Bayer Files for U.S., EU Approval of Aliqopa/Rituximab Combo
June 21 2021 - 11:35AM
Dow Jones News
By Colin Kellaher
Bayer AG on Monday said it has filed for U.S. and European
expanded approval of its cancer drug Aliqopa in combination with
rituximab, a drug used to treat certain types of cancer and
rheumatoid arthritis.
The Leverkusen, Germany, chemical and pharmaceutical company
said it is seeking U.S. Food and Drug Administration approval of
the combination in relapsed indolent B-cell non-Hodgkin's
lymphoma.
The company said it also has filed for European Medicines Agency
approval for the treatment of relapsed marginal zone lymphoma, a
subtype of indolent B-cell non-Hodgkin's lymphoma.
Bayer in April said a Phase III study showed a significant
progression-free survival benefit for the combination in patients
with relapsed indolent non-Hodgkin's lymphoma.
The FDA in 2017 approved Aliqopa for adults with relapsed
follicular lymphoma who have received at least two prior systemic
therapies.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
June 21, 2021 11:26 ET (15:26 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
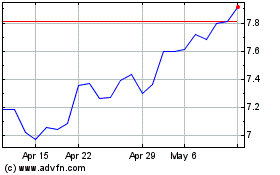
Bayer Aktiengesellschaft (PK) (USOTC:BAYRY)
Historical Stock Chart
From Apr 2024 to May 2024
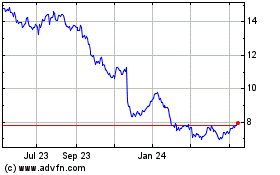
Bayer Aktiengesellschaft (PK) (USOTC:BAYRY)
Historical Stock Chart
From May 2023 to May 2024