SINGAPORE, May 24, 2020 /PRNewswire/ -- Singapore
medtech company HistoIndex's AI-based stain-free digital pathology
platform has long been established as a suitable tool for global
clinical guidelines that outline the assessment, prevention
and treatment of Chronic Hepatitis B (CHB) infections. In
the latest edition of China's
Guidelines for the Prevention and Treatment of Chronic Hepatitis B
[1], the digital pathology platform was recommended for use in
providing an automated and quantitative analysis of the liver's
morphological characteristics in unstained liver biopsies with high
repeatability and accuracy.
Challenges in Treating CHB Patients
Hepatitis B has been known as a complex disease to treat, due to
the challenge in distinguishing patients with an active infection.
These patients require intervention as opposed to patients with an
inactive infection who require only standard monitoring without
treatment. For patients with an active CHB infection, the treatment
decision is usually based on the increased level of alanine
aminotransferase (ALT) indicating an inflammation of the liver,
high HBV-DNA serum level and the presence of the Hepatitis B
e-antigen. However, these levels do not determine the presence and
level of fibrosis in the liver.
To prevent the progression of CHB to cirrhosis or Hepatocellular
Carcinoma (HCC), it is important to identify patients whose livers
are affected by advanced fibrosis prior to therapy, as the post
treatment prognosis depends on the stage of fibrosis. This can only
be achieved with a fibrosis assessment by performing a liver
biopsy, the current gold standard in evaluating the degree of
inflammation, necrosis, and fibrosis in all liver conditions.
Identifying Significant Morphological Features
Besides evaluating the severity of fibrosis and inflammation in
biopsies via conventional observations, it is important to
accurately assess dynamic changes in the extracellular matrix of
the liver, especially the various degrees of fibrous enlargement in
the liver. Having a very precise measurement of these features will
help clinicians to understand the stage of fibrosis in a patient
and carry out a suitable treatment management plan for their
patients. Enabled by Second Harmonic Generation (SHG), HistoIndex's
AI-based platform has been noted in multiple publications and
studies for its ability to accurately and automatically quantify
changes in liver fibrosis that are both observable and subtle, and
critical for treatment evaluation and modification.
In a study in 2018, fibrosis changes were evaluated with the SHG
platform CHB patients before and after 78 weeks' antiviral therapy,
with results encouraging the use of this tool to assess the
efficacy of new anti-fibrotic therapies in clinical trials [2]. An
interesting observation from this study was the reduction in
fibrous septa after treatment, which showed treatment efficacy and
was analyzed with precision by the SHG platform (Figure 1).
Therefore, this platform will be beneficial as an assistive tool
for CHB-related diagnosis, as well as for monitoring the efficacy
of antiviral therapies during CHB clinical trials. This is
synonymous with the SHG platform's recommendation by China's guidelines for its value in providing
an automated and quantitative analysis for the assessment of
CHB.
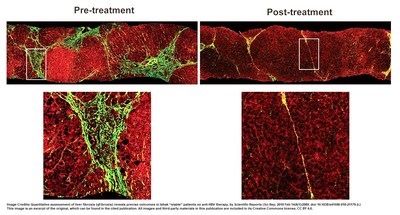
|
Image Credits:
Quantitative assessment of liver fibrosis (qFibrosis) reveals
precise outcomes in Ishak "stable" patients on anti-HBV therapy,
Scientific Reports [2]. This image is an excerpt of the original,
which can be found in the cited publication. All images and
third-party materials in this publication are included in its
Creative Commons license, CC BY 4.0.
|
Says Professor You Hong*, Chief Physician and Professor at the
Beijing Friendship Hospital (affiliated with the Capital Medical
University) and a co-author of the 2018 study, "As a clinician, we
need to make practical and reasonable decisions for our patients.
Including intelligent and reliable clinical tools such as the SHG
platform in the guidelines will greatly enhance our diagnostic
capability and personalize our treatment options in managing
patients with CHB. Having utilized this platform in our studies has
helped us to observe significant changes in the vital fibrosis
parameters that are crucial in determining the severity of CHB and
deciding the appropriate treatment route." Professor You is a Key
Opinion Leader (KOL) in the Hepatology field, and her additional
appointments include Deputy Director of the Chinese Society of
Hepatology and Steering Committee Member of APASL.
Dr Dean Tai, Chief Scientific
Officer of HistoIndex, adds, "We have substantial data from studies
conducted with the involvement of KOLs that show the potential of
our AI-based platform, in the area of CHB as well as Nonalcoholic
Steatohepatitis (NASH). I am confident that our platform will
contribute significantly if implemented in similar international
clinical guidelines, as well as in consortiums and workgroups that
have been set up globally for the management of CHB."
China: Bearing a Heavy
Disease Burden
As one of the countries battling CHB infections, China has the highest burden in the world,
with one third of the world's infected people living in
China [3]. Most of them are
unaware of their infection, making this disease a truly silent
epidemic. With limited access to diagnosis and treatment, these
people face a bleak future as their condition will eventually
progress – without treatment – to cirrhosis and eventually, liver
cancer. About 10 million people living with CHB will die by 2030
[4] [5], with most of these deaths being avoidable. As part of
efforts to help the nation in this devastating disease, experts
under the Chinese Medical Association's (CMA) Infectious Diseases
and Liver Diseases Branch released the updated guidelines in
2019.
EDITOR'S NOTES
* Professor You Hong is also the Assistant Dean at the
Capital Medical University, where she holds additional appointments
as Director of the Office of Academic Research and Director of the
Experimental Center.
A Major and Growing Global Health Concern
According to the World Health Organization (WHO), CHB has
resulted in an estimated 887,000 deaths in 2015, mostly from
cirrhosis and hepatocellular carcinoma (i.e. primary liver cancer),
and 257 million people are living with CHB as of the same year. As
of 2016, 27 million people were aware of their infection, while
only 4.5 million of the people diagnosed were on treatment [6].
Following WHO's call to eliminate viral hepatitis as a public
health problem, and bring down new infections by 90% and reduce
deaths by 65% by 2030, countries across the world have implemented
clinical guidelines to standardize and aid in the prevention,
diagnosis and antiviral therapy of CHB:
(a) According to the
American Association for the Study of Liver Diseases (AASLD)
practice guidance, a liver biopsy should be considered in patients
with persistently borderline normal or slightly elevated ALT
levels, particularly those who have been infected with HBV for a
long period of time and are now older than 40 years of age
[7].
(b) Similarly, in the
European Association for the Study of the Liver (EASL) 2017
Clinical Practice Guidelines on the management of Hepatitis B
Virus Infections, patients without cirrhosis should be considered
for treatment when their severity of liver disease assessed
traditionally by liver biopsy shows at least moderate
necroinflammation and/or at least moderate fibrosis [8].
(c) In the 2016 update of
the Asian-Pacific Association for the Study of the Liver (APASL)
Consensus Guidelines on Invasive and Non-invasive Assessment of
Hepatic Fibrosis, the liver biopsy remains the gold standard for
assessing liver fibrosis. Interestingly, the guidelines
acknowledged a possible shift from routine light microscopy to
digital image analysis, and the incorporation of numerical
measurements in conjunction with the integrated analysis of other
cellular information [9]. Since then, microscopy and analysis of
liver fibrosis has indeed been transformed; pathologists today are
assisted by smart digital pathology tools to make accurate and
objective assessments of fibrosis and other relevant features in
the liver.
|
References
|
|
1. Chinese
Medical Association Infectious Diseases Branch, Chinese Medical
Association Hepatology Branch. Guidelines for the Prevention
and Treatment of Chronic Hepatitis B (2019 Edition) [J].
Chinese Journal of Clinical Infectious Diseases, 2019, 12 (6):
401-428.: 10.3760 / cma.j.issn.1674-2397.2019.06.001.
|
|
Note: The Chinese
Journal of Clinical Infectious Diseases website is in Mandarin and
is enabled with an automatic prompt that offers the option for an
English translation.
|
|
2. Sun, Y.,
Zhou, J., Wu, X. et al. Quantitative assessment of liver
fibrosis (qFibrosis) reveals precise outcomes in Ishak "stable"
patients on anti-HBV therapy. Sci Rep 8, 2989 (2018).
https://doi.org/10.1038/s41598-018-21179-2
|
|
3. Shanquan
Chen, Jun Li, Dan Wang, Hong Fung, Lai-yi Wong, Lu Zhao. The
Hepatitis B epidemic in China should receive more
attention. The Lancet Volume 391, Issue 10130, P1572, April
21, 2018. https://doi.org/10.1016/S0140-6736(18)30499-9.
|
|
4. World Health
Organization. Global Hepatitis Report 2017. Accessed
29 April 2020.
|
|
5. World Health
Organization. Up to 10 million people in China could die from
chronic hepatitis by 2030 – Urgent action needed to bring an end to
the 'silent epidemic'. Accessed 29 April 2020.
|
|
6. World Health
Organization, Hepatitis B Fact Sheet. Accessed 29
April 2020.
|
|
7. Terrault,
N.A., Lok, A.S., McMahon, B.J., Chang, K.‐M., Hwang, J.P., Jonas,
M.M., Brown, R.S., Jr., Bzowej, N.H. and Wong, J.B. (2018),
Update on Prevention, Diagnosis, and Treatment of Chronic
Hepatitis B: AASLD 2018 Hepatitis B
Guidance. Hepatology, 67: 1560-1599.
doi:10.1002/hep.29800.
|
|
8. European
Association for the Study of the Liver, EASL 2017 Clinical
Practice Guidelines on the management of hepatitis B virus
infection. J Hepatol. Volume 67, Issue 2, P370-398, August
01, 2017. https://doi.org/10.1016/j.jhep.2017.03.021
|
|
9. Sarin, S.K.,
Kumar, M., Lau, G.K. et al. Asian-Pacific Clinical Practice
Guidelines on the Management of Hepatitis B: A 2015 Update.
Hepatol Int 10, 1–98 (2016).
https://doi.org/10.1007/s12072-015-9675-4
|
Media Contacts: Ms Cynthia Anne
Victor, Corporate Communications & PR, HistoIndex,
cynthia.victor@histoindex.com
Photo -
https://mma.prnewswire.com/media/1171910/Fibrous_Septa_Quantitative_assessment_of_liver_fibrosis_HistoIndex_Credits1.jpg