As filed
with
the
Securities
and
Exchange Commission
on December
5,
2016 Registration No. 333-214641
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C.
20549
PRE-EFFECTIVE
AMENDMENT
NO. 1
TO
FORM
S-3
REGISTRATION
STATEMENT UNDER THE SECURITIES ACT OF
1933
ImmuCell
Corporation
(Exact
name
of
registrant
as
specified in
its
charter)
|
Delaware
|
|
01-0382980
|
|
(State
or other
jurisdiction
of
incorporation
or organization)
|
|
(I.R.S.
Employer Identification Number)
|
56
Evergreen
Drive
Portland,
ME 04103
(207)
878-2770
(Address,
including zip
code,
and telephone number, including
area code, of
registrant’s
principal executive offices)
Michael
F. Brigham
President,
Chief Executive Officer and Treasurer
56
Evergreen
Drive
Portland,
ME 04103
(207)
878-2770
(Name,
address,
including zip
code,
and telephone number,
including
area code, of
agent
for
service)
Copy
to:
David
J.
Champoux
Pierce Atwood
LLP
254
Commercial Street
Portland,
ME 04101
(207)
791-1100
Approximate
date of
commencement
of
proposed
sale
to
the public:
From time
to
time after this Registration Statement
becomes
effective.
If
the
only
securities
being
registered
on
this
Form are being
offered pursuant
to dividend
or
interest reinvestment
plans,
please
check
the
following box:
¨
If
any of the
securities
being
registered
on
this
Form are
to
be
offered
on a
delayed
or
continuous
basis
pursuant to
Rule
415
under
the Securities Act
of 1933,
other than securities offered
only
in
connection with dividend
or
interest
reinvestment
plans,
check the
following
box:
þ
If
this
Form
is
filed
to register additional securities for
an
offering pursuant to Rule
462(b)
under the Securities Act, please check
the
following
box
and
list
the Securities Act
registration
statement number
of the earlier
effective
registration statement for the same
offering.
¨
If
this
Form
is
a
post-effective
amendment filed pursuant to Rule
462(c)
under the Securities Act,
check
the
following
box
and list
the
Securities
Act registration statement number
of the
earlier effective registration statement
for
the
same offering.
¨
If
this
Form
is
a
registration
statement pursuant to General Instruction
I.D. or a
post-effective amendment thereto
that shall become effective
upon
filing with
the Commission
pursuant
to Rule
462(e)
under the Securities Act,
check
the
following
box.
¨
If
this
Form
is
a
post-effective
amendment to
a
registration statement filed pursuant to
General
Instruction
I.D.
filed to register additional securities
or
additional
classes
of securities
pursuant to Rule
413(b)
under
the Securities Act,
check the
following
box.
¨
In
dicate
by
check
mark
whether
the
registrant is
a
large
accelerated
filer,
an
accelerated
filer,
a
non-accelerated filer
or a
smaller reporting company. See
the
definitions
of
“large accelerated filer”, “accelerated filer”
and
“smaller reporting company”
in
Rule
12b-2 of
the
Securities
Exchange
Act
of 1934.
|
Large
accelerated filer
☐
|
Accelerated
filer
☐
|
Non-accelerated
filer ☐
|
Smaller
reporting company ☒
|
|
|
|
|
|
CALCULATION
OF REGISTRATION FEE
|
Title
of each
class
of
securities
to
be registered
|
|
Amount
to
be registered
(1)(2)
|
|
Proposed
maximum
offering price per Share(2)
|
|
Aggregate
maximum offering price (2)
|
|
Amount
of
registration fee
|
|
Common
Stock, par value $0.10 per share (3)
|
|
659,880
shares
|
|
$
|
5.16
|
|
|
$
|
3,404,981
|
|
|
$
|
394.64
|
|
|
(1)
|
Pursuant
to Rule
416
under
the
Securities Act
of 1933
(the
“Securities Act”), this
registration
statement
also
covers
any additional
securities that
may be
offered
or
issued in
connection
with
any
stock split, stock dividend
or
similar transaction.
|
|
(2)
|
Estimated
solely
for
the
purpose of
calculating
the amount of
the
registration fee in
accordance
with Rule
457(c)
under
the
Securities Act. The
proposed
maximum
offering
price per
share
and proposed
maximum
aggregate offering
price are based
upon the average
of
the
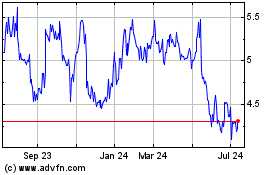
high ($5.24)
and
low ($5.08)
sales
price of
the registrant’s
common
stock
on
November
15, 2016, as
reported
on
the NASDAQ Capital Market.
|
|
(3)
|
Also
includes rights
issuable
in
respect of any
such shares
of
Common
Stock pursuant to the registrant’s
Common
Stock
Rights Plan,
as amended, as
more fully
described
in
the
prospectus forming a part of
this registration statement.
|
The
Registrant hereby amends this
Registration
Statement
on
such
date or dates as
may
be
necessary
to
delay its
effective
date
until the Registrant shall file
a
further amendment
which
specifically
states that
this
Registration
Statement
shall
thereafter
become
effective
in
accordance with
Section
8(a) of
the
Securities
Act of 1933 or
until the
Registration
Statement
shall
become
effective on
such
date as
the Securities
and
Exchange Commission,
acting pursuant
to
said Section
8(a),
may
determine.
The
information in this
preliminary
prospectus
is
not
complete
and
may
be
changed.
The
Selling Stockholders
may
not sell these securities until
the
registration
statement filed with the Securities
and Exchange Commission
is effective.
This
prospectus is not
an
offer to sell these
securities and the Selling Stockholders
are
not soliciting offers to
buy
these
securities in
any
state where
the
offer
or
sale
of
these securities
is
not
permitted.
|
|
Subject To
Completion,
Dated December
5, 2016
|
|
Prospectus
|
|
IMMUCELL
CORPORATION
659,880
Shares
of
Common
Stock
This
prospectus covers the
sale,
transfer
or
other disposition
of
up to 659,880 shares
of
common stock
by
certain
Selling
Stockholders, which,
as used
herein, includes
donees,
pledgees,
transferees, and other successors-in-interest selling shares
of
common
stock
received
after
the date of
this
prospectus from a Selling Stockholder as a
gift, pledge, partnership distribution
or
other transfer,
or
the Selling Stockholders.
The Selling Stockholders may,
from
time to
time,
sell,
transfer
or
otherwise
dispose of
any
or
all
of
their shares
of
common stock on any stock
exchange, market,
or
trading
facility
on
which the shares
are traded or
in
private transactions.
These
dispositions
may be at
fixed
prices,
at
prevailing market
prices at
the
time
of
sale,
at prices related
to
the
prevailing
market
price, at
varying
prices
determined
at
the time
of
sale,
or at
negotiated prices.
ImmuCell
is not offering
any
shares
of common stock
for
sale under this prospectus.
We
will
not
receive
any
of
the
proceeds from
the sale
or
other disposition
of the
shares
of common stock
by
the
Selling
Stockholders.
Our
common stock
is listed
on
the
NASDAQ
Capital
Market under
the
symbol
“ICCC.”
The
aggregate market
value
of our outstanding common
stock held
by
non-affiliates was
approximately $24,779,280 based on 4,847,390
shares
of
outstanding
common
stock,
of
which
863,583
shares
are
held
by
affiliates,
and a
price of $6.22 per
share, which was the last
reported
sale
price of
our
common
stock
as
quoted
on NASDAQ
Capital Market
on December 1, 2016.
INVESTING
IN OUR COMMON STOCK INVOLVES
RISKS.
YOU SHOULD REVIEW
CAREFULLY
THE
RISKS
AND
UNCERTAINTIES DESCRIBED
UNDER
THE
HEADING “RISK
FACTORS”
CONTAINED HEREIN
AND IN OUR ANNUAL REPORT ON
FORM
10-K FOR
THE
YEAR ENDED DECEMBER
31,
2015,
AND
UNDER SIMILAR
HEADINGS
IN
THE
OTHER DOCUMENTS THAT ARE INCORPORATED BY
REFERENCE
INTO THIS PROSPECTUS.
NEITHER
THE
SECURITIES AND
EXCHANGE
COMMISSION
NOR ANY STATE SECURITIES COMMISSION HAS APPROVED OR DISAPPROVED THESE SECURITIES OR
DETERMINED
IF THIS PROSPECTUS IS TRUTHFUL OR COMPLETE.
ANY
REPRESENTATION
TO
THE
CONTRARY
IS A CRIMINAL OFFENSE.
The
date of
this prospectus is December
5, 2016.
TABLE
OF
CONTENTS
CAUTIONARY
NOTE
REGARDING
FORWARD-LOOKING STATEMENTS
Some
of
the
statements
in
this
prospectus constitute “forward-looking
statements” within the meaning
of Section 27A of the
Securities Act
of 1933
and Section
21E of the
Securities Exchange
Act
of
1934. These
statements
relate
to future
events concerning our
business and
to our future
revenues,
operating results
and
financial
condition.
In some cases,
you
can identify
forward-looking
statements
by
terminology such as
“may”,
“will”, “could”, “would”, “should”, “expect”, “plan”,
“anticipate”, “aim”, “intend”, “believe”, “estimate”, “target”,
“forecast”, “predict”,
“project”,
“propose”,
“potential”,
or
“continue”,
or the
negative
of those terms or
other
comparable
terminology.
Any
forward-looking statements contained
in
this prospectus
are only estimates or
predictions
of
future events
based on
information
currently
available to our management and management’s current beliefs
about
the
potential outcome
of
future events. Whether
these future events will occur
as
management anticipates, whether
we will
achieve our
business
objectives,
and
whether our revenues, operating
results or
financial
condition will be
sustained
or improve
in
future
periods are subject
to numerous
risks. There are a number of
important factors that could cause actual
results
to differ materially
from the
results anticipated
by
these forward-looking statements.
These
important
factors include those that
we discuss
under the heading “Risk
Factors”
and in other sections
of
our Annual
Report on Form 10-K
for the year ended December
31, 2015
and
our
Form 10-Q for
the
quarterly period
ended
September
30, 2016,
all filed with
the
Securities
and
Exchange Commission (“SEC”),
as
well
as
in
our
other
reports
filed
from
time to time with
the SEC that are incorporated by
reference into this prospectus.
You
should
read
these
factors
and the other
cautionary
statements made in this prospectus and in
the documents we incorporate
by
reference into
this
prospectus
as
being applicable
to all related forward-looking statements wherever they
appear
in
this prospectus or the documents we incorporate
by
reference into this
prospectus. If
one
or
more
of
these factors materialize,
or
if any
underlying
assumptions
prove incorrect,
our
actual
results, performance
or
achievements
may vary
materially
from
any
future
results,
performance
or
achievements expressed
or
implied
by
these
forward-looking statements.
We
undertake no
obligation
to
publicly
update
any forward-looking
statements,
whether
as a
result
of new
information,
future events
or
otherwise,
except as
required
by
law.
ABOUT
THIS PROSPECTUS
This
document is called
a
prospectus and is
part of a registration
statement
that
we
have filed with
the
SEC,
using a
“shelf”
registration
process.
If
there
is
any inconsistency between the
information
in
this
prospectus
or
information
incorporated by
reference
having
a
later
date,
you
should
rely
on the incorporated
information having
a
later
date. We
urge you to
read
carefully this prospectus, together with
the
information
incorporated
herein
by
reference
as
described under
the heading “Where
You
Can Find
More
Information,”
before
buying
any of
the
securities being
offered.
You
should rely
only on
the information
we
have
provided or
incorporated
by
reference in this prospectus.
We
have
not,
and
the Selling Stockholders have not,
authorized
anyone
to
provide
you with different information.
No dealer,
salesperson
or
other
person
is authorized to give
any information or
to represent anything not contained in this
prospectus.
Neither
the
delivery
of
this
prospectus nor
any
sale made under it implies that there
has been
no
change in our affairs
or
that the information in this prospectus is
correct
as of any date
after the
date of
this prospectus.
You should
assume
that the
information
in
this prospectus
is
accurate
only
as
of the date on
the front
of this
prospectus, and
that
any
information
we
have
incorporated
by
reference is accurate
only as
of
the
date of the
document
incorporated
by
reference, regardless
of the
time
of delivery
of
this prospectus
or any
sale of a
security.
This
prospectus
contains summaries
of
certain
provisions
contained
in
some
of
the documents described
herein, but reference
is
made
to the actual
documents
for complete information.
All
of
the
summaries are
qualified
in
their
entirety
by the actual
documents.
Copies of
some
of
the
documents
referred to
herein
have
been
filed, will
be
filed
or
will
be
incorporated
by
reference
as
exhibits
to
the
registration statement
of
which
this
prospectus
is
a part, and
you
may obtain copies of
those
documents as described below under
“Where
You
Can
Find
More
Information”.
In
this prospectus, unless
the
context otherwise
requires, references to “we”, “us”, “our”
or
similar
terms,
as
well
as
references to “ImmuCell”
or
the “Company”, refer to
ImmuCell Corporation.
ABOUT
IMMUCELL
CORPORATION
We
are a
growing
animal
health
company
whose
purpose
is
to
create scientifically-proven and practical
products
that result in
a
measurable
economic impact
on animal
health and productivity in
the dairy and beef
industries.
We
were original
incorporated
in Maine
in
1982
and reincorporated in Delaware in
1987,
in
conjunction with our initial public offering
of common
stock.
We
have
developed products
that
provide
significant,
immediate immunity
to newborn
dairy
and
beef
cattle and
are
in the late stages
of developing a new product
that
addresses
mastitis,
the most
significant
cause
of
economic
loss to
the dairy
industry.
Across
all product lines, our product
sales for the year ended December
31, 2015
increased
by
35%,
or
$2,632,000,
to
$10,229,000
from
$7,597,000
in
2014,
and gross margins
as
a
percentage
of product sales
were
61%
in
2015, as
compared to
59%
during
2014.
Growth
in
sales
of
our lead
product,
First
Defense
® and
related
product
line extensions,
has
driven
the
increase
in our total
product
sales. Sales
of
First Defense
®,
and
related product line extensions, aggregated
92.8%
and
91.6% of
our total
product sales during the
years
ended
December
31, 2015
and
2014,
respectively. Sales
of
First Defense
®
and
related
product
line extensions increased
by
36%, 27% and 14%
during
the
years
ended
December
31, 2015, 2014
and
2013,
respectively, in
comparison
to the
prior
years.
First Defense
® is manufactured
from
hyperimmune
cows’ colostrum (the
milk
that a
cow
produces immediately
after
giving
birth)
utilizing our proprietary vaccine
and
milk
protein
purification
technologies.
The
target disease, bovine enteritis (calf scours),
causes
diarrhea
and dehydration
in
newborn
calves
and
often leads
to serious sickness
and even
death.
First
Defense
®
is the
only USDA-
licensed,
orally delivered scours
preventive
product on
the market for calves with claims against
E. coli
K99
and
coronavirus (two
leading causes of
scours).
First
Defense
®
provides
bovine
antibodies
that
newborn
calves need but
are
unable to
produce
on
their
own immediately
after birth. Our milk
antibody products provide
Immediate
Immunity
™
during
the
first
few critical days
of
life
when calves
need
this protection
most. Studies have shown
that calves
that
scour are
more susceptible to other diseases later
in
life and
under-perform calves that do
not
contract scours.
During
the
third quarter
of 2016,
our total product
sales
declined
20%, or $504,000,
to
$1,968,000
from $2,472,000
during
the
same
period
in
2015. For
the nine-month
period
ended September
30, 2016,
our total
product
sales declined
3%, or $204,000,
to
$7,330,000
from
$7,534,000
during the
same
period
in
2015.We
experienced
net operating income
of $50,000
during the third quarter
of 2016
(compared
to
$627,000
in the third quarter
of
2015).
We recorded
net operating income
of $771,000
during
the
nine-month
period
ended September
30, 2016,
compared to
$1,661,000
during
the comparable
period of 2015.
We
recorded
net
income
of $35,000, or
$0.01
per
diluted
share, during
the
third quarter
of 2016
compared to
$351,000, or $0.11 per
diluted
share during the
comparable period of 2015. We
recorded net income
of
$478,000, or
$0.11
per
diluted share,
during
the
nine-month
period
ended September
30,
2016
compared to
$924,000,
or
$0.29
per
diluted share, during the
comparable period of 2015.
We
believe that the
declines
experienced in
the
third quarter
of 2016
in
First Defense
®
sales
are
attributable to several factors:
|
-
|
|
Customer
order
patterns
were disrupted during the
prolonged period of order
backlog
from
first quarter
of 2015
until
the
early part of
the
third quarter
of
2016.
|
|
-
|
|
After
completing a
significant
investment to
double
First
Defense®
production capacity during early 2016,
the
distribution
chain
was re-supplied
with
product
largely
during
the
second quarter
of
2016.
|
|
-
|
|
When
customers were unable to secure
supply of
First
Defense®
,
some
moved to other products
(with and without claims), and some
opted
to
use no scours preventative
at
all.
|
|
-
|
|
A
competitive
product,
that
experienced interrupted
supply
to the market
during late
2014
and
through
the
first half
of
2015,
has returned to the market. Some customers
have reverted
back
to
using
this
product.
|
|
-
|
|
The
Company’s sales
team needs
to
regain momentum
lost
when it was not
able
to focus
on
acquiring
new
customers
during
the period of
scarce product supply.
|
|
-
|
|
Based
on
our informal
survey
of
market sources, the
value of a
bull
calf
has
dropped
from
approximately
$450
last year
to
about $50
to
$200
presently.
|
|
-
|
|
The
average Class
III
milk
price
has
declined from $15.80 per
hundred pounds during
2015
(compared to
$22.34
for
2014)
to
$14.38
during
the
first
nine
months
of
2016. The
average
price of $13.48
during the first
six
months
of the
year increased to
$16.18
during
the
third quarter
of
2016.
|
Although
some of
these
unfavorable
conditions
appear
to
be
improving,
we
do
not
expect
to see total sales for
2016
reaching
the
historic high
levels
experienced
in
2015.
The
majority of
our
product
development budget
from 2000
through
2016
has
been
focused
on
the development
of
Mast
Out
®,
a
Nisin-based
intramammary treatment
of
subclinical mastitis
in
lactating
dairy
cows.
During the
16.75-year period
that
began on January 1, 2000
(the
year
we
began
the
development of
Mast Out
®)
and
ended on September
30,
2016,
we
invested
the
aggregate
of
approximately $12,271,000
in
the development of
Mast
Out
®.
This estimated
allocation to
Mast Out
®
reflects
only direct
expenditures
and
includes
no
allocation of product development
or
administrative
overhead expenses. Approximately
$2,891,000 of
this
investment
was
offset
by
product
licensing
revenues
and grant
income
related to
Mast Out
®.
Nisin
is
an antibacterial peptide
that has
been
demonstrated
in
clinical
studies
to
be an
effective
aid
in the reduction
of
mastitis-causing organisms
in
dairy
cows.
Mastitis
is
a very common infection
in
dairy cows that results
in
inflammation
of
the
mammary gland.
Because
dairy producers are
required to discard
milk
for a period
during
and
after
treatment
with all currently
marketed
mastitis
treatment
products
due to
concerns about
antibiotic
residue in
milk,
it is generally current
practice to
only
treat mastitis when the
disease has
progressed
to the clinical
stage
where the milk
from
an
infected cow cannot be sold. We
believe
that
Mast Out®
could
revolutionize the way
that
mastitis is treated
by
making earlier treatment
of
subclinically
infected cows
economically feasible
by
not
requiring a
milk
discard
during,
or
for
a period of
time after, treatment.
No
other
FDA-approved
mastitis treatment
product on
the market
can
offer this value
proposition.
Commercial
introduction
of
Mast Out®
in
the
United States
is
subject
to approval
of
our
New
Animal
Drug
Application
by
the
U.S. Food
and
Drug
Administration’s
Center for
Veterinary Medicine
(FDA), which
approval
cannot
be
assured. Foreign
regulatory approvals
would
be
required for sales in
key
markets
outside of the
United States, which would involve some similar and
some
different
requirements.
During
the
third quarter
of 2016,
we
began
construction of a
major expansion
of
our manufacturing facilities to enable us to
produce
Nisin
for
use in the
production and
sale
of
Mast
Out®
.
We
anticipate
that construction
of
the building
will be completed by
the
end
of
the third quarter
of 2017,
and
that
equipment
installation will
be
completed
during the first quarter
of 2018. This facility
would then
produce
validation
batches
for submittal to and
review by FDA, and the
facility
will
be
inspected
by
FDA,
as
part of
our
NADA
application
process,
which
we hope
will
be
complete
in 2019,
enabling commercial
sales of
Mast Out®
to
commence.
The
estimated
cost
of
this
production
facility
is
$20
million.
As of
September
30,
2016,
our
cash
and cash
equivalents totaled
$9,607,000
(including
approximately
$5.3
million
in
net proceeds
from
our
public offering
of
common
stock
completed in
February 2016
and excluding
$343,000
held
temporarily in escrow).
We
previously arranged for secured
credit
facilities
provided by
TD
Bank
totaling
$4.5
million
and a
line
of credit totaling $500,000
to
be
available to fund
a portion of the cost
of
the
Nisin production
facility. Approximately
$651,000
had
been
paid
towards
this project as
of
September
30, 2016. The
remaining
cost of
the
production
facility
(approximately $4.4
million)
we expect
to fund with the
approximately
$3.2
million
in
net
proceeds
from
the common
stock issuance
described
in
“Private Placement
of
Common Shares”, and with
a combination
of cash
generated
by operations
in
the
fourth
quarter
of 2016
and the first and second quarters
of 2017
and
possibly some additional
bank borrowings.
Our
principal executive
offices are
located
at 56
Evergreen Drive,
Portland, ME 04103.
Our telephone
number is
(207) 878-2770.
Our website is located
at
www.immucell.com.
Information
contained
on, or
that
can be
accessed through, our website is not
part of
this
prospectus.
PRIVATE
PLACEMENT
OF
COMMON
SHARES
On
October 17, 2016
we
entered into
a
purchase
agreement (the “Securities Purchase Agreement”) with institutional and accredited
investors
as
part
of
a private placement
pursuant
to which
we
agreed to issue
a
total
of 659,880
share
of
our
common
stock
(the “Shares”) for
an
aggregate
purchase
price of $3,464,370.
Closing
of the
private
placement pursuant to the Securities Purchase Agreement occurred
on October
21,
2016.
In
connection with the Securities Purchase Agreement,
we
also entered
into
a
Registration Rights
Agreement
(the
“Registration Rights Agreement”) pursuant to which
we
have filed with
the SEC
the
registration statement
of
which this prospectus
forms a part,
relating
to the offer
and
sale
or
other disposition
by
the holders
of the
Shares.
Pursuant to
the
Registration Rights Agreement,
we
are
obligated to file
the
registration
statement
by
November
16, 2016
and
to use commercially
reasonable efforts
to
cause the
registration
statement to
be declared
effective
by
January 15, 2017,
which
is
90
days
from the date of
the Securities Purchase Agreement. Failure to meet
those
and related obligations,
or
failure to maintain
the effective registration
of
the Shares, will
subject
ImmuCell
to payment
of
liquidated damages.
Copies
of
the Securities
Purchase
Agreement
and
the
Registration
Rights Agreement
are
incorporated
by
reference
as
exhibits
to the registration statement
of
which this
prospectus
forms
a part. The
foregoing
summaries of each
of
the transaction
documents,
including
the
warrants,
are
qualified in their
entirety
by
reference to such
documents.
THE
OFFERING
|
Common stock outstanding:
|
|
4,847,390
shares
(1)
|
|
|
|
|
|
Common
Stock that
may
be sold or
otherwise
disposed of by the Selling
Stockholders:
|
|
659,880
shares
|
|
|
|
|
|
NASDAQ Capital Market symbol for common
stock:
|
|
ICCC
|
|
|
|
|
|
Use of proceeds:
|
|
We
will
not
receive
any of
the proceeds from the
sale or other disposition of the Shares covered
by
this
prospectus
|
|
|
|
|
|
Risk factors:
|
|
See “Risk
Factors”
in
our Annual
Report on Form 10-K
for
the
year
ended
December
31, 2015 as
well
as
our subsequently
filed
periodic
and current
reports,
for
a
discussion
of
factors
to consider
before
investing
in
shares
of our common
stock.
|
|
|
|
|
|
(1)
|
|
The
number
of
shares
shown to
be
outstanding is
based
on
the number
of
shares
of
our
common
stock outstanding
as
of
December
1, 2016,
and
does not
include shares reserved for issuance
upon the
exercise
of options
granted
or
available under stock option
plans.
|
WHERE
YOU
CAN FIND
ADDITIONAL INFORMATION
We
have filed with
the SEC a
registration statement
on
Form
S-3 under the Securities Act
of
1933, as
amended (“Securities Act”),
with
respect
to the securities
covered
by
this
prospectus. This
prospectus, which
is
a part of
the registration statement,
does not
contain
all
of
the
information
set
forth
in
the registration statement
or
the exhibits and schedules filed therewith.
For
further
information
with
respect
to us and
the
securities
covered by
this
prospectus,
please see the registration statement and the
exhibits
filed
with the registration statement.
A copy
of
the
registration statement and the
exhibits
filed
with the registration
statement
may be
inspected without charge
at
the
Public
Reference
Room
maintained
by the
SEC, located
at 100 F
Street,
N.E.,
Washington, D.C.
20549. Please
call the
SEC
at 1-800-SEC-0330
for more information
about the operation of
the
Public Reference Room.
The
SEC also
maintains a
website
that contains
reports, proxy and
information statements
and
other
information
regarding
registrants
that
file
electronically
with the SEC. The
address of
the
website
is
http://www.sec.gov.
We
are
subject to the information and
periodic
reporting
requirements
of the
Securities Exchange Act
of 1934, as
amended
(the “Exchange Act”) and, in
accordance
therewith,
we
file
periodic
reports,
proxy
statements and
other
information with
the
SEC.
Such periodic reports, proxy
statements
and
other
information are
available
for inspection and copying
at the
Public Reference
Room
and
website
of
the
SEC
referred to above.
We
maintain
a
website
at
www.immucell.com.
You
may
access
our Annual
Report
on Form 10-K,
Quarterly
Reports on Form 10-Q,
Current
Reports on Form 8-K
and amendments to those
reports
filed
pursuant to Sections
13(a)
or
15(d) of
the
Exchange Act with the
SEC
free
of
charge
at
our website
as
soon as reasonably
practicable after such material is electronically filed with, or furnished
to,
the
SEC.
Our website
and
the
information
contained
on
that site,
or
connected
to that site,
are
not incorporated into and
are
not
a part of
this prospectus.
INCORPORATION
OF INFORMATION BY
REFERENCE
The
SEC allows us to “incorporate
by
reference”
the
information
we
file
with it, which means
that
we
can
disclose
important
information
to you
by
referring
you to
those documents. The
information
we
incorporate
by
reference
is
an
important
part of
this prospectus,
and
certain
information that
we
will
later
file with
the SEC
will
automatically update
and supersede this information.
We
incorporate
by
reference
the
documents
listed below,
as
well
as any
future filings made with the
SEC under
Sections
13(a),
13(c),
14 or 15(d) of the
Exchange
Act
from the date of the initial
registration statement and
prior
to
the effectiveness
of
this registration statement, and
any
filings
made after the
date of
this
prospectus
until
we sell all
of the
securities
under
this
prospectus,
except
that
we
do
not incorporate any
document or portion of a
document
that
was furnished and deemed
by
the rules
of the
SEC not to have
been
filed:
|
·
|
|
Our
Annual
Report on Form 10-K
for
the fiscal year ended December
31, 2015,
filed
with the SEC
on
March
25,
2016;
|
|
·
|
|
Our
Quarterly
Reports on Form 10-Q
for
the quarters ended
March 31, 2016,
June
30,
2016 and September
30,
2016,
filed with
the
SEC,
respectively,
on May 11, 2016,
August
11,
2016
and November
10, 2016;
|
|
·
|
|
Our
Current
Reports on Form
8-K
filed with
the
SEC
on
January 7, 2016, January 29, 2016, January 29, 2016, February 3, 2016,
February
10, 2016, March 7, 2016, March 31, 2016, May 11, 2016, May 23, 2016,
June
15,
2016,
June
16,
2016, August
11,
2016,
October 11, 2016, October 18,
2016, October 18, 2016,
October 21,
2016
and November
10, 2016;
and
|
|
·
|
|
Our
definitive
proxy statement on
Schedule
14A
filed
on
April
28, 2016
for our annual meeting
of
shareholders held
on
June
15, 2016.
|
|
·
|
|
Our
Form 8-A
filed
with
the SEC on March 18, 1987
with
respect
to our Common Stock.
|
|
·
|
|
Our
Form 8-A
filed
with
the SEC on
September
13, 1995,
as
amended
by
Form 8-A/A
filed with the SEC
on
June
30, 2008,
with
respect
to our Common
Stock
Purchase
Rights.
|
Additionally,
all
reports and
other
documents subsequently
filed
by
us
pursuant to
Sections 13(a), 13(c), 14
and
15(d) of
the
Exchange Act
after
(i) the
date of
the
initial registration statement and
prior
to
effectiveness
of
the
registration
statement;
and
(ii)
the date of
this prospectus and
prior
to
the
termination
or completion
of
this offering,
shall
be deemed
to
be
incorporated
by
reference in this
prospectus and
to
be part
hereof
from the date
of
filing
of
such
reports and other
documents.
Any
information that
we
subsequently
file with the SEC that is incorporated
by
reference
as described
above
will automatically update and supersede
any previous
information
that
is
part of
this prospectus.
We
hereby undertake to
provide
without charge
to
each person,
including
any
beneficial
owner,
to whom
a
copy
of
this
prospectus
is delivered,
upon
written
or oral
request
of any such
person,
a copy of
any
and
all
of
the
information
that has
been or
may
be
incorporated
by
reference in
this
prospectus, other
than
exhibits to
such
documents.
Requests for such
copies
should
be
directed to our
Corporate
Secretary
at 56
Evergreen Drive,
Portland, ME 04103.
Our
telephone
number
is
(207) 878-2770.
RISK
FACTORS
An
investment
in
our
common
stock involves risks.
Prior
to
making
a
decision
about
investing in
our
common
stock, you should
consider
carefully
the risks together with
all
of
the other information
contained
or incorporated
by
reference in this prospectus,
including any risks described
in the
section
entitled
“Risk Factors” contained in our Annual
Report on Form 10-K
for the fiscal
year ended December
31, 2015
and our subsequent
filings
with
the SEC.
DESCRIPTION
OF COMMON STOCK
The
description
below
of
our
common stock and
provisions
of
our certificate
of
incorporation
and
bylaws
are
summaries
and are
qualified
by
reference to
the
certificate
of
incorporation and the bylaws.
These documents are filed as
exhibits
to the registration statement
of
which
this
prospectus
is
a
part.
Our
authorized capital stock consists
of
10,000,000
shares
of common
stock.
As
of
December 1, 2016,
there were
4,847,390
shares
of common stock
outstanding.
The
holders
of common stock are
entitled to receive
ratably
dividends,
if any,
as
may
be declared
from time
to time
by
the Board of
Directors out
of
funds
legally
available for that
purpose. In
the event
of
our
liquidation,
dissolution or
winding up, whether voluntary
or
involuntary,
the
holders
of
common stock are
entitled
to share
ratably
in
all
assets remaining
after payment
of
or provision
for liabilities.
The common
stock has no preemptive
or
conversion
rights or
other subscription rights.
There are
no
redemption
or sinking
fund provisions
applicable
to
the
common
stock. All
outstanding
shares
of common stock are fully paid
and nonassessable, and the shares
of common
stock to
be
issued
upon the
closing
of
this
offering will be
fully
paid
and nonassessable.
The
holders
of
common
stock are
entitled
to one vote
per
share
on
all matters
to
be
voted
upon by
the shareholders.
There
is no cumulative voting.
Effect
of Certain
Provisions
of our
Certificate
of
Incorporation,
Bylaws and
Common
Stock
Rights
Plan
Provisions
of
our
certificate
of
incorporation,
our bylaws,
our Common
Stock Rights
Plan or
Delaware
law may
discourage,
delay
or
prevent
a
merger,
acquisition or
other
change
in
control
that
stockholders may
consider
favorable,
including transactions
in
which
stockholders
might otherwise receive
a premium
for
their
shares
of
our
common stock. These
provisions
may also
prevent
or
frustrate attempts
by
our stockholders to
replace
or
remove our management.
These
provisions
include:
|
·
|
|
limitations
on the
removal
of
directors; advance notice requirements
for stockholder proposals and nominations;
|
|
·
|
|
the
ability
of
our
Board of
Directors
to alter
or repeal
our bylaws;
|
|
·
|
|
the
ability of
our
Board of Directors
to refuse to
redeem
rights
issued
under
our
Common Stock
Rights
Plan or
otherwise
to limit
or
suspend its
operation
that would work to dilute the stock ownership
of a
potential hostile
acquirer, likely
preventing acquisitions
that
have
not
been approved by
our
Board of
Directors; and
|
|
·
|
|
Section
203 of
the
Delaware
General
Corporation
Law,
which prohibits
a
publicly-held Delaware
corporation from
engaging
in
a
business
combination
with
an
interested
stockholder
(generally
defined
as
a person
which together with its affiliates
owns,
or
within the
last three
years
has
owned, 15% of
our voting stock, for
a period of
three years after
the
date
of the
transaction
in
which
the person
became
an
interested
stockholder) unless the business
combination
is
approved
in
a prescribed
manner.
|
The
existence
of
the
foregoing provisions
and
anti-takeover measures could
depress the
trading
price of
our
common
stock
or
limit
the price
that investors might
be
willing to
pay in
the
future
for shares
of
our
common
stock.
They
could also
deter
potential acquirers
of
our Company,
thereby
reducing
the
likelihood of
obtaining
a premium
for our
common
stock
in an
acquisition.
These
provisions,
summarized
below,
are
expected
to discourage coercive takeover
practices
and inadequate takeover
bids
and
to promote stability in our management.
These
provisions
are
also
designed to encourage persons
seeking
to acquire control
of us
to
first negotiate with
our board of
directors.
|
·
|
|
Shareholder
Meetings
. Our bylaws
provide that a
special
meeting
of
shareholders
may be
called
only
by
the
President
or by
the
Board of
Directors
or by
shareholders holding
a
majority
of the outstanding
shares
of
our
common
stock.
|
|
·
|
|
Requirements
for
Advance
Notification
of
Shareholder
Nominations
and
Proposals
.
Our bylaws establish advance notice
procedures
with
respect
to
shareholder proposals
and
the nomination
of
candidates
for
election
as
directors, other than nominations
made
by
or at
the
direction of
our
Board of
Directors
or a
committee
of
the
Board of
Directors.
|
|
·
|
|
Board
of
Directors
Vacancies
. Under our bylaws,
any
vacancy
on
the
Board of
Directors,
including
a vacancy
resulting
from
an
enlargement
of
the
Board of
Directors,
may
only be
filled
by
vote
of a
majority
of
the
remaining
directors.
Any
director
may
be
removed
by
vote
of the holders of a
majority
of
the outstanding shares
of
our
common
stock.
The
limitations
on the
removal
of directors
and filling
of
vacancies
would have
the
effect
of
making it more difficult for
a
third
party
to acquire control
of us, or
of
discouraging
a
third
party from
acquiring control
of
us.
|
|
·
|
|
Board
of
Directors Size
.
Within
the
range specified
by
our bylaws, our
Board of
Directors
determines
the
size
of
our
board
and may create
new
directorships and elect
new directors,
which
may enable an incumbent board
to
maintain control
by
adding
directors.
|
|
·
|
|
Indemnification
.
Our certificate
of
incorporation
and our bylaws,
as
amended, provide
that
we
will indemnify officers
and directors
against
losses as they
incur
in investigations
and
legal
proceedings
resulting
from
their
services to us, which
may
include service
in
connection
with takeover defense measures.
|
In
September
1995,
our
Board of
Directors
adopted a
Common Stock
Rights Plan
and
declared a
dividend
of
one
common
share purchase
right (a
“Right”)
for
each of the then
outstanding shares
of
the
common stock of the
Company.
Each Right
entitles
the registered
holder to purchase
from the Company
one
share
of common stock at an initial purchase price of $70.00 per
share, subject to
adjustment.
The
description and
terms of the
Rights
are
set forth
in
a
Rights Agreement
between
the
Company and
American
Stock
Transfer
& Trust
Company, LLC,
as
Rights Agent.
The
Rights
(as amended) become
exercisable and
transferable
apart
from
the common
stock
upon
the
earlier
of
i)
10
days
following a
public announcement
that a person or group
(Acquiring
Person)
has, without
the prior
consent
of the
Continuing Directors
(as such term
is
defined
in
the Rights Agreement), acquired
beneficial
ownership
of 20% or
more
of
the
outstanding common stock or
ii)
10 days following commencement
of a tender
offer
or
exchange offer the consummation
of
which
would result in ownership
by
a person or
group
of 20% or
more
of
the outstanding
common stock
(the earlier
of
such
dates
being
called the Distribution
Date).
Upon
the Distribution Date,
the
holder
of
each Right
not
owned
by
the
Acquiring
Person
would
be
entitled to
purchase
common
stock
at a
discount
to the initial purchase
price of $70.00 per
share, effectively
equal
to
one
half of the
market
price of a
share
of common stock on
the
date
the Acquiring
Person
becomes
an
Acquiring
Person.
If, after the Distribution Date, the
Company
should
consolidate
or
merge
with
any
other
entity
and
the
Company
were
not the surviving
company, or,
if the
Company
were the surviving
company,
all
or part of
the
Company’s common stock
were
changed
or
exchanged into the securities
of
any
other
entity,
or
if
more than
50% of
the
Company’s
assets
or
earning
power
were
sold, each
Right
would entitle
its
holder to purchase,
at the Rights’
then-current purchase
price, a
number
of
shares
of the
acquiring company’s
common stock
having
a
market value
at
that time equal to twice
the
Right’s
exercise price.
At
any
time
after
a person or group
becomes
an
Acquiring
Person
and
prior
to the acquisition
by
such
person or
group
of 50% or
more
of
the
outstanding
common
stock,
the Board of
Directors
of
the
Company
may exchange the Rights
(other
than Rights owned
by
such person
or
group which have become void), in whole
or
in
part, at an
exchange ratio
of
one
share of common stock per
Right
(subject
to
adjustment).
At
any
time
prior
to
14
days
following
the
date that any person
or
group becomes an
Acquiring
Person (subject
to extension
by
the
Board of
Directors), the
Board of
Directors
of
the
Company may redeem
the then
outstanding Rights
in
whole,
but not in
part, at a price of $0.005 per
Right,
subject
to
adjustment.
On
June
8, 2005,
our
Board of
Directors
voted to authorize
an
amendment
of
the Rights Agreement to extend the Final Expiration Date
by
an
additional three years, to September
19, 2008.
As
of June 30,
2005,
we
entered
into
an
amendment to the Rights Agreement with the Rights Agent reflecting
such
extension.
On June 6, 2008
our Board
of
Directors voted to authorize
an
amendment
of
the
Rights Agreement to extend the
Final Expiration Date
by an
additional
three years, to September
19, 2011,
and to increase the ownership threshold for determining
“Acquiring
Person”
status
from 15%
to
18%. As of
June
30, 2008,
we
entered
into
an
amendment to
the
Rights
Agreement with
the
Rights Agent reflecting such extension
and
threshold
increase.
On
August
5, 2011,
our
Board of
Directors voted to authorize amendments
of
the Rights Agreement to extend
the
Final
Expiration Date
by
an additional
three
years to September
19, 2014
and to increase the ownership threshold for determining
“Acquiring
Person”
status
from 18%
to
20%.
As
of
August
9, 2011,
we
entered into
an
amendment to
the
Rights Agreement with
the
Rights Agent
reflecting
such
extension
and
threshold
increase.
On
June
10, 2014,
our
Board
of
Directors voted to authorize
an
amendment
to the Rights Agreement to extend the final expiration
date by an additional
three
years to September
19,
2017.
As
of
June
16, 2014,
we
entered
into
an amendment
to the
Rights
Agreement
with
the
Rights Agent reflecting
such
extension.
As of
April
15, 2015,
we
entered
into
an
amendment to the Rights Agreement with
the Rights Agent
deleting
the
provisions
requiring
that redemptions of
the Rights, waivers
or
consents
avoiding “Acquiring
Person”
status
or
certain
amendments
to the
Rights
Agreement
be approved by “Continuing
Directors”.
No
other
changes have
been
made to the
terms
of
the Rights
or
the Rights Agreement.
Our
Board of
Directors believes that
there
is
some risk
that
the
potential value
of
the
Mast Out
® product
development
initiative
is
not fairly reflected in the
market
price of
our
common
stock,
as
it fluctuates
from time
to time, and
that
opportunistic
buyers
could
take
advantage
of that disparity
to
the
detriment
of
our
stockholders. If
this
were to
happen and
result
in
a
potential
threat
through
an
unsolicited
acquisition
effort
or
otherwise, our
Board of
Directors feels
that the
Common
Stock
Rights
Plan could
enhance stockholder
value by providing
management
with negotiating leverage.
Listing
Our
common stock
is listed
on
the
NASDAQ
Capital
Market
under the symbol
“ICCC”.
Transfer
Agent and Registrar
The
transfer agent
and
registrar for our common
stock
is
American
Stock
Transfer
& Trust
Company, LLC.
PLAN
OF DISTRIBUTION
Each
Selling
Stockholder
(the
“Selling Stockholders”)
of
the
Shares and
any
of
their
pledgees, assignees and successors-in-interest may, from time to time, sell
any or
all
of
the Shares covered
hereby on
the
NASDAQ Capital
Market or
any other stock exchange, market
or
trading
facility on
which the
Shares are traded or
in
private transactions. These sales may
be at
fixed
or
negotiated
prices.
A
Selling
Stockholder
may
use any one or
more
of
the following methods
when selling Shares:
|
*
|
|
ordinary
brokerage
transactions
and
transactions
in
which
the
broker-dealer solicits purchasers;
|
|
|
|
|
|
*
|
|
block
trades
in which
the
broker-dealer
will attempt to
sell Shares as
agent
but
may
position
and
resell
a portion of
the
block as
principal
to facilitate
the
transaction;
|
|
|
|
|
|
*
|
|
purchases
by
a
broker-dealer
as
principal
and resale
by
the
broker-dealer
for its account;
|
|
|
|
|
|
*
|
|
an
exchange distribution
in
accordance
with the
rules of the
applicable
exchange;
|
|
|
|
|
|
*
|
|
privately
negotiated transactions;
|
|
|
|
|
|
*
|
|
settlement
of short
sales;
|
|
|
|
|
|
*
|
|
in
transactions
through
broker-dealers
that agree with the Selling Stockholders to sell
a
specified
number
of
such
Shares at a
stipulated
price per
Share;
|
|
|
|
|
|
*
|
|
through
the
writing
or
settlement
of
options
or
other
hedging
transactions,
whether
through an
options exchange
or
otherwise;
|
|
|
|
|
|
*
|
|
a
combination
of
any
such methods
of
sale;
or
|
|
|
|
|
|
*
|
|
any
other
method permitted pursuant to
applicable
law.
|
The
Selling Stockholders may also sell
Shares under
Rule
144 or any
other
exemption from
registration
under the Securities Act
of 1933, as
amended (the “
Securities Act
”),
if available, rather than under this prospectus.
Broker-dealers
engaged
by the
Selling
Stockholders
may
arrange for other
broker-dealers
to participate in sales. Broker-dealers may
receive
commissions
or
discounts
from
the
Selling
Stockholders
(or,
if
any
broker-dealer
acts as
agent for the
purchaser
of
securities,
from
the purchaser)
in amounts to
be
negotiated, but,
except as
set
forth in
a
supplement to this prospectus, in
the case of an agency
transaction
not in
excess of a customary brokerage
commission
in
compliance with
FINRA
Rule
2440;
and in the case
of a
principal transaction
a
markup
or
markdown
in
compliance with
FINRA IM-2440.
In
connection with the sale
of
the
Shares
or interests therein, the Selling
Stockholders may enter into
hedging
transactions
with
broker-dealers or
other financial institutions, which
may
in
turn
engage in
short
sales
of
the
Shares
in
the
course
of hedging
the positions
they
assume.
The
Selling Stockholders may
also
sell
shares of common
stock
short
and
deliver Shares to close out their short positions,
or loan or
pledge Shares to broker-dealers that in
turn may sell
Shares.
The
Selling Stockholders may
also
enter
into
option or
other transactions with broker-dealers
or
other
financial
institutions or
create one
or
more
derivative securities which require
the delivery
to such
broker-dealer or
other financial institution
of
Shares offered
by this
prospectus, which
Shares such
broker-dealer
or
other financial institution
may resell
pursuant
to
this
prospectus
(as
supplemented
or
amended to reflect such transaction).
The
Selling Stockholders and
any broker-dealers or
agents
that are
involved
in
selling the
Shares
may be
deemed to
be
“underwriters”
within the meaning
of the
Securities Act in
connection
with
such sales.
In
such
event,
any
commissions
received
by
such
broker-dealers
or
agents
and
any
profit
on the
resale
of
the Shares
purchased
by
them may be
deemed to
be
underwriting
commissions
or discounts under
the Securities Act.
Each
Selling
Stockholder has
informed
the
Company that
it
does
not have
any
written
or oral
agreement
or
understanding,
directly or
indirectly, with
any
person
to distribute the Shares.
The
Company
is
required
to
pay
certain
fees
and
expenses
incurred
by
the Company incident
to
the
registration
of
the
Shares.
The Company has
agreed to
indemnify
the
Selling
Stockholders against
certain
losses,
claims, damages
and
liabilities,
including
liabilities
under the Securities
Act.
The
Company
has
agreed to keep the registration
statement that includes this prospectus effective until the earlier
of
(i)
the date on
which the
Shares
may
be
resold
by
the
Selling
Stockholders without registration
and
without regard to
any
volume
or
manner-of-sale limitations
by
reason of
Rule
144,
without
the
requirement for
the Company
to
be
in compliance with
the
current public
information
requirement
under
Rule
144
under the Securities Act
or any
other
rule of
similar effect
or
(ii) all
of
the Shares
have
been
sold pursuant to this
prospectus or
Rule
144 under the
Securities
Act
or
any
other
rule
of
similar effect.
The
Shares will
be
sold
only through
registered or licensed
brokers or
dealers if required under
applicable
state
securities laws.
In
addition, in certain states,
the Shares
covered
hereby
may not
be
sold unless
they
have
been
registered
or
qualified
for sale in the
applicable
state
or an
exemption
from
the
registration or
qualification
requirement
is
available and is
complied
with.
Under
applicable
rules and
regulations
under
the Exchange
Act,
any
person
engaged
in
the distribution of
the Shares
may
not
simultaneously
engage
in
market
making activities with
respect
to the
Company’s common stock
for
the
applicable
restricted
period,
as
defined
in
Regulation
M, prior
to the
commencement
of
the
distribution.
In
addition,
the
Selling Stockholders will
be
subject
to
applicable
provisions
of
the
Exchange Act and the rules and regulations
thereunder,
including
Regulation
M,
which
may limit the
timing
of
purchases and sales
of the common
stock
by
the
Selling Stockholders
or
any other
person.
The Company
will make
copies of
this
prospectus available
to the
Selling
Stockholders and has
informed
them
of
the need to deliver
a copy of
this prospectus to
each
purchaser
at
or
prior to the time
of the sale
(including
by
compliance with Rule
172
under
the Act).
SELLING
STOCKHOLDERS
The
shares
of common
stock
being
offered
by
the Selling
Stockholders
are
those
previously
issued to the Selling Stockholders.
For
additional information regarding
the issuances of those
shares
of
common
stock, see
"Private
Placement
of Common
Shares"
above.
We are
registering
the
Shares in
order
to permit the Selling Stockholders to offer the
Shares
for
resale
from
time to time.
Except
for
the ownership
of
the
Shares
and other
shares
of common stock previously acquired by
such
Selling
Stockholders,
the Selling Stockholders have not had
any
material relationship with
the Company
within the
past
three years.
The
table
below
lists
the
Selling
Stockholders and other information regarding
the beneficial
ownership
of
the
shares
of
common stock
by
each of
the Selling Stockholders. The
second column lists
the
number
of
shares
of common
stock beneficially
owned
by
each Selling
Stockholder
as of
November
16, 2016.
The
third column lists the
shares of common stock being offered by
this
prospectus
by
the
Selling Stockholders.
In
accordance
with
the terms of
the Registration
Rights Agreement, this prospectus
generally
covers
the
resale
of
the
sum
of
the
number
of shares of common stock
issued to the
Selling
Stockholders
in
the
transaction
described above,
in
“Private Placement
of Common
Shares”,
as of the trading day
immediately
preceding
the applicable date of
determination and all
subject
to
adjustment
as provided
in the Registration Rights Agreement.
The
fourth
column
assumes the
sale
of
all
of
the
Shares
offered
by
the
Selling
Stockholders pursuant
to
this prospectus.
|
|
|
|
Number
of shares of Common Stock Owned Prior to this
|
|
|
|
Maximum
Number
of
shares
of
Common
Stock to
be
Sold
Pursuant to this
|
|
|
|
Number
of
shares
of
Common
Stock
Owned
after this Offering
|
|
|
Name
of
Selling
Stockholder
|
|
|
Offering
|
|
|
|
Prospectus
|
|
|
|
Numbers
|
|
|
|
Percentage
|
|
|
Norman
H. Pessin SEP IRA fbo Norman H. Pessin
|
|
|
480,000
|
|
|
|
230,000
|
|
|
|
250,000
|
|
|
|
5.16
|
%
|
|
Dolphin Offshore Partners,
L.P.
|
|
|
150,000
|
|
|
|
150,000
|
|
|
|
0
|
|
|
|
0
|
|
|
Craton Capital, L.P.
|
|
|
171,428
|
|
|
|
95,238
|
|
|
|
76,190
|
|
|
|
1.57
|
%
|
|
Brian Lee Pessin
|
|
|
40,000
|
|
|
|
20,000
|
|
|
|
20,000
|
|
|
|
*
|
|
|
Sudbury Capital Fund,
L.P.
|
|
|
35,000
|
|
|
|
10,000
|
|
|
|
25,000
|
|
|
|
*
|
|
|
CVI Investments, Inc.(1)
|
|
|
29,762
|
|
|
|
29,762
|
|
|
|
0
|
|
|
|
0
|
|
|
Iroquois Master Fund,
Ltd.
|
|
|
19,000
|
|
|
|
19,000
|
|
|
|
0
|
|
|
|
0
|
|
|
Iroquois Capital Investment
Group LLC
|
|
|
5,000
|
|
|
|
5,000
|
|
|
|
0
|
|
|
|
0
|
|
|
Warberg WF IV L.P.
|
|
|
7,500
|
|
|
|
7,500
|
|
|
|
0
|
|
|
|
0
|
|
|
Anthony Polak
|
|
|
9,983
|
|
|
|
9,524
|
|
|
|
459
|
|
|
|
*
|
|
|
Domaco Venture Capital
Fund
|
|
|
30,524
|
|
|
|
9,524
|
|
|
|
21,000
|
|
|
|
*
|
|
|
Maura Kelly
|
|
|
9,524
|
|
|
|
9,524
|
|
|
|
0
|
|
|
|
0
|
|
|
RL Capital Partners,
L.P.
|
|
|
9,523
|
|
|
|
9,523
|
|
|
|
0
|
|
|
|
0
|
|
|
Jamie Polak
|
|
|
4,762
|
|
|
|
4,762
|
|
|
|
0
|
|
|
|
0
|
|
|
Margrit Polak
|
|
|
4,762
|
|
|
|
4,762
|
|
|
|
0
|
|
|
|
0
|
|
|
RBC Capital Markets,
LLC (Custodian fbo Ronald Lazar IRA)
|
|
|
10,661
|
|
|
|
4,761
|
|
|
|
5,900
|
|
|
|
*
|
|
|
Steven Farber
|
|
|
72,500
|
|
|
|
5,000
|
|
|
|
67,500
|
|
|
|
1.39
|
%
|
|
John Lipman
|
|
|
18,000
|
|
|
|
18,000
|
|
|
|
0
|
|
|
|
0
|
|
|
Kevin Harris
|
|
|
18,000
|
|
|
|
18,000
|
|
|
|
0
|
|
|
|
0
|
|
|
* Less than one percent
|
(1)
Heights Capital Management, Inc., the
authorized
agent
of
CVI Investments, Inc.
(“CVI”),
has discretionary
authority
to vote and
dispose of
the shares held
by
CVI
and
may
be deemed
to
be
the
beneficial owner
of
these shares. Martin
Kobinger,
in
his capacity
as
Investment Manager
of Heights
Capital Management, Inc., may also
be
deemed
to have investment discretion and
voting
power over the shares held
by
CVI.
Mr.
Kobinger disclaims
any
such
beneficial ownership to
the
shares.
CVI investments,
Inc.
is affiliated with one
or
more
FINRA
member, none
of
whom
are
currently
expected
to participate in the sale
pursuant to this prospectus
of
shares purchased
by
CVI
in
the private
offering
described
elsewhere
herein.
LEGAL
MATTERS
Pierce
Atwood LLP
will
pass upon
legal
matters in
connection
with
the validity
of
the securities offered hereby.
EXPERTS
The
financial statements
of ImmuCell Corporation as of
December
31, 2015
and
2014
and for
each
of
the
two
years
in
the
period
ended
December
31, 2015
incorporated
by
reference
in
this
prospectus from the
Annual
Report on Form 10-K
for
the
year
ended
December
31, 2015
have
been
so
incorporated
in reliance
on
the
report of
Baker Newman
& Noyes
LLC,
an
independent registered public accounting firm,
incorporated
herein
by reference,
given
on
the authority
of
said firm
as
experts
in
auditing
and
accounting.
INTERESTS
OF
NAMED
EXPERTS
AND
COUNSEL
No
expert
or
counsel named
in
this
prospectus
as
having
prepared
or
certified
any part of
this
prospectus or having
given
an
opinion
upon
the
validity of the
securities
being
registered
or upon
other
legal
matters
in
connection
with the registration
or offering of
the securities was employed
on a
contingency
basis,
or
had,
or
is to receive,
in
connection
with the offering,
a
substantial
interest,
direct or
indirect, in the registrant.
Nor
was
any
such
person
connected
with
the
registrant
as
a
promoter, managing
or
principal
underwriter, voting trustee,
director,
officer,
or
employee.
PART
II
INFORMATION
NOT
REQUIRED
IN PROSPECTUS
Item
14.
Other Expenses
of
Issuance
and
Distribution
The
estimated expenses
payable by the
registrant
in
connection
with
the
issuance and
distribution
of
the securities being registered
are as
follows:
|
SEC Registration Fee
|
|
$
|
395
|
|
|
Legal Fees and Expenses*
|
|
$
|
73,000
|
|
|
Accounting Fees and Expenses*
|
|
|
5,000
|
|
|
Miscellaneous Fees and Expenses*
|
|
$
|
9,500
|
|
|
TOTAL:
|
|
$
|
87,895
|
|
|
*
|
|
Estimated
solely
for
the
purposes of
this Item. Actual expenses
may
vary.
|
Item
15.
Indemnification
of
Directors
and Officers
We
are
incorporated in Delaware
and consequently are subject
to
the Delaware
General Corporation
Law
(the
“DGCL”).
Section 145 of the DGCL provides a
detailed
statutory
framework covering indemnification
of
directors and
officers who have
been or are
threatened
to
be or
have
been
made defendants in
legal proceedings
by reason of
their service
as directors or
officers.
Our bylaws
provide,
in effect, that
we
shall
indemnify
our
directors
and officers to the
maximum
extent permitted
by
Delaware law. Article
V,
Sections
1 through 9 of
our bylaws
provide
as
follows:
“Section
1.
Actions
other
than
by
or
in the Right
of
the Corporation.
The
corporation shall
indemnify
any
person
who was
or
is
a party or
is threatened to
be
made
a party
to
any threatened,
pending
or
completed action, suit
or
proceeding, whether
civil, criminal, administrative
or
investigative
(other
than
an
action
by
or
in
the
right
of
the
corporation) by reason of
the fact
that he
is
or
was
a director,
officer,
employee
or
agent
of
the
corporation, or
is or
was
serving
at the
request
of
the
corporation as a director,
officer, employee
or
agent
of
another
corporation,
partnership,
joint
venture, trust
or
other enterprise,
against
expenses (including
attorneys’ fees), judgments, fines and amounts
paid
in settlement
actually and reasonably
incurred
by
him
in
connection
with such action, suit
or proceeding
if he acted in
good
faith
and
in
a
manner
he
reasonably
believed to
be
in
or
not
opposed
to
the
best
interests
of the corporation,
and,
with
respect
to
any criminal action
or
proceedings, had no
reasonable
cause to
believe
his conduct
was unlawful.
The
termination
of any
action, suit
or proceeding by
judgment,
order,
settlement, conviction,
or upon a plea of
NOLO
CONTENDERE or
its equivalent, shall not,
of
itself,
create
a
presumption that
the person did
not
act
in
good
faith and
in
a
manner which he
reasonably
believed
to
be
in
or
not
opposed
to
the
best
interests
of the corporation,
and,
with
respect
to
any criminal
action
or
proceeding, had reasonable cause to believe
that
his
conduct
was unlawful.
Section
2.
Actions
by
or
in
the
Right
of the
Corporation.
The
corporation shall
indemnify any person
who
was
or
is
a party or
is threatened to
be
made
a party
to
any threatened,
pending or
completed action
or
suit
by
or
in
the
right
of
the
corporation
to
procure a
judgment in his favor
by reason of
the
fact
that he is
or
was
a director,
officer, employee
or
agent
of
the
corporation, or
is
or
was
serving
at
the
request of the corporation as a director,
officer,
employee
or
agent
of another
corporation,
partnership,
joint
venture, trust
or
other
enterprise against
expenses
(including attorney’s fees)
actually and
reasonably
incurred
by
him
in
connection with the
defense or
settlement
of
such
action or
suit
if
he acted
in
good
faith
and
in
a
manner
he
reasonably believed
to
be
in
or
not
opposed
to
the
best
interests
of the corporation
and
except that no indemnification shall
be
made in
respect of any
claim,
issue
or
matter
as
to which such
person
shall have
been adjudged
to
be
liable unless and
only
to
the
extent
that the Court of Chancery of the
State
of
Delaware
or
the court in which
such action or
suit was brought shall
determine upon
application
that, despite the
adjudication of liability
but
in
view
of
all
the
circumstances
for the case,
such person
is fairly
and reasonably
entitled
to indemnity for such
expenses
which the Court
of Chancery
of
the
State of
Delaware
or
such
other court shall
deem proper.
Section
3.
Success
on the
Merits.
To
the extent
that any person
described
in Section
1 or 2 of
this Article
V
has
been
successful
on
the
merits
or otherwise
in
defense of any
action,
suit
or proceeding
referred to in said Sections,
or
in
defense of any
claim,
issue or
matter
therein, he shall
be
indemnified against expenses
(including
attorneys’
fees)
actually and reasonably
incurred
by
him
in
connection therewith.
Section
4.
Specific Authorization. Any indemnification
under
Section
1 or 2 of
this
Article
V
(unless
ordered by a
court) shall
be
made
by
the corporation
only
as
authorized in the specific case
upon a determination that
indemnification
of any person described
in
said
Sections is
proper
in the circumstances
because he
has
met
the
applicable
standard
of
conduct set forth
in
said Sections.
Such
determination shall
be
made
(1) by
the
Board of
Directors
by a
majority vote
of a quorum
consisting
of directors
who
were not parties to
such action, suit
or
proceeding,
or
(2)
if such
a quorum
is not obtainable,
or
even
if
obtainable
a quorum of
disinterested
directors
so directs,
by
independent
legal counsel
in
a
written opinion,
or
(3)
by
the stockholders to the
corporation.
Section
5.
Advance
Payment.
Expenses
incurred in defending
a
civil
or
criminal
action, suit
or proceeding
may
be
paid by
the
corporation
in
advance of
the
final
disposition of
such action, suit
or proceeding upon receipt of an
undertaking
by or on behalf of
any
person described
in said Section to
repay
such
amount
if
it
shall
ultimately be
determined
that
he
is
not entitled to indemnification
by
the corporation as
authorized in this Article
V.
Section
6.
Non-Exclusivity. The indemnification
and
advancement
of expenses provided
by,
or
granted pursuant
to,
the other Sections
of this
Article
V
shall not
be
deemed exclusive
of
any
other
rights
to which those
provided
indemnification
or
advancement
of expenses
may
be
entitled under
any
By-law,
agreement,
vote
of
stockholders
or
disinterested
directors or
otherwise,
both as
to action
in
his
official
capacity and as
to action in another
capacity
while
holding such office.
Section
7.
Insurance.
The
Board
of
Directors may
authorize, by a
vote
of the
majority
of
the
full
board,
the
corporation
to purchase and
maintain insurance
on behalf of any person
who is
or
was
a director,
officer, employee
or
agent
of
the
corporation, or
is
or
was
serving at
the
request of the corporation
as a director,
officer, employee
or
agent
of
another
corporation, partnership,
joint
venture,
trust
or
other enterprise against
any
liability asserted
against
him and
incurred
by him
in
any
such
capacity,
or
arising out
of his
status
as
such, whether
or
not the corporation
would have
the
power to indemnify
him
against
such liability under
the provisions
of
this Article
V.
Section
8.
Continuation
of Indemnification
and
Advancement
of
Expenses.
The
indemnification
and advancement
of
expenses
provided
by,
or
granted pursuant
to,
this Article
V
shall continue
as
to
a person
who has
ceased
to
be a
director,
officer, employee
or
agent
and
shall
inure
to the benefit
of
the
heirs,
executors and
administrators of
such
a
person.
Section
9.
Intent
of
Article. The intent
of
this
Article
V
is to
provide
for indemnification
and
advancement
of
expenses to the fullest
extent permitted
by
Section
145
of the
General
Corporation Law of
Delaware.
To
the extent
that
such
Section or any successor
action may
be
amended
or
supplemented
from
time to time, this
Article
V
shall
be
amended automatically
and
construed
so as
to permit indemnification
and advancement
of expenses
to the fullest extent
from
time
to time
permitted by
law.”
Reference
is made to Article Eighth
of our
certificate
of
incorporation,
which
provides
as
follows:
“EIGHTH.
A director of
the
corporation
shall not
be personally
liable to the
corporation or
its
stockholders
for
monetary damages for
breach of fiduciary duty as a director,
except for liability (i) for
any breach of
the
director’s
duty
of
loyalty to
the corporation or
its
stockholders, (ii) for
acts
or
omissions not in
good
faith
or
which involve intentional misconduct
or a
knowing
violation
of
law, (iii)
under
Section
174 of
the General
Corporation Law
of
Delaware,
or
(iv)
for any
transaction
from
which
the
director
derived
an improper
personal benefit.
In addition
to
and not in limitation
of the
foregoing,
a director of
the
corporation
shall
not
be
liable to the
corporation or
its
stockholders for monetary damages
for breach of fiduciary duty as a director
to the
fullest extent
provided by the General Corporation Law
of
Delaware
as the
same
may
hereafter
be
amended.”
Reference
is
also
made to
Section 145 of
the
DGCL, which
provides as
follows:
“(a)
A
corporation
shall have
power
to indemnify
any person who
was
or
is
a party or
is
threatened to
be
made
a party
to
any
threatened,
pending
or
completed
action, suit
or
proceeding,
whether
civil, criminal, administrative
or
investigative
(other than
an action
by
or
in the
right of the corporation) by reason of the fact
that
the person
is
or
was
a director,
officer, employee
or
agent
of
the
corporation, or
is
or
was
serving
at the
request
of
the
corporation as a
director, officer, employee
or
agent
of
another
corporation,
partnership,
joint venture, trust
or
other enterprise, against
expenses
(including
attorneys’ fees), judgments, fines
and
amounts
paid
in
settlement
actually and reasonably
incurred
by
the
person
in connection with such
action,
suit
or proceeding
if
the person
acted in
good
faith and
in
a
manner the
person reasonably
believed
to
be
in
or
not
opposed
to
the
best
interests
of the
corporation,
and, with
respect
to
any
criminal
action or
proceeding, had no reasonable cause to believe
the
person’s
conduct was unlawful.
The
termination
of
any
action, suit
or proceeding by
judgment,
order,
settlement, conviction,
or upon a plea of
NOLO
CONTENDERE or
its equivalent, shall not,
of
itself,
create
a
presumption that
the person did
not
act
in
good
faith and
in
a
manner which the
person reasonably
believed
to
be
in
or
not
opposed
to
the
best
interests
of the
corporation,
and, with
respect
to
any
criminal
action or
proceeding, had reasonable cause to believe
that the
person’s
conduct was unlawful.
(b)
A
corporation
shall have
power
to indemnify
any
person who
was
or
is
a party or
is
threatened to
be
made
a party
to
any
threatened, pending
or
completed
action or
suit
by
or
in
the
right
of the corporation
to procure
a
judgment in
its
favor
by
reason of the
fact
that the person
is
or
was
a director,
officer, employee
or
agent
of the
corporation,
or
is
or
was
serving at
the
request
of
the
corporation, as
a director,
officer, employee
or
agent
of
another
corporation,
partnership,
joint
venture,
trust or
other enterprise against
expenses
(including
attorneys’ fees) actually and
reasonably
incurred
by
the
person
in
connection
with the
defense or
settlement
of
such
action or
suit
if
the
person
acted in
good
faith
and in
a
manner he
reasonably
believed
to
be
in
or
not opposed to the
best
interests
of the corporation
and except
that no indemnification shall
be
made in
respect of any
claim,
issue
or
matter
as
to which such
person
shall have
been adjudged
to
be
liable to the corporation unless
and only
to
the extent
that
the Court
of
Chancery
or
the court in which such action
or
suit was
brought shall determine
upon
application that, despite
the adjudication of
liability but in view
of
all
the
circumstances
of
the case,
such person
is
fairly
and
reasonably
entitled to indemnity for such
expenses
which
the
Court
of Chancery or
such other
court shall
deem proper.
(c)
To
the extent that
a present or
former
director or
officer
of a corporation
has
been
successful
on the
merits
or
otherwise
in
defense
of any action,
suit
or
proceeding
referred
to in subsections
(a)
and
(b) of
this section,
or
in defense
of any
claim, issue
or
matter therein,
such person
shall
be
indemnified against
expenses
(including attorneys’ fees) actually and
reasonably incurred by
such
person
in
connection
therewith.
(d)
Any
indemnification under subsections
(a)
and
(b)
of
this
section
(unless
ordered by a
court)
shall
be
made
by
the
corporation only as
authorized
in the specific
case upon a
determination
that
indemnification
of the director,
officer, employee
or
agent
is
proper
in the circumstances
because the person has
met
the applicable
standard
of
conduct
set
forth
in
subsections
(a)
and
(b) of
this
section.
Such
determination shall
be
made
with
respect
to
a person
who
is
a director or
officer
at
the
time
of such
determination
(1) by a
majority
vote
of
the
directors
who
are
not parties to such action, suit
or
proceeding,
even
though
less
than a
quorum,
or (2) by a
committee
of
such
directors designated by
majority vote
of
such
directors, even though
less
than
a
quorum,
or (3)
if there
are
no such directors,
or
if such
directors
so direct,
by
independent legal counsel
in
a
written
opinion, or (4) by
the
stockholders.
(e)
Expenses
(including attorneys’ fees) incurred
by
an
officer
or director
in defending
any
civil,
administrative
or
investigative action, suit
or
proceeding, may
be paid by
the
corporation
in advance
of the
final
disposition
of
such action, suit
or
proceeding
upon
receipt of an
undertaking
by
or on behalf
of such director or
officer to
repay such
amount
if
it shall ultimately
be
determined
that such
person
is not entitled to
be
indemnified
by the corporation as
authorized in
this
section.
Such expenses
(including
attorneys’ fees) incurred
by
former
directors
and officers
or
other employees
and
agents
may be
so
paid upon
such
terms
and
conditions,
if
any,
as the corporation
deems
appropriate.
(f) The
indemnification
and
advancement
of
expenses
provided
by,
or
granted
pursuant
to,
the other subsections
of
this
section shall
not
be
deemed
exclusive
of any other
rights to which
those
seeking
indemnification or
advancement
of expenses
may
be
entitled under
any
bylaw, agreement,
vote
of
stockholders
or
disinterested
directors
or
otherwise,
both as
to action
in
such
person’s
official
capacity
and
as
to action in another
capacity
while
holding
such office.
(g) A
corporation
shall have
power
to purchase and
maintain insurance
on behalf
of
any person
who
is
or
was
a director,
officer,
employee
or
agent
of the corporation, or
is
or
was
serving at
the request
of the corporation as a director,
officer, employee
or
agent
of
another
corporation,
partnership,
joint
venture,
trust or
other enterprise
against
any
liability asserted against such
person
and
incurred
by such person
in
any
such capacity,
or
arising out
of
such
person’s
status
as
such, whether
or
not the
corporation
would
have
the
power to indemnify
him
against
such liability under this section.
(h)
For
purposes of
this section,
references
to “the
corporation”
shall
include,
in
addition
to
the
resulting corporation,
any
constituent
corporation
(including
any
constituent
of
a
constituent)
absorbed
in
a
consolidation
or
merger which, if
its
separate existence had
continued, would have
had
power and
authority
to
indemnify
its
directors, officers,
and
employees
or
agents, so
that
any
person
who is
or
was
a director,
officer,
employee
or
agent
of such
constituent
corporation,
or
is
or
was
serving
at the
request
of
such
constituent
corporation as a
director, officer, employee
or
agent
of
another
corporation,
partnership,
joint venture, trust
or
other enterprise, shall stand in
the
same
position under this
section
with
respect
to
the
resulting or
surviving
corporation as
such
person
would have with
respect
to such
constituent
corporation
if its separate existence had
continued.
(i)
For
purposes of
this section,
references
to “other
enterprises” shall
include
employee benefit
plans;
references
to “fines” shall
include any excise
taxes assessed
on a person
with
respect
to
any
employee
benefit
plans; and references to “serving
at the
request
of the corporation”
shall include
any
service
as a director,
officer, employee
or
agent
of
the
corporation
which
imposes
duties
on, or
involves
services
by, such
director,
officer, employee
or
agent with
respect
to
an
employee
benefit
plan, its participants
or
beneficiaries;
and
a person
who
acted
in
good
faith and
in
a
manner
he
reasonably
believed to
be
in
the
interest
of the
participants
and beneficiaries
of
an
employee benefit
plan
shall
be deemed
to have acted
in
a
manner “not
opposed
to
the
best
interests
of
the
corporation”
as
referred to in this section.
(j)
The
indemnification
and
advancement
of
expenses
provided
by,
or
granted
pursuant
to,
this section shall, unless otherwise
provided
when
authorized
or
ratified, continue
as
to
a person
who
has
ceased to
be a director,
officer, employee
or
agent
and
shall
inure
to the benefit
of
the
heirs, executors and
administrators
of
such
a
person.
(k) The
Court
of Chancery
is hereby vested with exclusive
jurisdiction to hear and determine all actions for advancement
of expenses or
indemnification
brought
under
this
section or
under
any
bylaw, agreement, vote
of
stockholders
or
disinterested directors,
or
otherwise.
The
Court
of Chancery may summarily
determine
a
corporation’s obligation to advance
expenses
(including
attorneys’ fees).”
As
permitted
by
the
bylaws,
we
also presently maintain
a policy of
directors’
and officers’ liability insurance.
We
have also
entered
into
an
indemnification
agreement
(the
“Indemnification
Agreement”)
with
each of
our
directors
and executive
officers
which is
intended
to complement
the
indemnity and
protection available under our certificate
of incorporation
and
bylaws
and
the directors’
and
officers’
liability
insurance
policy
maintained
by
us,
and
to
provide
for indemnification
of directors
and officers to the fullest extent
permitted by applicable
law.
While
our bylaws
provide,
in effect, that
we
shall indemnify
directors
and officers
to the
maximum extent
permitted
by Delaware
law,
the Indemnification
Agreement
provides a
number
of procedures,
presumptions and remedies used
in
the determination
of the
right
of the director or
officer to indemnification.
These procedures,
presumptions
and
remedies substantially
broaden
the
indemnity rights of directors
and officers beyond
those expressly
contained
in
the bylaws and
in Section
145 of
the
DGCL.
The
Indemnification Agreement
provides
that
we will
pay
certain
expenses
incurred by a director or
officer in connection with
any
threatened,
pending
or
completed action, suit, arbitration
or
proceeding,
whether civil, criminal, administrative,
or
investigative, and specifically
including
actions
by
or
in
our name (“derivative suits”), where the individual’s
involvement
is
by
reason of
the fact
that
he
or
she is
or
was
a director or
officer. Such amounts
include
attorneys’
fees
and
other
expenses
customarily
incurred in
connection
with legal
proceedings
and,
in
the
case
of
proceedings
other than derivative suits, judgments, fines
and
amounts
paid
in
settlement. Indemnification would
be
available for actions, suits, arbitrations
or
proceedings
commenced
after the effective
date of
the Indemnification Agreement.
A director or
officer
will
not receive indemnification if
the director or
officer is found not to have acted
in
good
faith and
in
a
manner
he
reasonably
believed to
be
in
or
not
opposed
to
our
best
interests.
The
Indemnification Agreement further
provides
that
if
an action
against
an
indemnified
party
is dismissed,
with
or
without
prejudice,
the defense is deemed to have
been
successful
and
indemnification
is
required
to
be
made.
The
Indemnification Agreement
also provides
that litigation
expenses
must
be
advanced within
twenty days of
any
request, against
an
undertaking
to
repay
if
the
payee
is
ultimately
determined not
be
entitled
to indemnification.
A determination of
entitlement must
be
made
within
sixty days of
an
indemnification
request, and payment is to
be
made within
ten days
after
a
favorable determination. (Otherwise
a determination
in
favor of
the indemnified
party
is deemed
to have
been
made.)
If
there is
a
change in
control of
the
Company (as
defined in
the
Indemnification
Agreement), the indemnified
party
is
presumed to be
entitled
to indemnification (although
the Company
may
overcome
this
presumption), and
the
indemnified
party may require
that
independent
counsel
(as
defined in
the
Indemnification Agreement) make
the determination of
entitlement
and
may choose
such
counsel, subject
to
objection
by
us
on
limited grounds specified in
the
Indemnification
Agreement.
If a determination of
entitlement is made,
we
are
bound, but if
the
indemnified
party
is denied
indemnification
pursuant to
the
terms
of
the Indemnification Agreement,
he
or
she is entitled to seek
a de novo
determination
from a court. The
indemnified
party
is
entitled
to enforce the Indemnification Agreement in court
and
we
are
precluded from challenging the
validity
of
the
procedures
and presumptions contained
in
the Indemnification Agreement.
Item
16.
Exhibits
|
Exhibit
Number
|
|
Exhibit
Description
|
|
5.1
|
|
Opinion
of Pierce
Atwood
LLP
|
|
10.1
|
|
Securities
Purchase Agreement (incorporated
by
reference
to Exhibit
10.1 of
the registrant’s Current
Report
on
Form
8-K filed
on October 21, 2016
(file
no.
1-12934))
|
|
10.2
|
|
Registration
Rights Agreement (incorporated
by
reference
to Exhibit
10.2 of
the registrant’s Current
Report
on
Form
8-K filed
on October 21, 2016
(file
no.
1-12934))
|
|
23.1
|
|
Consent
of Pierce
Atwood
LLP
(included in Exhibit
5.1).
|
|
23.2
|
|
Consent
of Baker
Newman
&
Noyes LLC.
|
|
24.1
|
|
Power
of
Attorney
(See signature page).
|
Item
17.
Undertakings
(a)
The
undersigned registrant
hereby
undertakes:
(1)
To
file,
during
any
period
in
which offers
or
sales
are
being made,
a post-effective
amendment to this registration statement:
(i)
To
include
any
prospectus
required
by
section 10(a)(3) of
the Securities
Act
of
1933.
(ii)
To
reflect in the
prospectus
any
facts or
events arising after
the
effective
date of the
Registration Statement
(or
the
most recent post-effective amendment thereof) which, individually
or
in the
aggregate,
represent
a
fundamental change in
the
information
set forth
in
the
Registration Statement.
Notwithstanding
the foregoing,
any
increase
or decrease
in volume
of
securities offered
(if
the total
dollar
value
of
securities
offered would not
exceed
that which was registered) and
any
deviation
from the low or
high
end of
the
estimated maximum offering range
may be
reflected
in
the
form of
prospectus
filed with the
Commission
pursuant to Rule
424(b)
if,
in
the
aggregate,
the changes
in
volume and
price
represent
no more
than a 20%
change
in
the
maximum aggregate offering
price
set forth in
the
“Calculation
of
Registration Fee” table
in
the effective registration statement.
(iii)
To
include
any
material information
with
respect
to the
plan of
distribution
not previously disclosed in
the
registration statement
or any
material
change to such
information
in
the
registration
statement.
Provided,
however,
that paragraphs (a)(1)(i),
(a)(1)(ii)
and (a)(1)(iii)
above do
not
apply
if
the
registration
statement is
on Form S-3 or Form F-3
and
the information required to
be
included
in
a
post-effective amendment
by
those
paragraphs is contained in
reports
filed with
or
furnished to the Commission
by
the Registrant pursuant to Section
13
or
15(d) of
the Securities
Exchange
Act
of 1934
that
are incorporated by
reference
in
the registration
statement,
or
is
contained
in
a form
of
prospectus
filed pursuant to Rule
424(b)
that is
part of
the
registration statement.
(2)
That,
for the
purpose of
determining
any
liability
under the Securities Act
of
1933, each
such post-effective amendment
shall be
deemed to
be a
new
registration
statement
relating
to
the
securities
offered therein, and the offering
of
such securities
at
that
time shall
be
deemed to
be
the initial
bona
fide offering thereof.
(3)
To
remove
from
registration
by
means
of
a
post-effective
amendment any of the
securities
being
registered
which remain
unsold at
the termination
of
the offering.
(4)
That,
for the
purpose of
determining liability
under the Securities Act
of
1933
to any
purchaser:
(i)
If
the registrant is
relying
on
Rule
430B:
(A)
Each
prospectus filed
by the
registrant
pursuant
to
Rule
424(b)(3)
shall
be
deemed to
be part of
the registration
statement
as of the date
the filed prospectus was
deemed part of
and
included in the registration statement; and
(B)
Each
prospectus required to
be
filed
pursuant to
Rule 424(b)(2),
(b)(5)
or
(b)(7)
as part of a
registration statement in reliance
on
Rule
430B
relating to
an offering
made pursuant
to Rule
415(a)(1)(i),
(vii)
or
(x) for
the
purpose of
providing
the information
required
by section 10(a) of
the Securities Act
of 1933
shall
be
deemed to
be part of
and included
in
the
registration statement
as
of
the earlier
of
the
date such
form
of
prospectus
is
first
used
after effectiveness
or the date of
the
first contract
of
sale
of
securities
in
the
offering
described
in
the
prospectus.
As provided
in Rule
430B,
for liability
purposes of
the
issuer
and
any person
that is
at that date
an
underwriter, such
date
shall
be
deemed
to
be a new
effective
date
of
the registration statement relating to
the
securities
in
the
registration statement to which
that prospectus relates, and the
offering
of
such
securities
at that
time shall
be
deemed
to
be
the initial
bona
fide offering
thereof.
Provided,
however,
that
no
statement made
in
a
registration statement
or
prospectus that is
part of the
registration
statement
or
made in
a document incorporated
or
deemed incorporated
by
reference into the registration statement
or
prospectus
that is
part of the
registration statement
will,
as
to
a
purchaser
with
a
time
of
contract
of
sale
prior
to such effective
date, supersede or
modify
any
statement that
was made in the registration statement
or
prospectus that was
part
of
the registration statement
or
made in
any
such
document
immediately
prior
to such effective
date; or
(ii)
If
the registrant
is
subject
to
Rule
430C, each prospectus
filed pursuant to Rule
424(b) as part of a
registration
statement relating to
an
offering, other than registration statements
relying
on
Rule
430B or
other than prospectuses filed
in reliance
on
Rule
430A,
shall
be
deemed to
be part of
and included in the registration
statement
as of
the
date
it is first
used after effectiveness.
Provided,
however, that no statement made in
a
registration
statement
or
prospectus that is
part of
the
registration statement
or
made in
a
document
incorporated
or
deemed incorporated
by
reference
into
the
registration statement
or
prospectus
that is
part of the
registration statement will,
as
to
a
purchaser with
a
time
of
contract
of
sale
prior
to such first use, supersede
or modify any
statement that was made in
the
registration
statement or
prospectus that was
part of
the
registration statement
or
made in
any such
document
immediately prior
to such
date of
first
use.
(5)
That,
for the
purpose of
determining
liability
of
the registrant
under
the Securities Act
of 1933
to
any
purchaser
in
the
initial
distribution
of the
securities.
The
undersigned registrant undertakes that
in
a primary
offering
of
securities
of
the
undersigned registrant pursuant to this registration statement, regardless
of the
underwriting method used to sell the securities to
the
purchaser,
if the securities
are
offered
or
sold
to such purchaser
by
means
of any
of the
following communications, the
undersigned
registrant
will be a
seller to
the
purchaser and
will be considered
to offer
or
sell
such securities to such purchaser:
(i)
Any
preliminary prospectus or prospectus
of
the
undersigned registrant
relating
to
the
offering required to
be
filed pursuant
to Rule
424;
(ii)
Any free writing prospectus relating to
the
offering
prepared by or on behalf of
the undersigned registrant
or
used
or
referred
to
by
the
undersigned registrant;
(iii)
The portion of any
other free writing
prospectus
relating
to the offering containing material information
about the
undersigned registrant
or
its securities
provided by or on
behalf
of
the undersigned registrant; and
registrant to the purchaser.
(iv)
Any
other
communication that
is
an
offer
in
the
offering
made
by
the
undersigned
(b)
The
undersigned registrant
hereby
undertakes
that, for
purposes of
determining
any
liability
under
the Securities Act
of
1933,
each
filing
of the
registrant’s annual
report
pursuant to
Section 13(a) or
Section
15(d) of
the Securities Exchange Act
of
1934
(and, where
applicable, each
filing
of
an
employee
benefit
plan’s
annual report
pursuant to Section
15(d) of
the
Securities Exchange Act
of
1934)
that
is incorporated
by
reference
in
the
registration statement shall
be
deemed
to
be a new
registration statement
relating
to
the
securities
offered
therein,
and the
offering
of
such securities
at that
time shall
be
deemed to
be
the
initial
bona
fide offering thereof.
(c)
The
undersigned registrant
hereby
undertakes
that, for
purposes of
determining
any
liability
under
the
Securities Act
of 1933,
the information omitted
from
the
form
of prospectus
filed
as part of
this registration
statement in reliance
upon
Rule
430A
and
contained in
a form of
prospectus filed
by the
registrant
pursuant to Rule
424(b)(1) or
(4)
or 497(h)
under
the Securities Act
of 1933
shall
be
deemed
to
be part of
this registration statement
as of the
time
it was
declared
effective.
(d)
The
undersigned registrant
hereby
undertakes
that,
for
the
purpose of determining
any
liability under the
Securities Act
of
1933, each
post-effective amendment
that
contains
a form of prospectus
shall
be
deemed
to
be a
new
registration statement
relating
to the securities offered
therein, and
the
offering
of
such securities
at
that time shall
be
deemed to
be
the
initial
bona fide
offering thereof.
Insofar
as
indemnification for liabilities
arising under
the Securities Act
of 1933
may
be
permitted
to directors, officers
and
controlling
persons of the
registrant
pursuant
to
the provisions
described
in
Item 15 above, or
otherwise, the registrant
has
been
advised
that
in
the
opinion of
the Securities
and
Exchange
Commission such
indemnification
is
against
public
policy as
expressed in the Act and is, therefore, unenforceable.
In
the event that
a claim
for indemnification
against such liabilities (other than the payment
by
the
registrant
of
expenses incurred
or paid by a director,
officer
or
controlling
person of
the registrant
in
the
successful
defense of any
action,
suit
or
proceeding) is asserted
by
such
director,
officer or controlling
person
in
connection
with the securities being
registered, the registrant
will,
unless in the
opinion of
its
counsel
the matter
has been
settled
by
controlling precedent, submit to
a
court
of
appropriate jurisdiction the question whether such indemnification
by
it is
against
public
policy
as
expressed
in
the
Act and
will be
governed
by the
final
adjudication of such
issue.
SIGNATURES
Pursuant
to the requirements
of
the Securities Act
of 1933, as
amended, the registrant certifies
that
it
has reasonable grounds to believe that it meets all
of
the
requirements
for filing
on Form S-3
and has
duly
caused
this Pre-
Effective Amendment
No. 1
to the Registration Statement to
be
signed
on
its
behalf by the
undersigned, thereunto
duly
authorized, in
the City
of
Portland, State
of Maine on December 5, 2016.
|
|
IMMUCELL CORPORATION
|
|
|
|
|
|
By:
|
/s/ Michael F. Brigham
|
|
|
|
Michael F. Brigham
President,
Chief Executive Officer and
Treasurer
|
|
Dated:
December
5, 2016
|
By:
|
/s/ Michael F. Brigham
|
|
|
|
/s/ Michael F. Brigham
|
|
|
|
President,
Chief
Executive Officer,
Treasurer
and
Director
|
|
|
|
(Principal
Executive
Officer
and
Principal
Financial
and
Accounting Officer)
|
|
|
|
|
|
Dated:
December
5, 2016
|
By:
|
*
|
|
|
|
Joseph H. Crabb,
Vice President and
Chief
Scientific
Officer and Director
|
|
|
|
|
|
Dated:
December
5, 2016
|
By:
|
*
|
|
|
|
David S. Cunningham,
Director
|
|
|
|
|
|
Dated:
December
5, 2016
|
By:
|
*
|
|
|
|
Linda Rhodes, Director
|
|
|
|
|
|
Dated:
December
5, 2016
|
By:
|
*
|
|
|
|
Jonathan
E.
Rothschild,
Director
|
|
|
|
|
|
|
By:
|
*
|
|
Dated:
December
5, 2016
|
|
David S. Tomsche, Director
|
|
|
|
|
|
|
By:
|
*
|
|
Dated:
December
5, 2016
|
|
Paul
R. Wainman, Director
|
|
|
|
|
|
*By:
/s/
Michael
F. Brigham
|
|
|
|
Michael
F. Brigham, Attorney-in-Fact
|
|
|
ImmuCell
Corporation
EXHIBIT
INDEX
|
Exhibit 5.1
|
|
Opinion of Pierce Atwood LLP
|
|
|
|
|
|
Exhibit 23.1
|
|
Consent of Pierce Atwood LLP (included in Exhibit 5.1)
|
|
|
|
|
|
Exhibit 23.2
|
|
Consent of Baker Newman & Noyes LLC
|
ImmuCell (NASDAQ:ICCC)
Historical Stock Chart
From Mar 2024 to Apr 2024
ImmuCell (NASDAQ:ICCC)
Historical Stock Chart
From Apr 2023 to Apr 2024