Mereo BioPharma Appoints Christine Fox as Chief Financial Officer and Heidi Petersen as Senior Vice President, Regulatory Aff...
October 20 2020 - 7:00AM

Mereo BioPharma Group plc (NASDAQ: MREO, AIM: MPH) (“Mereo” or “the
Company”), a clinical stage biopharmaceutical company focused on
oncology and rare diseases, today announced the appointment of
Christine Fox, CPA, as Chief Financial Officer (CFO) and Heidi
Petersen as Senior Vice President, Regulatory Affairs.
Ms. Fox brings an extensive array of financial
experience to Mereo, including a background in financial operations
and reporting, technical accounting and external audit. She
replaces Interim CFO and current Mereo Board member Mr. Michael
Wyzga, who will continue to serve on Mereo’s Board. Ms. Petersen
brings more than 25 years of experience in the biopharmaceutical
industry, with regulatory expertise across a range of therapeutic
areas including immuno-oncology. Ms. Petersen’s appointment will be
effective November 2, 2020 and Ms. Fox in January 2021.
Denise Scots-Knight, Chief Executive Officer of
Mereo, said: “Christine and Heidi are seasoned financial and
regulatory executives and we are excited to welcome them to Mereo.
The remainder of 2020 and 2021 will be an important period for
Mereo as we expect multiple milestones across our pipeline,
including the initiation of our Phase 1b/2 combination study with
our anti-TIGIT antibody, etigilimab, in the coming weeks. With the
addition of Christine and Heidi, we believe we have an exceptional
senior management team in place as we continue to execute on our
clinical development and business objectives in preparation for our
next stage of growth.”
Christine Fox, CPA, as Chief Financial
OfficerMs. Fox is a Certified Public Accountant (CPA) with
over 18 years of experience in financial operations and reporting,
technical accounting and external audit. She has a proven track
record of delivering high-quality financial information under
international accounting standards and experience in the execution
of corporate transactions. Prior to joining Mereo, Ms. Fox served
as Group Financial Controller and Treasurer of Travelport where she
managed a global financial operations team. Prior to joining
Travelport, Ms. Fox served more than 10 years at KPMG in the U.S.
and Switzerland, in positions of increasing responsibility, where
she was primarily focused on large multi-national clients reporting
under U.S. GAAP and IFRS across a wide variety of industries,
including the pharmaceutical industry. Ms. Fox received a B.S. in
accounting from Butler University.
Heidi Petersen as Senior Vice President,
Regulatory AffairsMs. Petersen is a seasoned regulatory
affairs professional with 25 years of experience. Prior to joining
Mereo, she served as Vice President, Regulatory Affairs at Kartos
Therapeutics. Prior to Kartos, Ms. Petersen served as Vice
President, Regulatory Affairs at Immune Design (acquired by Merck
in 2019) where she provided global regulatory strategy on the
development of the company’s immuno-oncology programs across all
stages of clinical development. Previously, Ms. Petersen served as
Vice President, Regulatory Affairs and Quality at
BN-Immunotherapeutics (fully acquired by Bavarian Nordic in 2009)
where she led global regulatory strategy for the company’s early
and late-stage immuno-oncology programs. Earlier in her career, Ms.
Petersen served as an independent regulatory consultant, and held
business development and regulatory affairs positions with
Pharmacyclics (subsequently acquired by AbbVie). She also held
regulatory positions with Chiron Corporation (subsequently acquired
by Novartis) and ALZA corporation (subsequently acquired by Johnson
& Johnson). She received a master’s in public health from
Columbia University and a B.S. in biology from Tulane
University.
About Mereo BioPharmaMereo
BioPharma is a biopharmaceutical company focused on the
development and commercialization of innovative therapeutics that
aim to improve outcomes for oncology and rare diseases. Mereo's
lead oncology product candidate, etigilimab (Anti-TIGIT), has
completed a Phase 1a dose escalation clinical trial in patients
with advanced solid tumors and has been evaluated in a Phase 1b
study in combination with nivolumab in select tumor types. Mereo's
rare disease product portfolio consists of setrusumab, which has
completed a Phase 2b dose-ranging study in adults with osteogenesis
imperfecta (OI), as well as alvelestat, which is being investigated
in a Phase 2 proof-of-concept clinical trial in patients with
alpha-1 antitrypsin deficiency (AATD) and in a Phase 1b/2 clinical
trial in COVID-19 respiratory disease.
Additional Information The
person responsible for arranging the release of this information on
behalf of the Company is Charles Sermon, General Counsel.
Forward-Looking StatementsThis
Announcement contains "forward-looking statements." All statements
other than statements of historical fact contained in this
Announcement are forward-looking statements within the meaning of
Section 27A of the United States Securities Act of 1933, as amended
and Section 21E of the United States Securities Exchange Act of
1934, as amended. Forward-looking statements usually relate to
future events and anticipated revenues, earnings, cash flows or
other aspects of our operations or operating results.
Forward-looking statements are often identified by the words
"believe," "expect," "anticipate," "plan," "intend," "foresee,"
"should," "would," "could," "may," "estimate," "outlook" and
similar expressions, including the negative thereof. The absence of
these words, however, does not mean that the statements are not
forward-looking. These forward-looking statements are based on the
Company's current expectations, beliefs and assumptions concerning
future developments and business conditions and their potential
effect on the Company. While management believes that these
forward-looking statements are reasonable as and when made, there
can be no assurance that future developments affecting the Company
will be those that it anticipates.
All of the Company's forward-looking statements
involve known and unknown risks and uncertainties some of which are
significant or beyond its control and involve assumptions that
could cause actual results to differ materially from the Company's
historical experience and its present expectations or projections.
These forward-looking statements are subject to risks and
uncertainties, including, among other things, those described in
the Company’s latest Annual Report on Form 20-F, Reports on Form
6-K and other documents filed from time to time by the Company with
the United States Securities and Exchange Commission. The Company
wishes to caution investors not to place undue reliance on any
forward-looking statements, which speak only as of the date hereof.
The Company undertakes no obligation to publicly update or revise
any forward-looking statements, whether as a result of new
information, future events or otherwise, except to the extent
required by law.
Mereo BioPharma Contacts:
|
Mereo |
+44 (0)333 023 7300 |
|
Denise Scots-Knight, Chief Executive Officer |
|
|
|
|
|
N+1 Singer (Nominated Adviser and Broker
to Mereo) |
+44 (0)20 7496 3081 |
|
Phil Davies |
|
|
Will Goode |
|
| |
|
|
Burns McClellan (US Investor Relations Adviser
to Mereo) |
+01 212 213 0006 |
|
Lisa Burns |
|
|
Steve Klass |
|
|
|
|
|
FTI Consulting (UK Public Relations Adviser
to Mereo) |
+44 (0)20 3727 1000 |
|
Simon Conway |
|
|
Ciara Martin |
|
|
|
|
|
Investors |
investors@mereobiopharma.com |
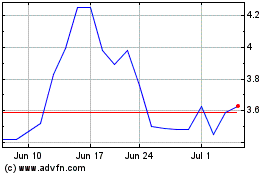
Mereo BioPharma (NASDAQ:MREO)
Historical Stock Chart
From Mar 2024 to Apr 2024
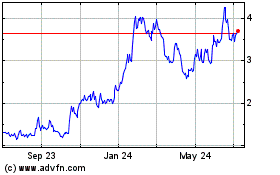
Mereo BioPharma (NASDAQ:MREO)
Historical Stock Chart
From Apr 2023 to Apr 2024