Intercept Pharmaceuticals Gets Orphan Drug Status for OCA and Bezafibrate to Treat Primary Biliary Cholangitis
May 16 2023 - 6:14PM
Dow Jones News
By Mary de Wet
Intercept Pharmaceuticals said Tuesday it received the Food and
Drug Administration's orphan drug designation for a combination of
obeticholic acid and bezafibrate to treat a rare bile-duct
disease.
Intercept has two Phase 2 studies to explore a range of
therapeutic doses for the combination of OCA and bezafibrate, the
biopharmaceutical company said.
The company expects to complete interim analyses from the
studies later this year.
The orphan-drug designation was given for a fixed dose to treat
primary biliary cholangitis, a chronic autoimmune disease that
affects the bile ducts in the liver and is most prevalent in women
over the age of 40.
The FDA's orphan-drug program gives special status to drugs and
biologics for diseases and disorders that affect fewer than 200,000
people in the U.S. and provides for an extended marketing
exclusivity period against competition.
Write to Mary de Wet at mary.dewet@dowjones.com
(END) Dow Jones Newswires
May 16, 2023 17:59 ET (21:59 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
Intercept Pharmaceuticals (NASDAQ:ICPT)
Historical Stock Chart
From May 2024 to Jun 2024
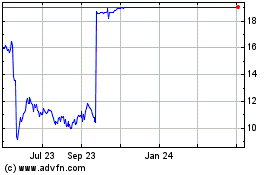
Intercept Pharmaceuticals (NASDAQ:ICPT)
Historical Stock Chart
From Jun 2023 to Jun 2024