per pre-funded warrant and accompanying
Series H Warrant, based on the combined public offering price per share and accompanying Series H Warrant or pre-funded warrant
and accompanying Series H Warrant, to brokers and dealers. After the shares of common stock and/or pre-funded warrants and accompanying
Series H Warrants are released for sale to the public, the underwriter may change the offering price, the concession, and other
selling terms at various times.
The following
table provides information regarding the amount of the discounts and commissions to be paid to the underwriter by us, before expenses:
|
|
|
Per Share
|
|
|
Per
Pre-Funded
Warrant
|
|
|
Per
Series H
Warrant
|
|
|
Total Per
Share and
Accompanying
Series H Warrant
|
|
|
Total Per
Pre-Funded Warrant
and Accompanying
Series H Warrant
|
|
|
Public offering price
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Underwriting discounts and commissions(1)
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Proceeds, before expenses, to us
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
|
(1)
|
We have agreed to pay the
underwriter a commission of 7.0% of the gross proceeds of this offering.
|
We estimate that our
total expenses of the offering, excluding the estimated underwriting discounts and commissions, will be approximately $ ,
which includes the fees and expenses for which we have agreed to reimburse the underwriter, provided that any such fees and expenses
in excess of an aggregate of $ will be subject to our prior written approval.
Rules of the Securities
and Exchange Commission may limit the ability of the underwriter to bid for or purchase shares before the distribution of the shares
is completed. However, the underwriter may engage in the following activities in accordance with the rules:
|
|
•
|
Stabilizing transactions
- the underwriter may make bids or purchases for the purpose of pegging, fixing or maintaining the price of the shares, so long
as stabilizing bids do not exceed a specified maximum.
|
|
|
•
|
Penalty bids - if the
underwriter purchases shares in the open market in a stabilizing transaction or syndicate covering transaction, it may reclaim
a selling concession from the underwriter and selling group members who sold those shares as part of this offering.
|
|
|
•
|
Passive market making
- market makers in the shares who are underwriters or prospective underwriters may make bids for or purchases of shares, subject
to limitations, until the time, if ever, at which a stabilizing bid is made.
|
Similar to other
purchase transactions, the underwriter's purchases to cover the syndicate short sales or to stabilize the market price of our common
stock may have the effect of raising or maintaining the market price of our common stock or preventing or mitigating a decline
in the market price of our common stock. As a result, the price of the shares of our common stock may be higher than the price
that might otherwise exist in the open market. The imposition of a penalty bid might also have an effect on the price of the shares
if it discourages resales of the shares.
Neither we nor
the underwriter make any representation or prediction as to the effect that the transactions described above may have on the price
of the shares. These transactions may occur on The Nasdaq Capital Market or otherwise. If such transactions are commenced, they
may be discontinued without notice at any time.
Electronic Delivery
of Prospectus: A prospectus in electronic format may be delivered to potential investors by the underwriter. The prospectus in
electronic format will be identical to the paper version of such prospectus. Other than the prospectus in electronic format, the
information on any underwriter's website and any information contained in any other website maintained by an underwriter is not
part of this prospectus or the registration statement of which this prospectus forms a part.
Over-allotment Option
We have granted to the underwriter an option,
exercisable not later than 30 days after the date of this prospectus, to purchase up to a number of additional shares of common
stock equal to 15% of the number of shares of common stock and shares of common stock underlying the pre-funded warrants sold in
the primary offering and/or up to a number of additional warrants to purchase shares of common stock equal to 15% of the number
of warrants sold in the primary offering, in any combination. Any shares so purchased will be sold at a price per share equal to
the public offering price, less the underwriting discount. Any warrants so purchased will be sold at a price per warrant equal
to the public offering price less the underwriting discount. The underwriter may exercise the option solely to cover over-allotments,
if any, made in connection with this offering. If any additional shares of common stock and/or warrants are purchased pursuant
to the over-allotment option, the underwriter will offer these shares of common stock and/or warrants on the same terms as those
on which the other securities are being offered hereby. The over-allotment option may be used to purchase shares of common stock,
preferred stock or warrants, or any combination thereof, as determined by the underwriter.
Determination of Offering Price
Our common stock is currently traded on
the Nasdaq Capital Market under the symbol “CLRB.” On May 7, 2020, the closing price of our common stock was $1.62
per share.
The public offering price of the securities
offered by this prospectus will be determined by negotiation between us and the underwriter. Among the factors considered in determining
the public offering price of the shares were:
|
|
·
|
our history and our prospects;
|
|
|
·
|
the industry in which we operate;
|
|
|
·
|
our past and present operating results;
|
|
|
·
|
the previous experience of our executive officers; and
|
|
|
·
|
the general condition of the securities markets at the time of this offering
|
The offering price stated on the cover
page of this prospectus should not be considered an indication of the actual value of the securities. That price is subject to
change as a result of market conditions and other factors, and we cannot assure you that the securities can be resold at or above
the public offering price.
Lock-up Agreements
Our officers and directors have agreed
with the underwriter to be subject to a lock-up period of 90 days following the date of this prospectus. This means that, during
the applicable lock-up period, such persons may not offer for sale, contract to sell, sell, distribute, grant any option, right
or warrant to purchase, pledge, hypothecate or otherwise dispose of, directly or indirectly, any shares of our common stock or
any securities convertible into, or exercisable or exchangeable for, shares of our common stock. Certain limited transfers are
permitted during the lock-up period if the transferee agrees to these lock-up restrictions. We have also agreed, in the underwriting
agreement, to similar lock-up restrictions on the issuance and sale of our securities for 90 days following the closing of this
offering, although we will be permitted to issue stock options or stock awards to directors, officers and employees under our existing
plans. The lock-up period is subject to an additional extension to accommodate for our reports of financial results or material
news releases. The underwriter may, in its sole discretion and without notice, waive the terms of any of these lock-up agreements.
Other Relationships
Upon
completion of an offering that meets certain criteria, we have granted the underwriter a right of first refusal to act as lead
underwriter, lead initial purchaser, lead placement agent or lead selling agent, as the case may be, in connection with any subsequent
financing by us. This right of first refusal extends for nine months from the effective date of this registration statement. The
terms of any such engagement of the underwriter will be determined by separate agreement.
Transfer Agent and Registrar
The transfer
agent and registrar for our common stock is American Stock Transfer and Trust Company.
Indemnification
We have agreed to indemnify the Oppenheimer
& Co. and selected dealers against certain liabilities, including certain liabilities arising under the Securities Act, or
to contribute to payments that the underwriter or selected dealers may be required to make for these liabilities.
LEGAL MATTERS
The validity of the securities being offered
by this prospectus has been passed upon for us by Michael Best & Friedrich LLP, Madison, Wisconsin. Ellenoff Grossman &
Schole LLP, New York, New York, is acting as counsel to the underwriter in this offering.
EXPERTS
The audited financial statements incorporated
by reference in this prospectus and elsewhere in the registration statement have been so incorporated by reference in reliance
upon the report of Baker Tilly Virchow Krause, LLP, independent registered public accountants, upon the authority of said firm
as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We are a reporting company and file annual,
quarterly and special reports, and other information with the SEC. Copies of the reports and other information may be read and
copied at the SEC’s Public Reference Room at 100 F Street NE, Washington, D.C. 20549. You can request copies of such documents
by writing to the SEC and paying a fee for the copying cost. You may obtain information on the operation of the Public Reference
Room by calling the SEC at 1-800-SEC-0330. The SEC maintains a web site at http://www.sec.gov that contains reports, proxy and
information statements and other information regarding registrants that file electronically with the SEC.
This prospectus is part of a registration
statement on Form S-1 that we filed with the SEC. Certain information in the registration statement has been omitted from this
prospectus in accordance with the rules and regulations of the SEC. We have also filed exhibits and schedules with the registration
statement that are excluded from this prospectus. For further information you may:
|
|
·
|
read a copy of the registration statement, including the exhibits and schedules, without charge at the SEC’s Public Reference
Room; or
|
|
|
·
|
obtain a copy from the SEC upon payment of the fees prescribed by the SEC.
|
We are subject to the information and reporting
requirements of the Exchange Act and, in accordance with this law, are required to file periodic reports, proxy statements and
other information with the SEC. We make available free of charge, on or through the investor relations section of our website,
annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed
or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act as soon as reasonably practicable after we electronically file
such material with, or furnish it to, the SEC. The information found on our website, other than as specifically incorporated by
reference in this prospectus, is not part of this prospectus.
INCORPORATION OF DOCUMENTS BY REFERENCE
The SEC allows us to “incorporate
by reference” information into this prospectus. This means that we can disclose important information to you by referring
you to another document filed separately with the SEC. The information incorporated by reference is considered to be a part of
this prospectus, except for any information that is superseded by other information that is included in this prospectus.
We incorporate by reference into this prospectus
the following document, which we have previously filed with the SEC:
|
|
·
|
our Annual Report on Form 10-K for the fiscal year ended December 31, 2019, filed with the SEC on March 9, 2020;
|
|
|
·
|
our Quarterly Report on Form 10-Q for the quarter year ended March 31, 2020, filed with the SEC on May 7, 2020;
|
|
|
·
|
our Definitive Proxy Statement on Schedule 14A for the annual meeting of stockholders, filed with the SEC on April 28, 2020;
|
|
|
·
|
our Current Report on Form 8-K, filed with the SEC on January 7, 2020; and
|
|
|
·
|
the description of our securities contained in our Registration Statement on Form 8-A filed on April 18, 2016, including any
amendment or report filed for the purpose of updating such description.
|
In addition, all documents subsequently
filed by us pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act prior to the termination of the offering will be
deemed to be incorporated by reference into this prospectus.
You should rely only on the information
contained in this prospectus, as updated and supplemented by any prospectus supplement, or that information to which this prospectus
or any prospectus supplement has referred you by reference. We have not authorized anyone to provide you with any additional information.
Any statement contained in a document incorporated
or deemed to be incorporated by reference herein will be deemed to be modified or superseded for purposes of this prospectus to
the extent that a statement contained herein modifies or supersedes such statement. Any statement so modified or superseded will
not be deemed, except as so modified or superseded, to constitute a part of this prospectus.
You may request and obtain a copy of any
of the filings incorporated herein by reference, at no cost, by writing or telephoning us at the following address or phone number:
Cellectar Biosciences,
Inc.
100 Campus Drive
Florham Park, New Jersey
07932
Attention: Chief Financial
Officer (608) 441-8120
GLOSSARY OF CERTAIN SCIENTIFIC TERMS
Cytotoxic — Cytotoxicity
is the quality of being toxic to cells (i.e. cell-killing). Many cancer chemotherapeutic drugs are cytotoxic to cancer cells (and,
to some extent, normal cells) thus resulting in unwanted side-effects, e.g. nausea/vomiting, hair loss, suppression of the immune
system.
Dexamethasone — Dexamethasone
is a corticosteroid (cortisone-like medicine or steroid). It works on the immune system to help relieve swelling, redness, itching
and allergic reactions and is used in the treatment of numerous medical conditions.
Dosimetry — Radiation
dosimetry is the calculation of absorbed dose and optimization of dose delivery in radiation therapy.
Lipid rafts — Lipid
rafts are specialized regions of the membrane phospholipid bilayer that contain high concentrations of cholesterol and sphingolipids
and serve to organize cell surface and intracellular signaling molecules (e.g. growth factor and cytokine receptors, the phosphatidylinositol
3-kinase (P13K)/Akt survival pathway).
Orphan drug status — Orphan
drug status confers seven years of marketing exclusivity under the Federal Food, Drug, and Cosmetic Act, and up to 10 years of
marketing exclusivity in Europe for a particular product in a specified indication.
Radiolabeled — Radiolabeled
refers to a molecule containing a radioisotope as a part of its structure.
Radioisotope — Also
referred to as radioactive isotopes or radionuclides, radioisotopes are variants of atoms of particular chemical elements (e.g.
iodine) with an unstable nucleus that can undergo radioactive decay during which ionizing radiation (e.g. gamma rays, subatomic
particles) is emitted.
Uptake — Uptake
is an act of taking in or absorbing, especially into a living organism, tissue or cell.
Xenograft — Xenograft
is a graft of tissue, organs or cells from an individual of one species transplanted into or grafted onto an individual of another
species.
PROSPECTUS
Shares of Common Stock,
Pre-Funded Warrants to Purchase Shares of Common Stock and
Series H Warrants to Purchase Shares of Common Stock

Sole Book-Running Manager
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution
We estimate that expenses in connection
with the distribution described in this registration statement (other than brokerage commissions, discounts or other expenses relating
to the sale of the shares by the selling stockholders) will be as set forth below. We will pay all of the expenses with respect
to the distribution, and such amounts, with the exception of the Securities and Exchange Commission registration fee and FINRA
fee, are estimates.
|
SEC registration fee
|
|
$
|
|
|
FINRA filing fee
|
|
|
|
|
Accounting fees and expenses
|
|
|
|
|
Legal fees and expenses
|
|
|
|
|
Printing and related expenses
|
|
|
|
|
Miscellaneous
|
|
|
|
|
Total
|
|
|
|
Item 14. Indemnification of Directors and Officers
Our charter contains provisions to indemnify
our directors and officers to the maximum extent permitted by Delaware law. We believe that indemnification under our charter covers
at least negligence on the part of an indemnified person. Our charter permits us to advance expenses incurred by an indemnified
person in connection with the defense of any action or proceeding arising out of the person’s status or service as our director,
officer, employee or other agent upon an undertaking by the person to repay those advances if it is ultimately determined that
the person is not entitled to indemnification.
Insofar as indemnification for liabilities
arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to
the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission
such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable.
The
underwriting agreement to be filed as Exhibit 1.1 to this Registration
Statement provides for indemnification by the underwriter of us and our directors and officers for certain liabilities under the
Securities Act, or otherwise.
Item 15. Recent Sales of Unregistered Securities
May 2019 Private Placement
On May 20, 2019, the Company issued and
sold 1,982,000 shares of common stock at an offering price of $2.50 per share. In a concurrent private placement, we issued to
the purchasers of our common stock, Series F warrants to purchase an aggregate of 1,982,000 shares of common stock. The Series
F warrants were immediately exercisable, expire five years after the date of issuance, and have an exercise price of $2.40.
In a separate concurrent private placement
transaction, the Company sold 2,018,000 shares of common stock together with Series G warrants to purchase an aggregate of up to
2,018,000 shares of common stock. The shares of common stock and Series G warrants were priced at $2.50 per fixed combination.
The warrants sold in the private placement were immediately exercisable, expire five years after the date of issuance, and have
an exercise price of $2.40.
Gross offering proceeds to the Company
were $10 million, with net proceeds to the Company of approximately $9.0 million after deducting placement agent fees and related
offering expenses. The unregistered common shares and warrants were offered pursuant to the exemption from registration afforded
by Section 4(a)(2) under the Securities Act and Regulation D promulgated thereunder. Such common shares, warrants and common shares
issuable upon exercise of such warrants had not been registered under the Securities Act, and therefore could not be offered or
sold in the United States. The offerings’ unregistered common shares and warrants were ultimately registered through our
May 31, 2019 filing of Form S-1 and acceptance of this Registration Statement by the SEC.
2017 Private Placement
On October 12, 2017, we issued and sold,
in a registered offering by us directly to the purchasers, an aggregate of 1,954,388 shares of common stock at an offering price
of $1.87375 per share. We also sold to those purchasers whose purchase of shares of common stock in the registered offering would
result in beneficial ownership of more than 4.99% of our outstanding common stock following the offering, the opportunity to purchase,
in lieu of the shares of our common stock that would result in ownership in excess of 4.99%, 41.0412949 shares of Series B Convertible
Preferred Stock convertible into an aggregate of 2,190,330 shares of common stock, convertible at any time at the holder’s
option into that number of shares of common stock equal to $100,000 divided by $1.87375, at a public offering price of $100,000
per share. Gross proceeds of the sale of the common stock and preferred stock were approximately $7.76 million before deducting
the placement agent fee and related offering expenses. In a concurrent private placement, we issued to the purchasers who participated
in the offering, Series D warrants to purchase an aggregate of 3,108,538 shares of common stock at an exercise price of $1.78 per
share. Each Series D warrant was immediately exercisable and expires seven years from the date of issuance. The private placement
of the Series D warrants was structured to comply with the requirements of Section 4(a)(2) under the Securities Act and Rule 506(b)
promulgated thereunder.
2017 Grant to Employees
During the three months ended June 30,
2017, we issued 46,000 shares of restricted stock to members of our executive team, 14,667 of which have since been forfeited.
The restricted stock was granted at a grant date fair value of either $20.80 or $21.00 per share, which was the closing price of
the stock on the date of issuance, and vested in equal annual amounts over three years. The restricted stock was issued in reliance
on the exemption from registration provided in Section 4(a)(2) of the Securities Act, for transactions not involving any public
offering. The restricted stock was subsequently registered pursuant to a reoffer prospectus on Form S-8 filed on November 9, 2017.
Item 16. Exhibits.
(a) Exhibits.
The exhibits to the registration statement
are listed in the Exhibit Index attached hereto and incorporated by reference herein.
(b) Financial Statement Schedules.
Financial statement schedules have been
omitted, as the information required to be set forth therein is included in the consolidated financial statements or notes thereto
appearing in the prospectus made part of this registration statement.
EXHIBIT INDEX
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Incorporated by Reference
|
Exhibit
No.
|
|
Description
|
|
Form
|
|
Filing Date
|
|
Exhibit
No.
|
|
2.1
|
|
Agreement and Plan of Merger by and among Novelos Therapeutics, Inc., Cell Acquisition Corp. and Cellectar, Inc. dated April 8, 2011
|
|
8-K
|
|
April 11, 2011
|
|
2.1
|
|
3.1
|
|
Second Amended and Restated Certificate of Incorporation
|
|
8-K
|
|
April 11, 2011
|
|
3.1
|
|
3.2
|
|
Certificate of Ownership and Merger of Cellectar Biosciences, Inc. with and into Novelos Therapeutics, Inc.
|
|
8-K
|
|
February 13, 2014
|
|
3.1
|
|
3.3
|
|
Certificate of Amendment to Second Amended and Restated Certificate of Incorporation
|
|
8-K
|
|
June 13, 2014
|
|
3.1
|
|
3.4
|
|
Certificate of Amendment to Second Amended and Restated Certificate of Incorporation
|
|
8-K
|
|
June 19, 2015
|
|
3.2
|
|
3.5
|
|
Certificate of Amendment to Second Amended and Restated Certificate of Incorporation
|
|
8-K
|
|
March 4, 2016
|
|
3.1
|
|
3.6
|
|
Certificate of Amendment to Second Amended and Restated Certificate of Incorporation
|
|
8-K
|
|
June 1, 2017
|
|
3.2
|
|
3.7
|
|
Certificate of Amendment of Second Amended and Restated Certificate of Incorporation
|
|
8-K
|
|
July 13, 2018
|
|
3.1
|
|
3.8
|
|
Amended and Restated By-laws
|
|
8-K
|
|
June 1, 2011
|
|
3.1
|
|
3.9
|
|
Form of Certificate of Designation of Series C Preferred Stock
|
|
S-1/A
|
|
July 18, 2018
|
|
3.11
|
|
4.1
|
|
Form of common stock certificate
|
|
S-1/A
|
|
November 9, 2011
|
|
4.1
|
|
4.2
|
|
Form of Series C Preferred Stock certificate
|
|
S-1/A
|
|
July 18, 2018
|
|
4.7
|
|
4.3†
|
|
Form of Series H Warrant
|
|
|
|
|
|
|
|
4.4†
|
|
Form of Pre-Funded Warrant
|
|
|
|
|
|
|
|
5.1†
|
|
Opinion of Michael Best & Friedrich LLP
|
|
|
|
|
|
|
|
10.1
|
|
2006 Stock Incentive Plan, as amended **
|
|
8-K
|
|
December 18, 2013
|
|
10.1
|
|
10.2
|
|
Form of Non-Statutory Stock Option under Novelos Therapeutics, Inc.’s 2006 Stock Incentive Plan**
|
|
8-K
|
|
December 15, 2006
|
|
10.2
|
|
10.3
|
|
Form of Convertible Debenture
|
|
8-K
|
|
February 10, 2014
|
|
4.1
|
|
10.4
|
|
Form of Series B Pre-Funded Warrant
|
|
8-K
|
|
September 30, 2015
|
|
4.1
|
|
10.5
|
|
Registration Rights Agreement dated September 28, 2015
|
|
8-K
|
|
September 30, 2015
|
|
10.2
|
|
10.6
|
|
Amendment and Exchange Agreement dated April 13, 2016
|
|
S-1/A
|
|
April 14, 2016
|
|
10.43
|
|
10.7
|
|
Form of Series A Warrant
|
|
S-1/A
|
|
April 14, 2016
|
|
4.2
|
|
10.8
|
|
Form of Series B Pre-Funded Warrant
|
|
S-1/A
|
|
April 14, 2016
|
|
4.3
|
|
10.9
|
|
Form of Warrant Agency Agreement
|
|
S-1/A
|
|
April 14, 2016
|
|
4.4
|
|
10.10
|
|
Form of Series C Warrant
|
|
S-1/A
|
|
November 18, 2016
|
|
4.3
|
|
10.11
|
|
Form of Warrant Agency Agreement
|
|
S-1/A
|
|
November 18, 2016
|
|
4.4
|
|
10.12
|
|
Form of Restricted Common Stock Agreement**
|
|
10-Q
|
|
August 14, 2017
|
|
10.1
|
|
10.13
|
|
Securities Purchase Agreement, dated as of October 10, 2017, by and among Cellectar Biosciences, Inc. and the Purchasers
|
|
8-K
|
|
October 11, 2017
|
|
10.1
|
|
10.14
|
|
Form of Series D Common Stock Purchase Warrant
|
|
8-K
|
|
October 11, 2017
|
|
4.1
|
|
10.15
|
|
Registration Rights Agreement, dated as of October 10, 2017, by and among Cellectar Biosciences, Inc. and the Purchasers
|
|
8-K
|
|
October 11, 2017
|
|
10.2
|
|
10.16
|
|
Form of Non-Statutory Stock Option**
|
|
S-8
|
|
November 9, 2017
|
|
10.2
|
|
10.17
|
|
Stock Option Agreement with James V. Caruso**
|
|
S-8
|
|
November 9, 2017
|
|
10.4
|
|
10.18
|
|
Stock Option Agreement with Jarrod Longcor**
|
|
S-8
|
|
November 9, 2017
|
|
10.5
|
|
10.19
|
|
Master Services Agreement for Clinical Research and Related Services between the Company and INC Research, LLC dated October 6, 2016*
|
|
10-K
|
|
March 21, 2018
|
|
10.33
|
|
10.20
|
|
Series E Common Stock Purchase Warrant
|
|
S-1/A
|
|
July 18, 2018
|
|
4.5
|
|
10.21
|
|
Form of Warrant Agency Agreement
|
|
S-1/A
|
|
July 18, 2018
|
|
4.7
|
|
10.22
|
|
Agreement of Lease between the Company and KBS II 100-200 Campus Drive, LLC
|
|
S-1/A
|
|
July 18, 2018
|
|
10.35
|
|
10.23
|
|
Form of Non-Statutory Stock Option (Definitive/Contingent – Employees)**
|
|
10-Q
|
|
November 13, 2018
|
|
10.3
|
|
10.24
|
|
Form of Non-Statutory Stock Option (Definitive/Contingent – Directors)**
|
|
10-Q
|
|
November 13, 2018
|
|
10.4
|
|
10.25
|
|
Amended and Restated Employment Agreement between the Company and James Caruso, dated April 15, 2019**
|
|
10-Q
|
|
May 6, 2019
|
|
10.1
|
|
10.26
|
|
Amended and Restated Employment Agreement between the Company and Jared Longcor, dated April 15, 2019**
|
|
10-Q
|
|
May 6, 2019
|
|
10.2
|
|
10.27
|
|
Placement Agency Agreement between the Company and Roth Capital Partners, LLC dated May 16, 2019
|
|
8-K
|
|
May 20, 2019
|
|
1.1
|
|
10.28
|
|
Form of Series F Common Stock Purchase Warrant
|
|
8-K
|
|
May 20, 2019
|
|
4.1
|
|
10.29
|
|
Form of Series G Common Stock Purchase Warrant
|
|
8-K
|
|
May 20, 2019
|
|
4.2
|
|
10.30
|
|
Securities Purchase Agreement, dated as of May 16, 2019, by and among Cellectar Biosciences, Inc. and the Purchasers
|
|
8-K
|
|
May 20, 2019
|
|
10.1
|
|
10.31
|
|
Private Placement Securities Purchase Agreement, dated as of May 16, 2019, by and among Cellectar Biosciences, Inc. and the Purchasers
|
|
8-K
|
|
May 20, 2019
|
|
10.2
|
|
10.32
|
|
Registration Rights Agreement, dated as of May 16, 2019, by and among Cellectar Biosciences, Inc. and the Purchasers
|
|
8-K
|
|
May 20, 219
|
|
10.3
|
|
10.33
|
|
Cellectar Biosciences, Inc. Amended and Restated Stock Incentive Plan**
|
|
8-K
|
|
June 14, 2019
|
|
10.1
|
|
10.34
|
|
Employment Agreement between the Company and Dov Elefant dated August 15, 2019**
|
|
10-Q
|
|
November 12, 2019
|
|
10.1
|
|
10.35
|
|
Amendment to Amended and Restated Employment Agreement between the Company and Jarrod Longcor dated November 10, 2019**
|
|
10-Q
|
|
November 12, 2019
|
|
10.2
|
|
10.36
|
|
Stock Option Agreement with Dov Elefant
|
|
10-K
|
|
March 9, 2020
|
|
10.36
|
|
10.37
|
|
Stock Option Agreement with Igor Grachev**
|
|
10-K
|
|
March 9, 2020
|
|
10.37
|
|
21.1*
|
|
List of Subsidiaries
|
|
|
|
|
|
|
|
23.1*
|
|
Consent of Independent Registered Public Accounting Firm
|
|
|
|
|
|
|
|
23.2†
|
|
Consent of Michael Best & Friedrich LLP (included in Exhibit 5.1)
|
|
|
|
|
|
|
|
24.1*
|
|
Power of Attorney (included on signature page)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
†
|
To be filed by amendment.
|
|
*
|
Filed herewith.
|
|
**
|
Management contract or compensatory plan or arrangement.
|
Item 17. Undertakings.
The undersigned registrant hereby undertakes
to provide to the underwriter at the closing specified in the underwriting agreement certificates in such denominations and registered
in such names as required by the underwriter to permit prompt delivery to each purchaser.
(a) The undersigned registrant hereby undertakes:
1. To file, during any period
in which offers or sales are being made, a post-effective amendment to this registration statement:
|
|
(i)
|
To include any prospectus required by section 10(a)(3) of the Securities Act of 1933;
|
|
|
(ii)
|
To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most
recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information
set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered
(if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or
high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant
to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than 20% change in the maximum aggregate
offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement.
|
2. That, for the purpose of
determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration
statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the
initial bona fide offering thereof.
3. To remove from registration
by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
4. That, for the purpose of
determining liability under the Securities Act of 1933 to any purchaser, each prospectus filed pursuant to Rule 424(b) as part
of a registration statement relating to the offering shall be deemed to be part of and included in the registration statement as
of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus
that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration
statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior
to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of
the registration statement or made in any such document immediately prior to such date of first use.
5. That, for the purpose of
determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities:
The undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration
statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or
sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser
and will be considered to offer or sell such securities to such purchaser:
|
|
(i)
|
Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant
to Rule 424;
|
|
|
(ii)
|
Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred
to by the undersigned registrant;
|
|
|
(iii)
|
The portion of any other free writing prospectus relating to the offering containing material information about the undersigned
registrant or its securities provided by or on behalf of the undersigned registrant; and
|
|
|
(iv)
|
Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
|
(b) The undersigned
registrant hereby undertakes that, for purposes of determining any liability under the Securities Act of 1933, each filing of the
registrant's annual report pursuant to section 13(a) or section 15(d) of the Securities Exchange Act of 1934 (and, where applicable,
each filing of an employee benefit plan's annual report pursuant to section 15(d) of the Securities Exchange Act of 1934) that
is incorporated by reference in the registration statement shall be deemed to be a new registration statement relating to the securities
offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
(c) Insofar as
indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling
persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion
of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Act and is, therefore,
unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant
of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action,
suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered,
the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court
of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Act and
will be governed by the final adjudication of such issue.
SIGNATURES
Pursuant to the requirements of the Securities
Act of 1933, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto
duly authorized, in the City of Florham Park, State of New Jersey, on May 8, 2020.
|
|
CELLECTAR BIOSCIENCES, INC.
|
|
|
|
|
|
By:
|
/s/ James V. Caruso
|
|
|
|
James V. Caruso
|
|
|
|
President and Chief Executive Officer
|
POWER OF ATTORNEY
We, the undersigned officers and directors
of Cellectar Biosciences, Inc., hereby severally constitute and appoint James V. Caruso and Dov Elefant, and each of them singly
(with full power to each of them to act alone), our true and lawful attorneys-in-fact and agents, with full power of substitution
and resubstitution in each of them for him and in his name, place and stead, and in any and all capacities, to sign for us and
in our names in the capacities indicated below any and all amendments (including post-effective amendments) to this registration
statement (or any other registration statement for the same offering that is to be effective upon filing pursuant to Rule 462(b)
under the Securities Act of 1933, as amended), and to file the same, with all exhibits thereto and other documents in connection
therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full
power and authority to do and perform each and every act and thing requisite or necessary to be done in and about the premises,
as full to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact
and agents or any of them, or their or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
|
Signature
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
/s/James V. Caruso
|
|
Chief Executive Officer and Director
|
|
May 8, 2020
|
|
James V. Caruso
|
|
(principal executive officer)
|
|
|
|
|
|
|
|
|
|
/s/ Dov Elefant
|
|
Chief Financial Officer
|
|
May 8, 2020
|
|
Dov Elefant
|
|
(principal financial officer and
|
|
|
|
|
|
principal accounting officer)
|
|
|
|
|
|
|
|
|
|
/s Frederick W. Driscoll
|
|
Director
|
|
May 8, 2020
|
|
Frederick W. Driscoll
|
|
|
|
|
|
|
|
|
|
|
|
/s/ Stephen A. Hill
|
|
Director
|
|
May 8, 2020
|
|
Stephen A. Hill
|
|
|
|
|
|
|
|
|
|
|
|
/s/ Stefan D. Loren, Ph.D.
|
|
Director
|
|
May 8, 2020
|
|
Stefan D. Loren, Ph.D.
|
|
|
|
|
|
|
|
|
|
|
|
/s/ John Neis
|
|
Director
|
|
May 8, 2020
|
|
John Neis
|
|
|
|
|
|
|
|
|
|
|
|
/s/ Douglas J. Swirsky
|
|
Director
|
|
May 8, 2020
|
|
Douglas J. Swirsky
|
|
|
|
|
Cellectar Biosciences (NASDAQ:CLRB)
Historical Stock Chart
From Aug 2024 to Sep 2024
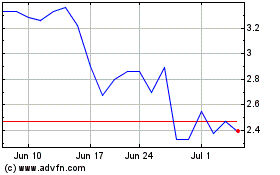
Cellectar Biosciences (NASDAQ:CLRB)
Historical Stock Chart
From Sep 2023 to Sep 2024