Filed Pursuant to Rule 424(b)(5)
Registration No. 333-233350
PROSPECTUS SUPPLEMENT
To Prospectus dated September 30, 2019

Up to $50,605,000
Common Stock
On July 27, 2018,
we entered into an At Market Issuance Sales Agreement, which, as amended as described below, we refer to as the sales agreement,
with B. Riley, Inc., Cantor Fitzgerald & Co., National Securities Corporation and Oppenheimer & Co Inc., each an Agent
and collectively, the Agents, relating to the sale of shares of our common stock offered by this prospectus supplement. In accordance
with the terms of the sales agreement, under this prospectus supplement we may offer and sell shares of our common stock, $0.0001
par value per share, having an aggregate offering price of up to $50.6 million from time to time through the Agents, acting as
agents. On August 16, 2019, we filed a new registration statement on Form S-3 (No. 333-233350), which became effective on September
30, 2019. On July 20, 2020, we entered into Amendment No. 1 to the Sales Agreement with the Agents to reflect the new registration
statement. As of the date of this prospectus supplement, we have sold 5,641,856 common shares under the sales agreement for approximately
$30.5 million in gross proceeds. As such, we may offer and sell the remaining balance of common shares having a maximum aggregate
sales price of up to $50.6 million pursuant to this prospectus supplement.
Our
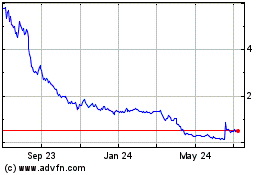
common stock is traded on The Nasdaq Capital Market, or the Exchange, under the symbol “MBIO.” The last reported sale
price of our common stock on July 17, 2020 was $3.48 per share.
Sales
of our common stock, if any, under this prospectus supplement will be made by any method permitted that is deemed an “at
the market offering” as defined in Rule 415(a)(4) under the Securities Act of 1933, as amended, or the Securities Act. The
Agents are not required to sell any specific amount, but will act as our sales agents using commercially reasonable efforts consistent
with its normal trading and sales practices. There is no arrangement for funds to be received in any escrow, trust or similar
arrangement.
The
Agents will be entitled to compensation at a commission rate of up to 3% of the gross sales price per share sold. In connection
with the sale of the common stock on our behalf, each Agent will be deemed to be an “underwriter” within the meaning
of the Securities Act and the compensation of each Agent will be deemed to be underwriting commissions or discounts. We have also
agreed to provide indemnification and contribution to the Agents with respect to certain liabilities, including liabilities under
the Securities Act.
Investing
in these securities involves a high degree of risk. Before buying shares of our common stock, you should carefully consider the
risk factors described in “Risk Factors” beginning on page S-3 of this prospectus supplement and in the documents
incorporated by reference into this prospectus supplement and any free writing prospectus that we have authorized for use in connection
with this offering.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities, or
determined if this prospectus supplement is accurate or complete. Any representation to the contrary is a criminal offense.
|
|
B. Riley FBR
|
|
|
Cantor
|
|
|
National Securities
Corporation
|
|
|
Oppenheimer
& Co.
|
The date of this
Prospectus Supplement is July 20, 2020
TABLE OF CONTENTS
Prospectus Supplement
Prospectus
ABOUT THIS PROSPECTUS
SUPPLEMENT
This
document is in two parts. The first part is this prospectus supplement, which describes the specific terms of this offering of
common stock and also adds to and updates information contained in the accompanying prospectus and the documents incorporated
by reference in this prospectus supplement and the accompanying prospectus. The second part, the accompanying prospectus, dated
September 30, 2019, provides more general information, some of which may not apply to this offering. Generally, when we refer
to this prospectus supplement, we are referring to both parts of this document combined. To the extent there is a conflict between
the information contained in this prospectus supplement and the information contained in the accompanying prospectus or any document
incorporated by reference that was filed with the U.S. Securities and Exchange Commission, or SEC, before the date of this prospectus
supplement, you should rely on the information in this prospectus supplement; provided that if any statement in one of these documents
is inconsistent with a statement in another document having a later date—for example, a document incorporated by reference
in the accompanying prospectus—the statement in the document having the later date modifies or supersedes the earlier statement.
We
further note that the representations, warranties and covenants made by us in any agreement that is filed as an exhibit to any
document that is incorporated by reference herein were made solely for the benefit of the parties to such agreement, including,
in some cases, for the purpose of allocating risk among the parties to such agreements, and should not be deemed to be a representation,
warranty or covenant to you. Moreover, such representations, warranties or covenants were accurate only as of the date when made.
Accordingly, such representations, warranties and covenants should not be relied on as accurately representing the current state
of our affairs.
We
are offering to sell, and seeking offers to buy, shares of our common stock only in jurisdictions where offers and sales are permitted.
The distribution of this prospectus supplement and the accompanying prospectus and the offering of the common stock in certain
jurisdictions may be restricted by law. Persons outside the United States who come into possession of this prospectus supplement
and the accompanying prospectus must inform themselves about, and observe any restrictions relating to, the offering of the common
stock and the distribution of this prospectus supplement and the accompanying prospectus outside the United States. This prospectus
supplement and the accompanying prospectus do not constitute, and may not be used in connection with, an offer to sell, or a solicitation
of an offer to buy, any securities offered by this prospectus supplement and the accompanying prospectus by any person in any
jurisdiction in which it is unlawful for such person to make such an offer or solicitation.
Unless
otherwise indicated in this prospectus supplement or the context otherwise requires, all references to “we,” “us,”
“our,” “the Company,” and “Mustang” refer Mustang Bio, Inc. and its subsidiaries.
FORWARD-LOOKING
STATEMENTS
Certain
matters discussed in this prospectus supplement may constitute forward-looking statements for purposes of the Securities Act of
1933, as amended, or the Securities Act, and the Securities Exchange Act of 1934, as amended, or the Exchange Act, and involve
known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be
materially different from the future results, performance or achievements expressed or implied by such forward-looking statements.
The words “anticipate,” “believe,” “estimate,” “may,” “expect,” “will,”
“could,” “project,” “intend” and similar expressions are generally intended to identify forward-looking
statements. Our actual results may differ materially from the results anticipated in these forward-looking statements due to a
variety of factors, including, without limitation, those discussed under the caption “Risk Factors” contained in this
prospectus supplement, the accompanying prospectus, any applicable free writing prospectus and under similar heading in the other
documents that are incorporated by reference into this prospectus. All written or oral forward-looking statements attributable
to us are expressly qualified in their entirety by these cautionary statements. Such forward-looking statements include, but are
not limited to, statements about our:
|
|
•
|
expectations
for increases or decreases in expenses;
|
|
|
•
|
expectations
for the clinical and pre-clinical development, manufacturing, regulatory approval, and
commercialization of our product candidates or any other products we may acquire or in-license;
|
|
|
•
|
our
use of clinical research centers and other contractors;
|
|
|
•
|
expectations
for incurring capital expenditures to expand our research and development and manufacturing
capabilities;
|
|
|
•
|
expectations
for generating revenue or becoming profitable on a sustained basis;
|
|
|
•
|
expectations
or ability to enter into marketing and other partnership agreements;
|
|
|
•
|
expectations
or ability to enter into product acquisition and in-licensing transactions;
|
|
|
•
|
expectations
or ability to build our own commercial infrastructure to manufacture, market and sell
our product candidates;
|
|
|
•
|
acceptance
of our products by doctors, patients or payors;
|
|
|
•
|
our
ability to compete against other companies and research institutions;
|
|
|
•
|
our
ability to secure adequate protection for our intellectual property;
|
|
|
•
|
our
ability to attract and retain key personnel;
|
|
|
•
|
availability
of reimbursement for our products;
|
|
|
•
|
estimates
of the sufficiency of our existing cash and cash equivalents and investments to finance
our operating requirements, including expectations regarding the value and liquidity
of our investments;
|
|
|
•
|
the
volatility of our stock price;
|
|
|
•
|
expectations
for future capital requirements.
|
The
forward-looking statements contained in this prospectus supplement reflect our views and assumptions only as of the date of this
prospectus supplement, respectively. Except as required by law, we assume no responsibility for updating any forward-looking statements.
We qualify all of our forward-looking statements by these cautionary statements. New risks and uncertainties arise from time to
time, and it is impossible for us to predict these events or how they may affect us.
PROSPECTUS SUPPLEMENT SUMMARY
This summary highlights information
contained elsewhere or incorporated by reference in this prospectus supplement, the accompanying prospectus and in the documents
we incorporate by reference. This summary does not contain all of the information that you should consider before deciding to
invest in our common stock. You should read this entire prospectus supplement carefully, including the “Risk Factors”
section contained in this prospectus supplement, the accompanying prospectus and in the documents we incorporate by reference.
Our Company
We are a clinical-stage
biopharmaceutical company focused on translating today’s medical breakthroughs in cell and gene therapies into potential
cures for hematologic cancers, solid tumors and rare genetic diseases. We aim to acquire rights to these technologies by licensing
or otherwise acquiring an ownership interest in the technologies, funding their research and development and eventually either
out-licensing or bringing the technologies to market.
Our pipeline is currently
focused in three core areas: gene therapy programs for rare genetic disorders, chimeric antigen receptor (“CAR”) engineered
T cell (“CAR T”) therapies for hematologic malignancies and CAR T therapies for solid tumors. For each therapy we
have partnered with world class research institutions. For our gene therapy program, we have partnered with St. Jude Children’s
Research Hospital (“St. Jude”) in the development of a first-in-class ex vivo lentiviral treatment of X-linked severe
combined immunodeficiency (“XSCID”) and for our CAR T therapies we have partnered with the City of Hope National Medical
Center (“COH”), Fred Hutchinson Cancer Research Center (“Fred Hutch”) and Nationwide Children’s
Hospital (“Nationwide”).
Gene Therapy
In partnership with
St. Jude, our gene therapy program is being conducted under an exclusive license to develop a potentially curative treatment for
XSCID, a rare genetic immune system condition in which affected patients do not live beyond infancy without treatment. This first-in-class
ex vivo lentiviral gene therapy is currently in two Phase 1/2 clinical trials: a multicenter trial in newly diagnosed infants
sponsored by St. Jude and a single-center trial in previously transplanted patients sponsored by the National Institutes of Health
(“NIH”).
In May 2020, Mustang
submitted an IND application with the U.S. Food and Drug Administration (“FDA”) to initiate a multicenter Phase 2
clinical trial of MB-107 in newly diagnosed infants with XSCID who are under the age of two. The trial is expected to enroll 10
patients who, together with 15 patients enrolled in the current multicenter trial led by St. Jude Children’s Research Hospital,
will be compared with 25 matched historical control patients who have undergone hematopoietic stem cell transplant (“HSCT”).
The primary efficacy endpoint will be event-free survival. The initiation of this trial is currently on hold pending CMC clearance
by the FDA. Mustang is targeting topline data from the trial in the second half of 2022.
Mustang further expects
to file an IND in the third quarter of 2020 for a registrational multi-center Phase 2 clinical trial of its lentiviral gene therapy
in previously transplanted XSCID patients. This product will be designated MB-207. Mustang anticipates enrolling 20 patients and
comparing them to matched historical control patients who have undergone a second HSCT. Mustang is targeting topline data for
this trial in the second half of 2022.
CAR T Therapies
Our pipeline of CAR
T therapies is being developed under exclusive licenses from several world class research institutions. Our strategy is to license
these technologies, support preclinical and clinical research activities by our partners and transfer the underlying technology
to our cell processing facility located in Worcester, Massachusetts, to conduct our own clinical trials.
We are developing
CAR T therapies for hematologic malignancies in partnership with COH targeting CD123 (MB-102) and CS1 (MB-104) and with Fred Hutch
targeting CD20 (MB-106). Phase 1 clinical trials sponsored by COH for MB-102 and MB-104 and by Fred Hutch for MB-106 are underway.
In the third quarter of 2019 the FDA approved our IND application to initiate a multicenter Phase 1/2 clinical trial of MB-102,
and our clinical trial began enrollment in the second quarter of 2020 for the treatment of patients with acute myeloid leukemia,
blastic plasmacytoid dendritic cell neoplasm, and high-risk myelodysplastic syndrome. We expect to file an IND for MB-106 in the
first half of 2021 and to initiate our own Phase 1 clinical trial shortly thereafter for the treatment of patients with non-Hodgkin
lymphoma and chronic lymphocytic leukemia.
We also plan to file
an IND and initiate our own clinical trial for MB-104 for the treatment of patients with multiple myeloma.
We are also developing
CAR T therapies for solid tumors in partnership with COH targeting IL13Rα2 (MB-101), HER2 (MB-103) and PSCA (MB-105). In
addition, we have partnered with Nationwide for the C134 oncolytic virus (MB-108) in order to enhance the activity of MB-101 for
the treatment of patients with GBM. Phase 1 clinical trials sponsored by COH for MB-101, MB-103 and MB-105 are underway. A Phase
1 clinical trial sponsored by the University of Alabama at Birmingham (“UAB”) for MB-108 began during the third quarter
of 2019 and, in the fourth quarter of 2020, COH plans to file an IND for the combination of MB-101 and MB-108 for the treatment
of patients with GBM. Mustang also plans to file INDs and initiate our own clinical trials for MB-103 for the treatment of patients
with metastatic breast cancer to brain and for MB-105 for the treatment of patients with prostate and pancreatic cancer.
Company Information
We are a majority-controlled
subsidiary of Fortress Biotech, Inc. (“Fortress”).
Our principal executive
offices are located at 377 Plantation Street, Worcester, Massachusetts 01605, and our telephone number is 781-652-4500. We maintain
a website on the Internet at www.mustangbio.com and our e-mail address is info@mustangbio.com. Our Internet website, and the information
contained on it, are not to be considered part of this prospectus supplement.
The Offering
|
Common stock offered by us pursuant to
this prospectus supplement
|
|
Shares of our common stock having an aggregate offering
price of up to $50.6 million.
|
|
|
|
|
|
Plan of Distribution
|
|
“At the market offering” that may be made from time
to time on The Nasdaq Capital Market or other market for our common stock in the United States through the Agents. See the
section titled “Plan of Distribution” on page S-7 of this prospectus supplement.
|
|
|
|
|
|
Use of Proceeds
|
|
We intend to use the net proceeds of this offering for the continued
development of novel, non-chemotherapy, immune-enhanced treatments for patients with hematologic malignancies and solid tumor
cancers; the acquisition, development and commercialization of other pharmaceutical products; and for general corporate purposes.
See the section titled “Use of Proceeds” on page S-5 of this prospectus supplement.
|
|
|
|
|
|
Risk factors
|
|
See “Risk Factors” beginning on page S-3 of this
prospectus supplement and the other information included in, or incorporated by reference into, this prospectus supplement
for a discussion of certain factors you should carefully consider before deciding to invest in shares of our common stock.
|
|
|
|
|
|
Nasdaq Capital Market Symbol
|
|
MBIO
|
RISK FACTORS
Investment in our common stock involves
risks. Before deciding whether to invest in our common stock, you should consider carefully the risk factors discussed below and
those contained in the section entitled “Risk Factors” contained in our Annual Report on Form 10-K for the year ended December 31, 2019, as filed with the SEC on March 16, 2020, which is incorporated herein by reference in its entirety,
together with other information in the accompanying prospectuses, the information and documents incorporated by reference herein
and therein, and in any free writing prospectus that we have authorized for use in connection with this offering. If any of the
risks or uncertainties described in our SEC filings actually occurs, our business, financial condition, results of operations
or cash flow could be materially and adversely affected. This could cause the trading price of our common stock to decline, resulting
in a loss of all or part of your investment. The risks and uncertainties we have described are not the only ones facing our company.
Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also affect our business
operations.
Risks Associated with this Offering
We have broad discretion in the use of the net proceeds
of this offering and may not use them effectively.
We intend to use
the net proceeds from this offering for general corporate purposes and to continue preclinical development and clinical trials
of our product candidates. However, our management will have broad discretion in the application of the net proceeds from this
offering and could spend the proceeds in ways that do not improve our results of operations or enhance the value of our common
stock. The failure by management to apply these funds effectively could result in financial losses that could have a material
adverse effect on our business, cause the price of our common stock to decline and delay the development of our product candidates.
You will experience immediate and substantial dilution.
The offering price
per share in this offering may exceed the net tangible book value per share of our common stock outstanding prior to this offering.
Assuming that shares of our common stock are sold at a price of $3.48 per share, the last reported sale price of our common stock
on the Nasdaq Capital Market on July 17, 2020, for aggregate gross proceeds of $50.6 million, and after deducting commissions
and estimated offering expenses payable by us, you will experience immediate dilution of $1.80 per share, representing the difference
between our as adjusted net tangible book value per share as of March 31, 2020 after giving effect to this offering and the assumed
offering price, net of commissions and offering expenses. The exercise of outstanding stock options and warrants will result in
further dilution of your investment. See the section entitled “Dilution” below for a more detailed illustration of
the dilution you would incur if you participate in this offering.
You may experience future dilution as a result of future
equity offerings.
In order to raise
additional capital, we may in the future offer additional shares of our common stock or other securities convertible into or exchangeable
for our common stock at prices that may not be the same as the price per share in this offering. We may sell shares or other securities
in any other offering at a price per share that is less than the price per share paid by investors in this offering, and investors
purchasing shares or other securities in the future could have rights superior to existing stockholders. The price per share at
which we sell additional shares of our common stock, or securities convertible or exchangeable into common stock, in future transactions
may be higher or lower than the price per share paid by investors in this offering.
Fortress will continue to control a voting majority of our
common stock following the offering.
Pursuant to the terms
of the Class A Preferred Stock held by Fortress, Fortress is entitled to cast, for each share of Class A Preferred Stock held
by Fortress, the number of votes that is equal to one and one-tenth (1.1) times a fraction, the numerator of which is the sum
of the shares of outstanding common stock and the denominator of which is the number of shares of outstanding Class A Preferred
Stock. Accordingly, as long as Fortress owns any shares of Class A Preferred Stock, it will be able to control or significantly
influence all matters requiring approval by our stockholders, including the election of directors and the approval of mergers
or other business combination transactions. The consummation of this offering will not impact Fortress’s holdings of Class
A Preferred Stock, so Fortress will continue to be able to exercise such control and influence over the Company. The interests
of Fortress may not always coincide with the interests of other stockholders, and Fortress may take actions that advance its own
interests and are contrary to the desires of our other stockholders. Moreover, this concentration of voting power may delay, prevent
or deter a change in control of us even when such a change may be in the best interests of all stockholders, could deprive our
stockholders of an opportunity to receive a premium for their common stock as part of a sale of Mustang or our assets, and might
affect the prevailing market price of our common stock.
Our stock price can be volatile, which increases the risk
of litigation, and may result in a significant decline in the value of your investment.
The trading price
of our common stock has been and is likely to continue to be highly volatile and subject to wide fluctuations in price in response
to various factors, many of which are beyond our control. These factors include:
|
|
·
|
announcements relating
to the clinical development of our product candidates;
|
|
|
·
|
announcements concerning
the progress of our efforts to obtain regulatory approval for and commercialize our product candidates or any future product candidate,
including any requests we receive from the FDA, or comparable regulatory authorities outside the United States, for additional
studies or data that result in delays in obtaining regulatory approval or launching these product candidates, if approved;
|
|
|
·
|
the depth and liquidity
of the market for our common stock;
|
|
|
·
|
investor perceptions
about us and our business;
|
|
|
·
|
market conditions in
the pharmaceutical and biotechnology sectors or the economy as a whole;
|
|
|
·
|
price and volume fluctuations
in the overall stock market;
|
|
|
·
|
the failure of one or
more of our product candidates or any future product candidate, if approved, to achieve commercial success;
|
|
|
·
|
developments concerning
product development results or intellectual property rights of others;
|
|
|
·
|
litigation or public
concern about the safety of our potential products;
|
|
|
·
|
announcements of the
introduction of new products by us or our competitors;
|
|
|
·
|
actual fluctuations in
our quarterly operating results, and concerns by investors that such fluctuations may occur in the future;
|
|
|
·
|
deviations in our operating
results from the estimates of securities analysts or other analyst comments;
|
|
|
·
|
developments concerning
current or future strategic collaborations;
|
|
|
·
|
discussion of us or our
stock price by the financial and scientific press and in online investor communities;
|
|
|
·
|
health care reform legislation,
including measures directed at controlling the pricing of pharmaceutical products, and third-party coverage and reimbursement
policies; and
|
|
|
·
|
additions or departures
of key personnel.
|
In addition, equity
markets in general, and the market for biotechnology and life sciences companies in particular, have experienced extreme price
and volume fluctuations that have often been unrelated or disproportionate to the operating performance of companies traded in
those markets. These broad market and industry factors may materially affect the market price of our common stock, regardless
of our development and operating performance. In the past, following periods of volatility in the market price of a company’s
securities, securities class-action litigation has often been instituted against that company. Such litigation, if instituted
against us, could cause us to incur substantial costs to defend such claims and divert management’s attention and resources,
which could seriously harm our business.
USE OF PROCEEDS
We may issue and sell
shares of our common stock having aggregate sales proceeds of up to $50.6 million from time to time. Because there is no minimum
offering amount required as a condition to close this offering, the actual total public offering amount, commissions, expenses,
and proceeds to us, if any, are not determinable at this time but will be reported in our periodic reports.
We intend to use the
net proceeds of this offering for the continued development of novel, non-chemotherapy, immune-enhanced treatments for patients
with hematologic malignancies and solid tumor cancers; the acquisition, development and commercialization of other pharmaceutical
products; and for general corporate purposes.
The timing and amounts
of our actual expenditures will depend on several factors, including the progress of our research and development programs, the
results of other pre-clinical and clinical studies and the timing and costs of regulatory approvals. Pending the uses described
above, we will invest the net proceeds in short-term and long-term, investment grade, interest-bearing securities.
DIVIDEND POLICY
We have never declared
or paid any cash dividends on our common stock and do not anticipate paying any cash dividends in the foreseeable future. Any
future determination to pay dividends will be at the discretion of our board of directors.
DILUTION
If you invest in
our common stock, your interest will be diluted to the extent of the difference between the price per share you pay in this offering
and the net tangible book value per share of our common stock immediately after this offering. Our net tangible book value of
our common stock as of March 31, 2020 was approximately $45.8 million, or approximately $1.09 per share of common stock based
upon 42,076,840 shares outstanding. Net tangible book value per share is equal to our total tangible assets, less our total liabilities,
divided by the total number of shares outstanding as of March 31, 2020.
After giving effect
to the sale of our common stock in the aggregate amount of $50.6 million at an assumed offering price of $3.48 per share, the
last reported sale price of our common stock on the Nasdaq Capital Market on July 17, 2020, and after deducting estimated offering
commissions and expenses payable by us, our net tangible book value as of March 31, 2020 would have been approximately $94.8 million,
or $1.68 per share of common stock. This represents an immediate increase in net tangible book value of $0.59 per share to our
existing stockholders and an immediate dilution in net tangible book value of $1.80 per share to new investors in this offering.
The following table
illustrates this calculation on a per share basis1:
|
Offering price per share
|
|
|
|
$3.48
|
|
Net tangible book value per share
|
|
$1.09
|
|
|
|
Increase in net tangible book value per share attributable to the offering
|
|
$0.59
|
|
|
|
As-adjusted net tangible book value per share after giving effect to the offering
|
|
$1.68
|
|
|
|
Dilution in net tangible book value per share to new investors
|
|
|
|
$1.80
|
1
This table excludes the effect of our offering of common stock on June 12, 2020 and sales under our ATM program between
April 1, 2020 and the date hereof.
The number of shares
of our common stock to be outstanding immediately after this offering is based on 42,076,840 shares of our common stock outstanding
as of March 31, 2020. The number of shares outstanding as of March 31, 2020 excludes:
|
|
·
|
1,787,164 shares of common stock shares of common stock
reserved for future issuance under our incentive plan and employee stock purchase plan;
|
|
|
·
|
980,667 shares of common stock issuable upon the vesting
of restricted stock units;
|
|
|
·
|
1,241,675 shares issuable upon exercise of outstanding
options with a weighted average exercise price of $5.73 per share; and
|
|
|
·
|
5,402,670 shares issuable upon exercise of outstanding
warrants with a weighted average exercise price of $8.21 per share
|
To
the extent that any options or warrants are exercised, new options are issued under our equity incentive plan or we otherwise
issue additional shares of common stock in the future at a price less than the public offering price, there will be further dilution
to new investors.
PLAN OF DISTRIBUTION
On July 27, 2018, we
entered into an At Market Issuance Sales Agreement, with B. Riley FBR, Inc., Cantor Fitzgerald & Co., National Securities Corporation
and Oppenheimer & Co. Inc. (the “Agents”), under which we may issue and sell shares of our common stock having
an aggregate gross sales price of up to $75 million from time to time through the Agents. The sales agreement has been filed
as an exhibit to our Current Report on Form 8-K filed with the SEC on July 27, 2018. On August 16, 2019, we filed a new registration
statement on Form S-3 (No. 333-233350), which became effective on September 30, 2019. On July 20, 2020, we entered into Amendment
No. 1 to the Sales Agreement with the Agents to reflect the new registration statement. As of the date of this prospectus supplement,
we have sold 5,641,856 common shares under the sales agreement for approximately $30.5 million in gross proceeds. As such, we may
offer and sell the remaining balance of common shares having a maximum aggregate sales price of up to $50.6 million pursuant to
this prospectus supplement.
Upon delivery of a
placement notice and subject to the terms and conditions of the sales agreement, the Agents may sell our common stock by
any method permitted by law deemed to be an “at the market offering” as defined in Rule 415(a)(4) promulgated
under the Securities Act. We may instruct the Agents not to sell common stock if the sales cannot be effected at or above
the price designated by us from time to time. We or the Agents may suspend the offering of common stock upon notice and subject
to other conditions.
We will pay the Agents commissions,
in cash, for their services in acting as an agent in the sale of our common stock. The Agents will be entitled to compensation
at a commission rate of up to 3% of the gross sales price per share sold. Because there is no minimum offering amount
required as a condition to close this offering, the actual total public offering amount, commissions, expenses, and proceeds to
us, if any, are not determinable at this time but will be reported in our periodic reports. We have also agreed to reimburse the
Agents for certain specified expenses, including the fees and disbursements of its legal counsel in an amount not to exceed
$20,000.
Settlement for sales
of common stock will generally occur on the second business day following the date on which any sales are made (or
such earlier day as is industry practice for regular-way trading), or on some other date that is agreed upon by us and the Agents in
connection with a particular transaction, in return for payment of the net proceeds to us. Sales of our common stock as contemplated
in this prospectus supplement will be settled through the facilities of The Depository Trust Company or by such other means as
we and the Agents may agree upon. There is no arrangement for funds to be received in an escrow, trust or similar arrangement.
The Agents will use
its commercially reasonable efforts, consistent with its sales and trading practices, to solicit offers to purchase the common
stock shares under the terms and subject to the conditions set forth in the sales agreement. In connection with the sale of the
common stock on our behalf, the Agents will be deemed to be “underwriters” within the meaning
of the Securities Act and the compensation of the Agents will be deemed to be underwriting commissions or discounts. We have
agreed to provide indemnification and contribution to the Agents against certain civil liabilities, including liabilities
under the Securities Act.
The offering of our
common stock pursuant to the sales agreement will terminate upon the earlier of (1) the sale of all shares of our common
stock subject to the sales agreement having an aggregate offering price of $50.6 million, (unless the parties agree to extend
the sales agreement) or (2) termination of the sales agreement as permitted therein. We and the Agents may each
terminate the respective sales agreement at any time upon five days’ prior notice.
The Agents and its
affiliates may in the future provide various investment banking, commercial banking and other financial services for us and our
affiliates, for which services they may in the future receive customary fees. To the extent required by Regulation M, the
Agents will not engage in any market making activities involving our common stock while the offering is ongoing under this
prospectus supplement.
LEGAL MATTERS
The validity of the
common stock offered hereby will be passed upon by Alston & Bird LLP, New York, New York. Duane Morris LLP, New York, New
York, is counsel for the Agents in connection with this offering.
EXPERTS
The
financial statements as of December 31, 2019 and 2018 and for each of the two years in the period ended December 31, 2019 incorporated
by reference in this Prospectus Supplement have been so incorporated in reliance on the report of BDO USA, LLP, an independent
registered public accounting firm, incorporated herein by reference, given on the authority of said firm as experts in auditing
and accounting.
WHERE YOU CAN
FIND ADDITIONAL INFORMATION ABOUT US
We file reports with
the SEC on an annual basis using Form 10-K, quarterly reports on Form 10-Q and current reports on Form 8-K. You may read and copy
any such reports and amendments thereto at the SECs Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549 on official
business days during the hours of 10:00 a.m. to 3:00 p.m. Please call the SEC at 1-800-SEC-0330 for information on the Public
Reference Room. Additionally, the SEC maintains a website that contains annual, quarterly, and current reports, proxy statements,
and other information that issuers (including us) file electronically with the SEC. The SECs website address is http://www.sec.gov.
You can also obtain copies of materials we file with the SEC from our Internet website found at www.mustangbio.com. Our stock
is quoted on the Nasdaq Capital Market under the symbol “MBIO.”
This prospectus supplement
is only part of a registration statement on Form S-3 that we have filed with the SEC under the Securities Act and therefore omits
certain information contained in the registration statement. We have also filed exhibits and schedules with the registration statement
that are excluded from this prospectus supplement, and you should refer to the applicable exhibit or schedule for a complete description
of any statement referring to any contract or other document. You may inspect a copy of the registration statement, including
the exhibits and schedules, without charge, at the public reference room or obtain a copy from the SEC upon payment of the fees
prescribed by the SEC.
INCORPORATION OF CERTAIN INFORMATION
BY REFERENCE
The SEC allows us
to “incorporate by reference” the information we file with them, which means that we can disclose important information
to you by referring you to those documents instead of having to repeat the information in this prospectus supplement and accompanying
prospectus. The information incorporated by reference is considered to be part of this prospectus supplement and accompanying
prospectus, and later information that we file with the SEC will automatically update and supersede this information. This prospectus
supplement incorporates by reference the documents listed below (other than, unless otherwise specifically indicated, current
reports furnished under Item 2.02, Item 7.01 and Item 9.01 of Form 8-K and exhibits filed on such form that are related to such
items):
|
|
·
|
Our Current Reports on Form 8-K filed with the SEC
on June 15, 2020 and June 19, 2020 (excluding information furnished pursuant to Item 9.01); and
|
All
reports and other documents we subsequently file pursuant to Section 13(a), 13(c), 14 or 15(d) of the Exchange Act prior to the
termination of this offering, including all such documents we may file with the SEC after the date of the initial registration
statement and prior to the effectiveness of the registration statement, but excluding any information furnished to, rather than
filed with, the SEC, will also be incorporated by reference into this prospectus supplement and deemed to be part of this prospectus
supplement from the date of the filing of such reports and documents.
PROSPECTUS
$75,000,000

Common Stock
Preferred Stock
Warrants
Debt Securities
Units
We may offer and sell an indeterminate number
of shares of our common stock, preferred stock, warrants to purchase our common stock, preferred stock, debt securities, or units
comprised of two or more such securities (the “Securities”) from time to time under this prospectus. You should read
this prospectus and any prospectus supplement carefully before you invest.
We may offer the Securities in one or more
offerings in amounts, at prices, and on terms determined at the time of the offering. We may sell the Securities through agents
we select or through underwriters and dealers we select. If we use agents, underwriters or dealers, we will name them and describe
their compensation in a prospectus supplement.
This prospectus provides a general description
of the Securities we may offer. Each time we sell Securities, we will provide specific terms of the Securities offered in a supplement
to this prospectus. The prospectus supplement may also add, update or change information contained in this prospectus. You should
read this prospectus and the applicable prospectus supplement carefully before you invest in any Securities. This prospectus may
not be used to consummate a sale of Securities unless accompanied by the applicable prospectus supplement.
Our common stock are listed for trading on
the Nasdaq Global Market under the symbol “MBIO.”
We are an “emerging growth
company” as defined in the Jumpstart Our Business Startups Act and a “smaller reporting company” as defined in
the Exchange Act, and will therefore be subject to reduced reporting requirements.
Investing in our Securities involves risks.
Neither the Securities and Exchange Commission
nor any state securities commission has approved or disapproved of these securities or passed upon the accuracy or adequacy of
this prospectus. Any representation to the contrary is a criminal offense.
The date of this
Prospectus is September 30, 2019
TABLE
OF CONTENTS
SUMMARY
We are a clinical-stage
biopharmaceutical company focused on translating today’s medical breakthroughs in cell and gene therapies into potential
cures for hematologic cancers, solid tumors and rare genetic diseases. We aim to acquire rights to these technologies by licensing
or otherwise acquiring an ownership interest in the technologies, funding their research and development and eventually either
out-licensing or bringing the technologies to market.
Our pipeline is currently
focused in three core areas: gene therapy programs for rare genetic disorders, chimeric antigen receptor (“CAR”) engineered
T cell (“CAR T”) therapies for hematologic malignancies and CAR T therapies for solid tumors. For each therapy we have
partnered with world class research institutions. For our gene therapy programs, we have partnered with St. Jude Children’s
Research Hospital (“St. Jude”) in the development of a first-in-class ex vivo lentiviral treatment of X-linked severe
combined immunodeficiency (“XSCID”) and for our CAR T therapies we have partnered with the City of Hope National Medical
Center (“COH”), Fred Hutchinson Cancer Research Center (“Fred Hutch”) and Nationwide Children’s Hospital
(“Nationwide”).
Gene Therapy
In partnership with St.
Jude, our gene therapy program (MB-107) is being conducted under an exclusive license to develop a potentially curative treatment
for XSCID, a rare genetic immune system condition in which affected patients do not live beyond infancy without treatment. This
first-in-class ex vivo lentiviral gene therapy is currently in two Phase 1/2 clinical trials sponsored by St. Jude and the
National Institutes of Health (“NIH”). Results in these two trials have been promising and we plan to transfer St.
Jude’s Investigational New Drug Application (“IND”) to Mustang in the second half of 2019 following completion
of the technology transfer process.
CAR T Therapies
Mustang’s pipeline
of CAR T therapies is being developed under exclusive licenses from several world class research institutions. Our strategy is
to license these technologies, support preclinical and clinical research activities by our partners and transfer the underlying
technology to our cell processing facility located in Worcester, Massachusetts to conduct Mustang sponsored clinical trials.
We are developing CAR T therapies for hematologic malignancies
in partnership with COH targeting CD123 (MB-102) and CS1 (MB-104) and with Fred Hutch targeting CD20 (MB-106). Phase 1 clinical
trials sponsored by COH for MB-102 and by Fred Hutch for MB-106 are underway, and a COH sponsored Phase 1 clinical trial for MB-104
opened in April 2019. The U.S. Food and Drug Administration (“FDA”) recently approved our IND application to initiate
a multi-center Phase 1/2 clinical trial of MB-102, and our clinical trial will begin later in 2019. We expect to file an IND for
MB-104 in the second half of 2019 and to initiate our own Phase 1 clinical trial shortly thereafter for the treatment of patients
with multiple myeloma. We also plan to file an IND and initiate our own clinical trial for MB-106 for the treatment of patients
with non-Hodgkin lymphoma and chronic lymphocytic leukemia.
We are also developing CAR T therapies for solid tumors in partnership
with COH targeting IL13Rα2 (MB-101), HER2 (MB-103) and PSCA (MB-105). In addition, we have partnered with Nationwide for the C134
oncolytic virus (MB-108) in order to enhance the activity of MB-101 for the treatment of patients with glioblastoma multiforme
(“GBM”). Phase 1 clinical trials sponsored by COH for MB-101 and MB-103 are underway, and a COH sponsored Phase 1 clinical
trial for MB-105 opened for accrual of patients with prostate cancer in the second quarter of 2019. A Phase 1 clinical trial sponsored
by the University of Alabama at Birmingham for MB-108 is scheduled to commence in the second half of 2019. We also plan to file
INDs and initiate our own clinical trials for MB-103 for the treatment of patients with metastatic breast cancer to brain, for
MB-105 for the treatment of patients with prostate and pancreatic cancer and for the combination of MB-101 and MB-108 for the treatment
of patients with GBM.
Additionally, we hold
complementary patent licenses relating to the use, delivery and possible enhancement of our proprietary CAR technologies. In particular,
we licensed intellectual property from Harvard University pertaining to CRISPR/Cas9 gene editing of CAR T cells, and we hope to
use this technology to enhance the activity of our CAR T cell therapies.
To date, we have not
received approval for the sale of our product candidates in any market and, therefore, have not generated any product sales from
our product candidates. In addition, we have incurred substantial operating losses since our inception, and expect to continue
to incur significant operating losses for the foreseeable future and may never become profitable. As of June 30, 2019, we have
an accumulated deficit of $99.1 million.
Company Information
We are a majority-controlled subsidiary of
Fortress Biotech, Inc. (“Fortress”).
Our principal executive offices are located at 2 Gansevoort Street,
9th Floor, New York, New York 10014, and our telephone number is 781-652-4500. We maintain a website on the Internet at www.mustangbio.com
and our e-mail address is info@mustangbio.com. Our Internet website, and the information contained on it, are not to be considered
part of this prospectus.
Important
information About This Prospectus
In this prospectus, unless
the context suggests otherwise, references to “Mustang Bio,” “Mustang,” the “Company,” “we,”
“us” and “our” refer to Mustang Bio, Inc.
This prospectus is part
of a “shelf” registration statement that we filed with the Securities and Exchange Commission (“SEC”).
By using a shelf registration statement, we may sell our securities, as described in this prospectus, from time to time in one
or more offerings. Each time we sell Securities, we will provide a prospectus supplement to this prospectus that contains specific
information about the terms of such offering. The prospectus supplement may also add, update or change information contained in
this prospectus. Before purchasing any securities, you should carefully read both this prospectus and any prospectus supplement,
together with the additional information incorporated into this prospectus or described under the heading “Where You Can
Find More Information.”
You should rely only on
the information contained or incorporated by reference in this prospectus and any prospectus supplement. We have not authorized
any other person to provide you with different information. If anyone provides you with different or inconsistent information,
you should not rely on it. We will not make an offer to sell Securities in any jurisdiction where the offer or sale is not permitted.
You should assume that the information appearing in this prospectus, as well as information we previously filed with the SEC and
have incorporated by reference, is accurate as of the date on the front cover of this prospectus only, or when such document was
filed with the SEC. Our business, financial condition, results of operations and prospects may have changed since the relevant
date.
Neither we, nor any of
our officers, directors, agents or representatives or underwriters, make any representation to you about the legality of an investment.
You should not interpret the contents of this prospectus, any prospectus supplement, or any free writing prospectus to be legal,
business, investment or tax advice. You should consult with your own advisors for that type of advice and consult with them about
the legal, tax, business, financial and other issues that you should consider before investing in our common stock.
We will not use this prospectus
to offer and sell Securities unless it is accompanied by a prospectus supplement that more fully describes the terms of the offering.
Description
Of Capital Stock
The following description
summarizes the material terms of Mustang capital stock as of the date of this registration statement. Because it is only a summary,
it does not contain all the information that may be important to you. For a complete description of our capital stock, you should
refer to our certificate of incorporation, our bylaws and to the provisions of applicable Delaware law.
Our common stock is
traded on The Nasdaq Capital Market under the symbol “MBIO.” The closing sale price of our common stock on August
14, 2019 was $3.73 per share.
Capital Stock
The Company is authorized
to issue 85,000,000 shares of common stock with a par value of $0.0001 per share, of which 1,000,000 shares are designated as Class
A common stock and 2,000,000 of preferred stock at $0.0001 par value of which 250,000 are designated as Class A preferred stock.
The holders of common
stock are entitled to one vote per share of common stock held.
The undesignated
preferred stock may be issued from time to time in one or more series. The Board of Directors is authorized to determine or alter
the dividend rights, dividend rate, conversion rights, voting rights, rights and terms of redemption (including sinking fund provisions,
if any), the redemption price or prices, the liquidation preferences and other designations, powers, preferences and relative,
participating, optional or other special rights, if any, and the qualifications, limitations and restrictions granted to or imposed
upon any wholly unissued series of preferred stock, and to fix the number of shares of any series of preferred stock (but not below
the number of shares of any such series then outstanding).
Class A Common Stock
The holders of Class A
common stock are entitled to the number of votes equal to the number of whole shares of common stock into which the shares of Class
A common shares held by such holder are convertible. For a period of ten years from issuance, the holders of the Class A common
stock have the right to appoint one member of the board of directors of Mustang. To date, the holders of Class A common stock have
not yet appointed such director.
Class A Preferred Stock
The Class A Preferred
Stock is identical to undesignated common stock other than as to voting rights, conversion rights, and the PIK dividend right.
The holders of the outstanding
shares of Class A Preferred Stock receive on each March 13 (each a “PIK Dividend Payment Date”) after the original
issuance date of the Class A Preferred Stock until the date all outstanding Class A Preferred Stock is converted into common stock
or redeemed (and the purchase price is paid in full), pro rata per share dividends paid in additional fully paid and non-assessable
shares of common stock such that the aggregate number of shares of common stock issued pursuant to such PIK dividend is equal to
2.5% of the Corporation’s fully-diluted outstanding capitalization on the date that is one business day prior to any PIK
Dividend Payment Date (“PIK Record Date”). In the event the Class A Preferred Stock converts into common stock, the
holders shall receive all PIK dividends accrued through the date of such conversion. No dividend or other distribution shall be
paid, or declared and set apart for payment (other than dividends payable solely in capital stock on the capital stock) on the
shares of common stock until all PIK dividends on the Class A Preferred Stock shall have been paid or declared and set apart for
payment. All dividends are non-cumulative.
On any matter presented
to the stockholders for their action or consideration at any meeting of stockholders (or by written consent of stockholders in
lieu of meeting), each holder of outstanding shares of Class A Preferred Stock shall be entitled to cast for each share of Class
A Preferred Stock held by such holder as of the record date for determining stockholders entitled to vote on such matter, the number
of votes that is equal to one and one-tenth (1.1) times a fraction, the numerator of which is the sum of (A) the number of shares
of outstanding common stock and (B) the whole shares of common stock in to which the shares of outstanding Class A Common Stock
and the Class A Preferred Stock are convertible, and the denominator of which is number of shares of outstanding Class A Preferred
Stock. Thus, the Class A Preferred Stock will at all times constitute a voting majority.
Each share of Class A Preferred Stock
is convertible, at the option of the holder, into one fully paid and nonassessable share of common stock, subject to certain adjustments.
If the Company, at any time effects a subdivision or combination of the outstanding common stock (by any stock split, stock dividend,
recapitalization, reverse stock split or otherwise), the applicable conversion ratio in effect immediately before that subdivision
is proportionately decreased or increased, as applicable, so that the number of shares of common stock issuable on conversion of
each share of Class A Preferred Stock shall be increased or decreased, as applicable, in proportion to such increase or decrease
in the aggregate number of shares of common stock outstanding. Additionally, if any reorganization, recapitalization, reclassification,
consolidation or merger involving the Company occurs in which the common stock (but not the Class A Preferred Stock) is converted
into or exchanged for securities, cash or other property, then each share of Class A Preferred Stock becomes convertible into the
kind and amount of securities, cash or other property which a holder of the number of shares of common stock of the Company issuable
upon conversion of one share of the Class A Preferred Stock immediately prior to such reorganization, recapitalization, reclassification,
consolidation or merger would have been entitled to receive pursuant to such transaction.
Additional Features
Other features of our
capital stock include:
|
|
·
|
Dividend Rights. The holders of outstanding shares of our common stock, including Class A common stock, are entitled to receive dividends out of funds legally available at the times and in the amounts that our board of directors may determine. All dividends are non-cumulative.
|
|
|
·
|
Voting Rights. The holders of our common stock are entitled to one vote for each share of common stock held on all matters submitted to a vote of the stockholders, including the election of directors. Our certificate of incorporation and bylaws do not provide for cumulative voting rights.
|
|
|
·
|
No Preemptive or Similar Rights. The holders of our common stock have no preemptive, conversion, or subscription rights, and there are no redemption or sinking fund provisions applicable to our common stock.
|
|
|
·
|
Right to Receive Liquidation Distributions. Upon our liquidation, dissolution, or winding-up, the assets legally available for distribution to our stockholders would be distributable ratably among the holders of our common stock, including Class A common stock, outstanding at that time after payment of other claims of creditors, if any.
|
|
|
·
|
Fully Paid and Non-Assessable. All of the outstanding shares of our common stock, including Class A common stock, and the Class A Preferred Stock are duly issued, fully paid and non-assessable.
|
DESCRIPTION
OF WARRANTS
We may issue warrants
to purchase shares of our common stock or preferred stock in one or more series together with other securities or separately, as
described in each applicable prospectus supplement.
The prospectus supplement
relating to any warrants we offer will include specific terms relating to the offering. These terms will include some or all of
the following:
|
|
·
|
the title of the warrants;
|
|
|
·
|
the aggregate number of warrants offered;
|
|
|
·
|
the designation, number and terms of the shares of common stock purchasable upon exercise of the warrants and procedures by which those numbers may be adjusted;
|
|
|
·
|
the exercise price of the warrants;
|
|
|
·
|
the dates or periods during which the warrants are exercisable;
|
|
|
·
|
the designation and terms of any securities with which the warrants are issued;
|
|
|
·
|
if the warrants are issued as a unit with another security, the date on and after which the warrants and the other security will be separately transferable;
|
|
|
·
|
if the exercise price is not payable in U.S. dollars, the foreign currency, currency unit or composite currency in which the exercise price is denominated;
|
|
|
·
|
any minimum or maximum amount of warrants that may be exercised at any one time;
|
|
|
·
|
any terms relating to the modification of the warrants;
|
|
|
·
|
any terms, procedures and limitations relating to the transferability, exchange or exercise of the warrants; and
|
|
|
·
|
any other specific terms of the warrants.
|
DESCRIPTION
OF DEBT SECURITIES
We may offer debt securities
which may be senior, subordinated or junior subordinated and may be convertible. Unless otherwise specified in the applicable prospectus
supplement, our debt securities will be issued in one or more series under an indenture to be entered into between us and a trustee.
We will issue the debt securities offered by this prospectus and any accompanying prospectus supplement under an indenture to be
entered into between us and the trustee identified in the applicable prospectus supplement. The terms of the debt securities will
include those stated in the indenture and those made part of the indenture by reference to the Trust Indenture Act of 1939, as
in effect on the date of the indenture. We have filed a copy of the form of indenture as an exhibit to the registration statement
in which this prospectus is included. The indenture will be subject to and governed by the terms of the Trust Indenture Act of
1939.
The following description
briefly sets forth certain general terms and provisions of the debt securities that we may offer. The particular terms of the debt
securities offered by any prospectus supplement and the extent, if any, to which these general provisions may apply to the debt
securities, will be described in the related prospectus supplement. Accordingly, for a description of the terms of a particular
issue of debt securities, reference must be made to both the related prospectus supplement and to the following description.
Debt Securities
The aggregate principal
amount of debt securities that may be issued under the indenture is unlimited. The debt securities may be issued in one or more
series as may be authorized from time to time pursuant to a supplemental indenture entered into between us and the trustee or an
order delivered by us to the trustee. For each series of debt securities we offer, a prospectus supplement accompanying this prospectus
will describe the following terms and conditions of the series of debt securities that we are offering, to the extent applicable:
|
|
·
|
title and aggregate principal amount;
|
|
|
·
|
whether the debt securities will be senior, subordinated or junior subordinated;
|
|
|
·
|
applicable subordination provisions, if any;
|
|
|
·
|
provisions regarding whether the debt securities will be convertible or exchangeable into other securities or property of the Company or any other person;
|
|
|
·
|
percentage or percentages of principal amount at which the debt securities will be issued;
|
|
|
·
|
interest rate(s) or the method for determining the interest rate(s);
|
|
|
·
|
whether interest on the debt securities will be payable in cash or additional debt securities of the same series;
|
|
|
·
|
dates on which interest will accrue or the method for determining dates on which interest will accrue and dates on which interest will be payable;
|
|
|
·
|
whether the amount of payment of principal of, premium, if any, or interest on the debt securities may be determined with reference to an index, formula or other method;
|
|
|
·
|
redemption, repurchase or early repayment provisions, including our obligation or right to redeem, purchase or repay debt securities under a sinking fund, amortization or analogous provision;
|
|
|
·
|
if other than the debt securities’ principal amount, the portion of the principal amount of the debt securities that will be payable upon declaration of acceleration of the maturity;
|
|
|
·
|
authorized denominations;
|
|
|
·
|
amount of discount or premium, if any, with which the debt securities will be issued, including whether the debt securities will be issued as “original issue discount” securities;
|
|
|
·
|
the place or places where the principal of, premium, if any, and interest on the debt securities will be payable;
|
|
|
·
|
where the debt securities may be presented for registration of transfer, exchange or conversion;
|
|
|
·
|
the place or places where notices and demands to or upon the Company in respect of the debt securities may be made;
|
|
|
·
|
whether the debt securities will be issued in whole or in part in the form of one or more global securities;
|
|
|
·
|
if the debt securities will be issued in whole or in part in the form of a book-entry security, the depository or its nominee with respect to the debt securities and the circumstances under which the book-entry security may be registered for transfer or exchange or authenticated and delivered in the name of a person other than the depository or its nominee;
|
|
|
·
|
whether a temporary security is to be issued with respect to such series and whether any interest payable prior to the issuance of definitive securities of the series will be credited to the account of the persons entitled thereto;
|
|
|
·
|
the terms upon which beneficial interests in a temporary global security may be exchanged in whole or in part for beneficial interests in a definitive global security or for individual definitive securities;
|
|
|
·
|
the guarantors, if any, of the debt securities, and the extent of the guarantees and any additions or changes to permit or facilitate guarantees of such debt securities;
|
|
|
·
|
any covenants applicable to the particular debt securities being issued;
|
|
|
·
|
any defaults and events of default applicable to the debt securities, including the remedies available in connection therewith;
|
|
|
·
|
currency, currencies or currency units in which the purchase price for, the principal of and any premium and any interest on, such debt securities will be payable;
|
|
|
·
|
time period within which, the manner in which and the terms and conditions upon which the Company or the purchaser of the debt securities can select the payment currency;
|
|
|
·
|
securities exchange(s) on which the debt securities will be listed, if any;
|
|
|
·
|
whether any underwriter(s) will act as market maker(s) for the debt securities;
|
|
|
·
|
extent to which a secondary market for the debt securities is expected to develop;
|
|
|
·
|
provisions relating to defeasance;
|
|
|
·
|
provisions relating to satisfaction and discharge of the indenture;
|
|
|
·
|
any restrictions or conditions on the transferability of the debt securities;
|
|
|
·
|
provisions relating to the modification of the indenture both with and without the consent of holders of debt securities issued under the indenture;
|
|
|
·
|
any addition or change in the provisions related to compensation and reimbursement of the trustee;
|
|
|
·
|
provisions, if any, granting special rights to holders upon the occurrence of specified events;
|
|
|
·
|
whether the debt securities will be secured or unsecured, and, if secured, the terms upon which the debt securities will be secured and any other additions or changes relating to such security; and
|
|
|
·
|
any other terms of the debt securities that are not inconsistent with the provisions of the Trust Indenture Act (but may modify, amend, supplement or delete any of the terms of the indenture with respect to such series of debt securities).
|
General
One or more series of debt
securities may be sold as “original issue discount” securities. These debt securities would be sold at a substantial
discount below their stated principal amount, bearing no interest or interest at a rate which at the time of issuance is below
market rates. One or more series of debt securities may be variable rate debt securities that may be exchanged for fixed rate debt
securities.
United States federal income
tax consequences and special considerations, if any, applicable to any such series will be described in the applicable prospectus
supplement.
Debt securities may be issued
where the amount of principal and/or interest payable is determined by reference to one or more currency exchange rates, commodity
prices, equity indices or other factors. Holders of such debt securities may receive a principal amount or a payment of interest
that is greater than or less than the amount of principal or interest otherwise payable on such dates, depending upon the value
of the applicable currencies, commodities, equity indices or other factors. Information as to the methods for determining the amount
of principal or interest, if any, payable on any date, the currencies, commodities, equity indices or other factors to which the
amount payable on such date is linked and certain additional United States federal income tax considerations will be set forth
in the applicable prospectus supplement.
The term “debt securities”
includes debt securities denominated in U.S. dollars or, if specified in the applicable prospectus supplement, in any other freely
transferable currency or units based on or relating to foreign currencies.
We expect most debt securities
to be issued in fully registered form without coupons and in denominations of $1,000 and any integral multiples thereof. Subject
to the limitations provided in the indenture and in the prospectus supplement, debt securities that are issued in registered form
may be transferred or exchanged at the principal corporate trust office of the trustee, without the payment of any service charge,
other than any tax or other governmental charge payable in connection therewith.
Global Securities
The debt securities of
a series may be issued in whole or in part in the form of one or more global securities that will be deposited with, or on behalf
of, a depositary identified in the prospectus supplement. Global securities will be issued in registered form and in either temporary
or definitive form. Unless and until it is exchanged in whole or in part for the individual debt securities, a global security
may not be transferred except as a whole by the depositary for such global security to a nominee of such depositary or by a nominee
of such depositary to such depositary or another nominee of such depositary or by such depositary or any such nominee to a successor
of such depositary or a nominee of such successor. The specific terms of the depositary arrangement with respect to any debt securities
of a series and the rights of and limitations upon owners of beneficial interests in a global security will be described in the
applicable prospectus supplement.
Governing Law
The indenture and the debt
securities shall be construed in accordance with and governed by the laws of the State of New York.
DESCRIPTION
OF UNITS
We may issue, in one more
series, units comprised of shares of our common stock, preferred stock, warrants to purchase common stock or preferred stock, debt
securities or any combination of those securities. Each unit will be issued so that the holder of the unit is also the holder of
each security included in the unit. Thus, the holder of a unit will have the rights and obligations of a holder of each included
security. The unit agreement under which a unit is issued may provide that the securities included in the unit may not be held
or transferred separately, at any time or at any time before a specified date.
We may evidence units by
unit certificates that we issue under a separate agreement. We may issue the units under a unit agreement between us and one or
more unit agents. If we elect to enter into a unit agreement with a unit agent, the unit agent will act solely as our agent in
connection with the units and will not assume any obligation or relationship of agency or trust for or with any registered holders
of units or beneficial owners of units. We will indicate the name and address and other information regarding the unit agent in
the applicable prospectus supplement relating to a particular series of units if we elect to use a unit agent.
We will describe in the
applicable prospectus supplement the terms of the series of units being offered, including:
|
|
·
|
the designation and terms of the units and of the securities comprising the units, including whether and under what circumstances those securities may be held or transferred separately;
|
|
|
·
|
any provisions of the governing unit agreement that differ from those described herein; and
|
|
|
·
|
any provisions for the issuance, payment, settlement, transfer or exchange of the units or of the securities comprising the units.
|
The other provisions regarding
our common stock, preferred stock, warrants and debt securities as described in this section will apply to each unit to the extent
such unit consists of shares of our common stock, warrants and/or debt securities.
Plan
Of Distribution
We may sell the securities covered in this
prospectus in any of three ways (or in any combination):
|
|
·
|
through underwriters or dealers;
|
|
|
·
|
directly to a limited number of purchasers or to a single purchaser; or
|
Each time that we use
this prospectus to sell securities, we will also provide a prospectus supplement that contains the specific terms of the offering.
The prospectus supplement will set forth the terms of the offering of the securities, including:
|
|
·
|
the name or names of any underwriters, dealers or agents and the amounts of any securities underwritten or purchased by each of them; and
|
|
|
·
|
the public offering price of the common stock and the proceeds to us and any discounts, commissions or concessions allowed or reallowed or paid to dealers.
|
Any public offering price
and any discounts or concessions allowed or reallowed or paid to dealers may be changed from time to time.
If underwriters are used
in the sale of any securities, the securities will be acquired by the underwriters for their own account and may be resold from
time to time in one or more transactions, including negotiated transactions, at a fixed public offering price or at varying prices
determined at the time of sale. The securities may be either offered to the public through underwriting syndicates represented
by managing underwriters, or directly by underwriters. Generally, the underwriters’ obligations to purchase the securities
will be subject to certain conditions precedent. The underwriters will be obligated to purchase all of the securities if they purchase
any of securities.
We may sell the securities
through agents from time to time. The prospectus supplement will name any agent involved in the offer or sale of the securities
and any commissions we pay to them. Generally, any agent will be acting on a best efforts basis for the period of its appointment.
We may authorize underwriters,
dealers or agents to solicit offers by certain purchasers to purchase the securities from us at the public offering price set forth
in the prospectus supplement pursuant to delayed delivery contracts providing for payment and delivery on a specified date in the
future. The contracts will be subject only to those conditions set forth in the prospectus supplement, and the prospectus supplement
will set forth any commissions we pay for solicitation of these contracts.
Agents and underwriters
may be entitled to indemnification by us against certain civil liabilities, including liabilities under the Securities Act of 1933,
as amended, or to contribution with respect to payments which the agents or underwriters may be required to make in respect thereof.
Agents and underwriters may be customers of, engage in transactions with, or perform services for us in the ordinary course of
business.
We may enter into derivative
transactions with third parties, or sell securities not covered by this prospectus to third parties in privately negotiated transactions.
If the applicable prospectus supplement indicates, in connection with those derivatives, the third parties may sell securities
covered by this prospectus and the applicable prospectus supplement, including in short sale transactions. If so, the third party
may use securities pledged by us or borrowed from us or others to settle those sales or to close out any related open borrowings
of securities, and may use securities received from us in settlement of those derivatives to close out any related open borrowings
of securities. The third party in such sale transactions will be an underwriter and will be identified in the applicable prospectus
supplement (or a post-effective amendment).
Legal
Matters
Certain legal matters
will be passed upon for us by Alston & Bird LLP, New York, New York. Additional legal matters may be passed upon for us
or any underwriters, dealers or agents, by counsel that we will name in the applicable prospectus supplement.
Experts
The financial
statements as of December 31, 2018 and 2017 and for each of the two years in the period ended December 31, 2018 incorporated
by reference in this Prospectus have been so incorporated in reliance on the report of BDO USA, LLP, an independent
registered public accounting firm, incorporated herein by reference, given on the authority of said firm as experts in
auditing and accounting.
Where
You Can Find More Information
We have filed with the
SEC, Washington, D.C. 20549, a registration statement on Form S-3 under the Securities Act with respect to the common stock offered
hereby. This prospectus does not contain all of the information set forth in the registration statement and the exhibits and schedules
thereto. For further information with respect to the Company and its common stock, reference is made to the registration statement
and the exhibits and any schedules filed therewith. Statements contained in this prospectus as to the contents of any contract
or other document referred to are not necessarily complete and in each instance, if such contract or document is filed as an exhibit,
reference is made to the copy of such contract or other document filed as an exhibit to the registration statement, each statement
being qualified in all respects by such reference. A copy of the registration statement, including the exhibits and schedules thereto,
may be read and copied at the SEC’s Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549. Information on the
operation of the Public Reference Room may be obtained by calling the Securities and Exchange Commission (the “SEC”
or “Commission”) at 1-800-SEC-0330. In addition, the SEC maintains an Internet site at www.sec.gov, from which interested
persons can electronically access the registration statement, including the exhibits and any schedules thereto.
We are subject to the
information reporting requirements of the Exchange Act, and we file periodic reports and other information with the SEC. All documents
filed with the SEC are available for inspection and copying at the addresses set forth above. We also maintain an Internet site
at www.mustangbio.com. Our website and the information contained therein or connected thereto shall not be deemed to be
incorporated into this prospectus or the registration statement of which it forms a part.
INCORPORATION
OF CERTAIN DOCUMENTS BY REFERENCE
The SEC allows us to “incorporate
by reference” the information we file with them which means that we can disclose important information to you by referring
you to those documents instead of having to repeat the information in this prospectus and accompanying prospectus. The information
incorporated by reference is considered to be part of this prospectus and accompanying prospectus, and later information that we
file with the SEC will automatically update and supersede this information. This prospectus incorporates by reference the documents
listed below (other than, unless otherwise specifically indicated, current reports furnished under Item 2.02 or Item 7.01 of Form
8-K and exhibits filed on such form that are related to such items):
|
|
e)
|
The description of the Common Stock as included under the caption “Description of Registrant’s Securities to be Registered” in our Registration Statement on Form 10-12G, as amended, originally filed with the Commission on July 28, 2016, and our Registration Statement on Form 8-A12B filed with the Commission on August 21, 2017, and any amendment or report filed for the purpose of further updating such description.
|
All reports and other
documents we subsequently file pursuant to Section 13(a), 13(c), 14 or 15(d) of the Exchange Act prior to the termination of this
offering, including all such documents we may file with the SEC after the date of the initial registration statement and prior
to the effectiveness of the registration statement, but excluding any information furnished to, rather than filed with, the SEC,
will also be incorporated by reference.

Mustang Bio, Inc.
Up to $50,605,000
Common Stock
|
|
B. Riley FBR
|
|
|
Cantor
|
|
|
National Securities Corporation
|
|
|
Oppenheimer
& Co.
|
July 20, 2020
Mustang Bio (NASDAQ:MBIO)
Historical Stock Chart
From Mar 2024 to Apr 2024
Mustang Bio (NASDAQ:MBIO)
Historical Stock Chart
From Apr 2023 to Apr 2024