
PositiveID's ExcitePCR
FireflyDX Prototype Successfully Detects E. Coli in Less Than 30
Minutes
Detection at local processing or
restaurant level is key to preventing outbreaks
like romaine lettuce E. coli
and egg-borne salmonella
Delray Beach, FL --
May
01, 2018 --
InvestorsHub NewsWire -- PositiveID Corporation ("PositiveID" or
"Company") (OTC:PSIDD)
subsidiary
ExcitePCR today announced today that the
FireflyDX breadboard prototype pathogen detection system can
accurately detect the E. coli bacterium in less than 30
minutes. Further, the Company
commented on the
importance of its rapid, portable
pathogen
detection
products, currently under
development, to
prevent increasingly frequent foodborne disease
outbreaks.
In scenarios where
containment is key to preventing widespread accidental or
bioterror-driven health disasters,
immediate action can mean the difference between single
digit and hundreds of cases, as seen in recent
instances.
FireflyDX is designed to
detect E. coli and other pathogens at the point of
need,
eliminating the
delays that result from having to transport
samples for off-site lab testing
to receive
potentially
life-saving results.
The recent
multi-state
outbreak is the worst outbreak of E. coli since 2006 when illnesses
traced to spinach killed three people and sickened more than
270,
creating millions of dollars in legal
liability.
The ability to rapidly
and accurately diagnose pathogenic E. coli at the point of
need gives health care providers
information required for correct treatment protocols and
for public
information
campaigns.
Further, millions of
dollars in
legal awards are paid annually due to foodborne illness. Through
implementation of on-site detection at local food
processing facilities, legal liability for outbreaks can be
significantly
minimized. PositiveID believes that
completion and commercialization of
FireflyDX could be a solution that enables such
prevention.
FireflyDX
Evidence
The graph below shows FireflyDX
successful detection of E. coli using its own assay:
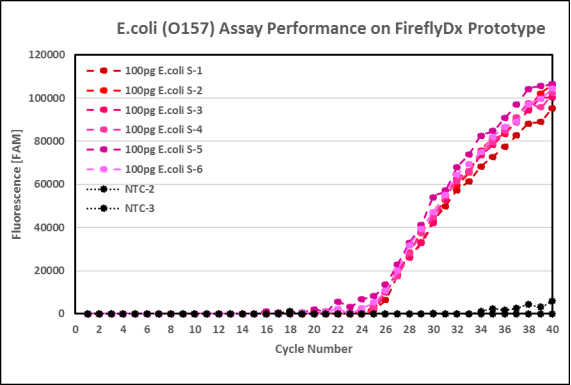
"FireflyDX is being developed
to prevent and more effectively respond to disease outbreaks and
minimize the spread of infection,"
commented
Lyle L.
Probst, ExcitePCR President and
CEO. "Use of FireflyDX at the farm,
food processing facility, or the restaurant level, can equip health
care professionals with an unprecedented level of information to stop such
food safety issues before they become a public health
concern."
E. coli is a bacterium commonly
found in the lower intestine of warm-blooded
animals. Most E. coli strains are harmless, but some
kinds, like E. coli O157:H7, can cause
serious disease in humans including bloody diarrhea and hemolytic
uremic syndrome, a type of kidney failure. Food contamination can occur
when food products come into contact with fecal material in soil,
water, or contaminated surfaces. This contamination can cause food
poisoning in humans and result in serious illness and costly
product recalls.
PositiveID expects that
FireflyDX vertical markets will include human clinical, such as
infectious disease, and hospital acquired infections, global food
and agricultural, and biothreat defense.
About PositiveID
Corporation
PositiveID Corporation
is a
holding
company with a
focus on life sciences tools and
diagnostics. PositiveID's ExcitePCR subsidiary
develops biological
detection and diagnostics systems, specializing in the development
of microfluidic systems for the automated preparation of and
performance of biological assays. PositiveID's Thermomedics subsidiary
markets the FDA-cleared Caregiver non-contact infrared
thermometer. PositiveID also is a minority
owner of E-N-G Mobile Systems, a leader in the
specialty vehicle
market. For
more information on PositiveID, please visit http://www.psidcorp.com, or connect with PositiveID
on Twitter, Facebook or LinkedIn.
On August 24, 2017, PositiveID
Corporation and its wholly-owned subsidiary PositiveID Diagnostics,
Inc. (collectively, the "Seller"), entered into an Asset Purchase
Agreement ("APA") with ExcitePCR Corporation. Pursuant to the APA,
at closing, the Seller will sell and deliver to ExcitePCR all
assets used in connection with the operation of the FireflyDX
technology. For more information on the APA, please read
PositiveID's Form 8-K filed on August 28, 2017, which can be
found here.
Statements about PositiveID's future expectations,
including the likelihood that the completion and
commercialization of FireflyDX could be a solution that
enables the ability to rapidly and accurately
diagnose pathogenic E. coli at the point of need
giving health care providers information required
for correct treatment protocols and for public information
campaigns, and enables on-site
detection at local food processing facilities to
minimize legal liability for outbreaks;
the likelihood that use of FireflyDX at the farm, food
processing facility, or the restaurant level, can equip health care
professionals with an unprecedented level of information to stop
such food safety issues before they become a public health
concern; the likelihood that FireflyDX
vertical markets will include human clinical, such as infectious
disease, and hospital acquired infections, global food and
agricultural, and biothreat defense; constitute
"forward-looking statements" within the meaning of Section 27A of
the Securities Act of 1933, Section 21E of the Securities Exchange
Act of 1934, and as that term is defined in the Private Litigation
Reform Act of 1995. Such forward-looking statements involve risks
and uncertainties and are subject to change at any time, and
PositiveID's actual results could differ materially from expected
results. These risks and uncertainties include, without limitation,
the Company's ability to raise capital; the Company's
ability to complete the testing and development of FireflyDX; the
Company's ability to commercialize FireflyDX; the Company's ability
to defend against litigation; the Company's ability to
close the APA with ExcitePCR; as well as other risks.
Additional information about these and other factors that could
affect the Company's business is set forth in the Company's various
filings with the Securities and Exchange Commission, including
those set forth in the Company's 10-K filed on April 2, 2018, and
10-Qs filed on November 13, 2017, August 14, 2017, and May 15,
2017, under the caption "Risk Factors." The Company undertakes no
obligation to update or release any revisions to these
forward-looking statements to reflect events or circumstances after
the date of this statement or to reflect the occurrence of
unanticipated events, except as required by law.
Contact:
PositiveID
Corporation
Allison Tomek
(561) 805-8044
atomek@psidcorp.com