Addex ADX71149 Receives IND Approval to Start a Phase 2a Clinical Study in Epilepsy
January 21 2021 - 1:00AM

First
patient treated with ADX71149 (JNJ-40411813) scheduled for Q2
2021
Geneva, Switzerland, January 21,
2021 – Addex Therapeutics (SIX: ADXN and Nasdaq: ADXN), a
clinical-stage pharmaceutical company pioneering allosteric
modulation-based drug discovery and development, today announced
that its partner Janssen Pharmaceuticals, Inc., part of the Janssen
Pharmaceutical Companies of Johnson & Johnson, has received
FDA’s Investigational New Drug (IND) approval to begin a Phase 2a
proof of concept study with the selective metabotropic glutamate
type 2 (mGlu2) receptor positive allosteric modulator (PAM),
JNJ-40411813 (ADX71149), in patients with epilepsy. The first
patient is expected to be treated during Q2 2021.
“IND approval for the mGlu2 epilepsy study is
great news to start 2021 and will be the first of three clinical
studies we expect to be initiate this year,” said Tim Dyer, CEO of
Addex. “There is a great opportunity for an allosteric modulation
approach in this difficult disease. We’d like to thank the team
involved for their continued support and moving quickly to get this
clinical study ready for patient enrolment.”
The multi-center study will assess the efficacy,
safety, tolerability and pharmacokinetics of adjunctive
JNJ-40411813 (ADX71149) administration in patients with focal onset
seizures with suboptimal response to levetiracetam. The primary
objective of the study is to evaluate the efficacy of JNJ-40411813
(ADX71149) in combination with levetiracetam using a time-to-event
endpoint.
“There is a strong preclinical rationale for
mGlu2 PAM in epilepsy, including a true synergistic effect with
levetiracetam,” said Robert Lütjens, Head of Discovery Biology of
Addex. "A highly synergistic anti-epileptic effect was observed
when the two drugs were administered together, and we look forward
to seeing this effect in epilepsy patients.”
Glutamate, mGlu2 Receptors and
EpilepsyGlutamate is the primary excitatory
neurotransmitter in the brain and plays a key role in the
initiation and spread of seizures. When activated, the mGlu2
receptor decreases the release of glutamate and consequently helps
to maintain neurotransmitter balance. In the presence of
agonist-induced activation, positive allosteric modulation of mGlu2
receptors could result in the normalization of the excessive
glutamate release seen during a seizure. There is still an urgent
need for more effective treatments for epilepsy, with improved
tolerability and safety. JNJ-40411813 (ADX71149) was described in
the Eilat 15 conference summary review as one of the most promising
novel approaches currently in development (Bialer et al., 2019.
Epilepsia). Proof of concept data with JNJ-40411813 (ADX71149) and
other mGlu2 PAMs in animal models of epilepsy have been published
in peer-reviewed journals (Metcalf et al., 2017 and 2018.
Epilepsia).
About Addex Therapeutics
Addex Therapeutics is a clinical-stage
pharmaceutical company focused on the development and
commercialization of an emerging class of novel orally available
small molecule drugs known as allosteric modulators for
neurological disorders. Allosteric modulators offer several
potential advantages over conventional non-allosteric molecules and
may offer an improved therapeutic approach to conventional
"orthosteric" small molecule or biological drugs. Addex's
allosteric modulator drug discovery platform targets receptors and
other proteins that are recognized as essential for therapeutic
intervention. Addex's lead drug candidate, dipraglurant (mGlu5
negative allosteric modulator or NAM), is poised to start a
pivotal registration clinical trial for Parkinson’s disease
levodopa induced dyskinesia (PD-LID) in H1 2021. Addex is also
investigating dipraglurant's therapeutic use in blepharospasm (a
type of dystonia), for which a clinical trial is expected to be
initiated in H1 2021. Addex's third clinical program, ADX71149
(mGlu2 positive allosteric modulator or PAM), developed in
collaboration with Janssen Pharmaceuticals, Inc, is scheduled to
enter a phase 2a proof of concept clinical study for the treatment
of epilepsy in Q2 2021. Addex’s GABAB PAM program has been
licensed to Indivior PLC for the treatment of addiction.
Preclinical programs include GABAB PAM for CMT1A, mGlu7 NAM
for PTSD, mGlu2 NAM for mild neurocognitive disorders, mGlu4 PAM
for Parkinson’s disease and mGlu3 PAM for neurodegenerative
disorders. Addex is listed on the SIX Swiss
Exchange and the NASDAQ Capital Market and trades under the ticker
symbol "ADXN".
Press Contacts:
|
Tim DyerChief Executive OfficerTelephone: +41 22 884 15
55Email: PR@addextherapeutics.com |
Mike SinclairPartner, Halsin Partners+44 (0)20 7318
2955msinclair@halsin.com |
Forward Looking
StatementsCertain statements made in this announcement are
forward-looking statements. These forward-looking statements are
not historical facts but rather are based on the Company's current
expectations, estimates, and projections about its industry; its
beliefs; and assumptions. Words such as 'anticipates,' 'expects,'
'intends,' 'plans,' 'believes,' 'seeks,' 'estimates,' and similar
expressions are intended to identify forward-looking statements.
These statements are not guarantees of future performance and are
subject to known and unknown risks, uncertainties, and other
factors, some of which are beyond the Company's control, are
difficult to predict, and could cause actual results to differ
materially from those expressed or forecasted in the
forward-looking statements. The Company cautions securityholders
and prospective securityholders not to place undue reliance on
these forward-looking statements, which reflect the view of the
Company only as of the date of this announcement. The
forward-looking statements made in this announcement relate only to
events as of the date on which the statements are made. The Company
will not undertake any obligation to release publicly any revisions
or updates to these forward-looking statements to reflect events,
circumstances, or unanticipated events occurring after the date of
this announcement except as required by law or by any appropriate
regulatory authority.
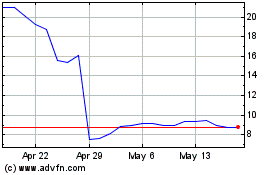
Addex Therapeutics (NASDAQ:ADXN)
Historical Stock Chart
From Apr 2024 to May 2024
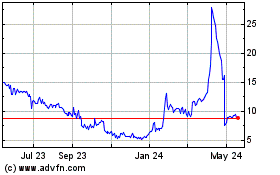
Addex Therapeutics (NASDAQ:ADXN)
Historical Stock Chart
From May 2023 to May 2024