U.K. Drugmaker Charged -- WSJ
April 11 2019 - 3:02AM
Dow Jones News
Prosecutors seek at least $3 billion from Indivior, which has
denied the allegations
By Maria Armental
This article is being republished as part of our daily
reproduction of WSJ.com articles that also appeared in the U.S.
print edition of The Wall Street Journal (April 11, 2019).
Federal prosecutors charged a U.K. pharmaceutical company with
organizing a multibillion-dollar fraud to drive up sales of
Suboxone Film, an opioid-addiction treatment.
As part of an indictment filed Tuesday in Virginia, the Justice
Department said Indivior PLC set up a telephone and online program
intended to connect callers with doctors that Indivior knew were
prescribing Suboxone or opioids in a "careless and clinically
unwarranted manner."
The criminal charges come as the U.S. government has taken a
more aggressive approach to fighting the opioid crisis, with
thousands of lawsuits filed against opioid manufacturers and
distributors.
In 2017, President Trump declared the opioid crisis a public
health emergency and has taken steps to crack down on international
and domestic drug-supply chains.
The Justice Department is seeking at least $3 billion and
control of other property from Indivior, previously the
pharmaceuticals business of Reckitt Benckiser Group PLC. Indivior,
which became a stand-alone company in 2014, denied the charges. In
a statement, it accused the Justice Department of pursuing
"self-serving headlines on a matter of national significance."
The company said it worked with the Justice Department for years
as part of its investigation and "made numerous attempts to reach a
settlement that went far beyond what we believe the facts of this
case support."
"Indivior's top priority has always been the treatment of
patients struggling with opioid addiction," the company said.
"Indivior does not make pain pills in the U.S. and is not a
contributor to the opioid crisis."
The government says that for more than a decade, the company
played down the risks of Suboxone Film, a prescription medicine
that dissolves in the mouth and contains the active ingredients
buprenorphine and naloxone. For example, prosecutors said,
promotional statements described the product as being safer with a
"lower risk of child exposure" and "less divertable and abusable,"
meaning it is less likely to be sold illegally, shared or
smuggled.
However, the government said company managers discussed that
"under no circumstances" could claims be made that the product is
safer or better at reducing pediatric exposures among other things.
Prosecutors said managers "did not contact patients, physicians,
pharmacists, health care benefit programs, or others to correct or
retract the promotional statements Indivior salespeople had already
made."
Write to Maria Armental at maria.armental@wsj.com
(END) Dow Jones Newswires
April 11, 2019 02:47 ET (06:47 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
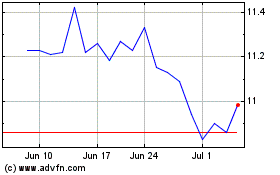
Reckitt Benckiser (PK) (USOTC:RBGLY)
Historical Stock Chart
From Mar 2024 to Apr 2024
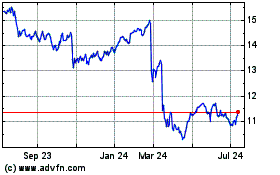
Reckitt Benckiser (PK) (USOTC:RBGLY)
Historical Stock Chart
From Apr 2023 to Apr 2024