AtriCure Announces the First Patient Enrolled in the ICE-AFIB™ Clinical Trial
February 05 2019 - 8:00AM
Business Wire
Trial will evaluate the safety and effectiveness of AtriCure’s
cryoICE® Ablation System for the treatment of persistent and
long-standing persistent atrial fibrillation in open concomitant
cardiac surgery
AtriCure, Inc. (Nasdaq: ATRC), a leading innovator in surgical
treatment for atrial fibrillation (Afib) and left atrial appendage
management, today announced the first patient was treated in the
ICE-AFIB™ trial (NCT03732794). Following Investigational Device
Exemption (IDE) approval by the FDA, the first patient was treated
by Dr. Niv Ad, at Washington Adventist Hospital in Takoma Park,
Maryland. The trial will evaluate the safety and effectiveness of
the cryoICE Ablation System for the treatment of persistent and
long-standing persistent atrial fibrillation during concomitant
open-chest cardiac surgery.
The ICE-AFIB trial is a prospective, multicenter, single-arm
study of up to 150 patients at up to 20 U.S. centers. The study
will enroll patients with persistent and long-standing persistent
atrial fibrillation undergoing cardiac surgical procedure(s) for
heart valve repair or replacement and/or coronary artery bypass
procedures. The study defines persistent and long-standing
persistent atrial fibrillation in accordance with the Heart Rhythm
Society (HRS) 2017 AF expert consensus statement.
The cryoICE system in conjunction with an AtriClip® left atrial
appendage (LAA) occlusion device will be used to perform the
Cox-Maze III lesion set in this trial. Primary effectiveness is
defined as freedom from Afib, atrial flutter, and/or atrial
tachycardia lasting greater than 30 seconds and will be evaluated
12 months after the procedure. Long-term effectiveness will be
evaluated three years post procedure.
“Cryothermal energy has been a mainstay of surgical ablation for
a long time. The ICE-AFIB IDE trial is the first of its kind
designed to assess the safety and sustained effectiveness of
cryothermal ablation as a standalone energy source for surgical AF
ablation,” said Dr. Niv Ad, Director of Clinical Research at
Washington Adventist Hospital and the national principal
investigator of ICE-AFIB.
“While AtriCure’s ABLATE™ clinical trial confirmed that the
usage of the Isolator® Synergy™ Ablation System (radiofrequency
ablation (heat)) when supplemented with select lesions created with
cryothermal energy (cold) is an effective treatment for persistent
and long-standing persistent Afib during cardiac surgery, the
ICE-AFIB trial is a unique opportunity to generate systematic
clinical evidence on the safety and effectiveness of cryosurgery
for the treatment of such patients,” said Michael Carrel, President
and Chief Executive Officer of AtriCure. “The ICE-AFIB trial is yet
another testament to AtriCure’s commitment to and leadership in
improving the lives of atrial fibrillation patients undergoing
cardiac surgery.”
The cryoICE ablation system was 510(k) cleared by the FDA in
2009 for the cryosurgical treatment of cardiac arrhythmias by
freezing target tissues, which creates an inflammatory response
(cryonecrosis) that blocks the electrical conduction pathway.
About AtriCure, Inc.
AtriCure, Inc. provides innovative technologies for the
treatment of Afib and related conditions. Afib affects more than 33
million people worldwide. Electrophysiologists and cardiothoracic
surgeons around the globe use AtriCure technologies for the
treatment of Afib and reduction of Afib related complications.
AtriCure’s Isolator Synergy Ablation System is the first and only
medical device to receive FDA approval for the treatment of
persistent Afib. AtriCure’s AtriClip Left Atrial Appendage (LAA)
Exclusion System products are the most widely sold LAA management
devices worldwide, with more than 170,000 implanted to date. For
more information, visit AtriCure.com or follow us on Twitter
@AtriCure.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190205005541/en/
Valerie Storch-WillhausMedia RelationsSenior Director, Corporate
Marketing and Communications(612)
605-3311vstorch-willhaus@AtriCure.com
Andy WadeInvestor RelationsSenior Vice President and Chief
Financial Officer(513) 755-4564awade@AtriCure.com
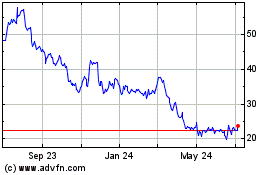
AtriCure (NASDAQ:ATRC)
Historical Stock Chart
From Mar 2024 to Apr 2024
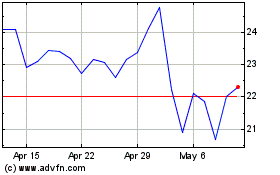
AtriCure (NASDAQ:ATRC)
Historical Stock Chart
From Apr 2023 to Apr 2024