Applied DNA Receives Kosher Certification from Orthodox Union for Qualifying Products
February 20 2020 - 9:00AM
Business Wire
Products within the SigNature® and CertainT®
Brand Certified in Strict Compliance with OU Standards as
Ingredients for Dietary Supplements, Pharmaceuticals and Food
Products
Applied DNA Sciences, Inc. (“Applied DNA”, the “Company”,
NASDAQ: APDN), announced receipt of certification from the Orthodox
Union (OU®) for products within the SigNature and CertainT line
following a thorough evaluation of the Company’s products and
manufacturing processes. The Pareve kosher designation will first
be used for the Nutrition21 Nitrosigine® sports supplement product
recently announced. The Company’s products and services, used by
its customers to manage traceability and authenticity in the name
of brand assurance, are also suitable for related markets such as
food and pharmaceuticals.
The OU (Orthodox Union) Kosher is the world’s largest and most
widely recognized international kosher certification agency,
certifying over 800,000 products produced in more than 9,011 plants
located in 104 countries around the world. Over $150 billion of
kosher certified products are consumed annually, with the OU kosher
symbol appearing on close to 70% of America’s kosher certified
foods.
“As we grow our presence in food, pharmaceutical and dietary
supplements markets, we are pleased to be supportive of the
business imperatives critical to meet market requirements that
maximize adoption. We are proud to have the OU certification on our
qualifying products. OU maintains strict guidelines for
certification and ongoing manufacturing, marketing and distribution
of products, making Applied DNA’s mission for enabling our
customers a platform for proof of authenticity and traceability
nicely complementary for OU-certified products,” said Dr. James
Hayward, president and CEO, Applied DNA.
The Company’s qualifying CertainT and SigNature molecular tags
enable companies and their supply chains to designate a unique
molecular identity to products with meaning specific to the tagged
material: for example, geographic facility location, brand, product
line or date of manufacture. This tag can be blended into
ingredients and packaging inks and varnishes in the pharmaceutical,
nutraceutical, and dietary supplements industry. The tag enables
its carrier to be verified for authenticity and blending detection,
either at the customer or in the Applied DNA laboratory, depending
on application requirements.
“Applied DNA Sciences is leading the way in supply chain
authentication,” commented Rabbi Menachem Genack, CEO, OU Kosher,
“and we are excited to partner with them as they expand into the
market of dietary supplements, food ingredients and nutraceuticals.
In turn,” he observed, “their molecular tag offers our supervision
program an additional instrument for validating the integrity of
our certified products.”
About Applied DNA Sciences
Applied DNA is a provider of molecular technologies that enable
supply chain security, anti-counterfeiting and anti-theft
technology, product genotyping and pre-clinical nucleic acid-based
therapeutic drug candidates.
Applied DNA makes life real and safe by providing innovative,
molecular-based technology solutions and services that can help
protect products, brands, entire supply chains, and intellectual
property of companies, governments and consumers from theft,
counterfeiting, fraud and diversion.
Visit adnas.com for more information. Follow us on Twitter and
LinkedIn. Join our mailing list.
Common stock listed on NASDAQ under the symbol APDN.
Forward-Looking Statements
The statements made by Applied DNA in this press release may be
“forward-looking” in nature within the meaning of the Private
Securities Litigation Reform Act of 1995. Forward-looking
statements describe Applied DNA’s future plans, projections,
strategies and expectations, and are based on assumptions and
involve a number of risks and uncertainties, many of which are
beyond the control of Applied DNA. Actual results could differ
materially from those projected due to our ability to successfully
enter into commercial contracts for the implementation of our
CertainT® platform, the possibility of failure to make timely
payment on its outstanding secured convertible notes and resulting
enforcement by noteholders of remedies on collateral which includes
substantially all of Applied DNA’s assets, the Company’s history of
net losses, limited financial resources, limited market acceptance
and various other factors detailed from time to time in Applied
DNA’s SEC reports and filings, including our Annual Report on Form
10-K filed on December 12, 2019 and our subsequent quarterly report
on Form 10-Q filed on February 6, 2020, and other reports we file
with the SEC, which are available at www.sec.gov. Applied DNA
undertakes no obligation to update publicly any forward-looking
statements to reflect new information, events or circumstances
after the date hereof to reflect the occurrence of unanticipated
events, unless otherwise required by law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200220005089/en/
investor contact: Sanjay M. Hurry, LHA Investor
Relations, 212-838-3777, shurry@lhai.com program contacts:
Judy Murrah, Chief Information Officer, 631-240-8819,
judy.murrah@adnas.com web: www.adnas.com twitter:
@APDN
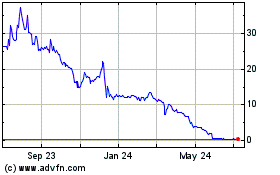
Applied DNA Sciences (NASDAQ:APDN)
Historical Stock Chart
From Mar 2024 to Apr 2024
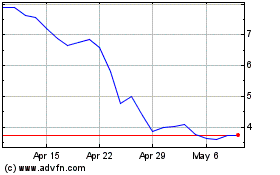
Applied DNA Sciences (NASDAQ:APDN)
Historical Stock Chart
From Apr 2023 to Apr 2024