Itonis Pharmaceuticals in Final Steps of NDC Registration With FDA for Emesyl Product
April 22 2014 - 9:00AM
Marketwired
Itonis Pharmaceuticals in Final Steps of NDC Registration With FDA
for Emesyl Product
LAGUNA HILLS, CA--(Marketwired - Apr 22, 2014) - Itonis, Inc.
(PINKSHEETS: ITNS) is pleased to share an update from its
manufacturer that the Company's Emesyl™ anti-nausea product is in
its final steps of completing the NDC registration with the Food
and Drug Administration ("FDA"), and is being readied for the first
production run. While the process has taken longer than
expected, the Company is pleased with recent progress to deliver
Emesyl™ to market.
The manufacturer's CEO said, "We are in the final steps of
helping Itonis get the [Emesyl™] product NDC registered with the
FDA, and doing the final sourcing of the key unique ingredients we
need to get a production run up on our filling line."
The complete update from the Emesyl™ manufacturer Oasis Health
Products is available at
http://itonisholdings.com/wp-content/uploads/2014/04/DK-Letter-to-Mark-Cheung-April-21-2014.pdf.
"We are excited that the Company will be clearing this final
governmental hurdle for the National Drug Code number that will
enable the final labeling of the Emesyl™ product. An NDC
number is a universal product identifier that is required for all
drug products whether pharmaceutical or homeopathic," says Mark
Cheung, Itonis' CEO.
About Itonis
Pharmaceuticals Itonis Pharmaceuticals, a division of
Itonis, Inc., is headed by Charles Hensley, Ph.D., and its mission
is to create and market over-the-counter and prescription
homeopathic products that better people's lives. Dr. Hensley is a
pioneer in the development and marketing of safe and effective
therapeutic nutraceutical and homeopathic preparations. He was
a founder of the company that launched the Zicam® Cold Remedy,
making the product a household name and forever changing the
marketing and product placement paradigm for homeopathic drugs in
the United States. Dr. Hensley and his team plan to use the
same methodologies to revolutionize the treatment of several other
common ailments and other chronic diseases.
About Itonis,
Inc. Itonis, Inc. was incorporated in the state of Nevada on
July 5, 2005 under the name of Kenshou, Inc., which later changed
to Itonis, Inc. on December 2, 2005. Please visit
www.itonisholdings.com.
Safe Harbor:
Statements in this press release may constitute forward-looking
statements and are subject to numerous risks and uncertainties,
including the failure to complete successfully the development of
new or enhanced products, the Company's future capital needs, the
lack of market demand for any new or enhanced products the Company
may develop, any actions by the Company's affiliates that may be
adverse to the Company, the success of competitive products, other
economic factors affecting the Company and its markets, seasonal
changes, and other risks detailed from time to time in the
Company's filings with the U.S. Securities and Exchange Commission.
The actual results may differ materially from those contained in
this press release. The Company disclaims any obligation to update
any statements in this press release.
Contact: Itonis, Inc. Email Contact
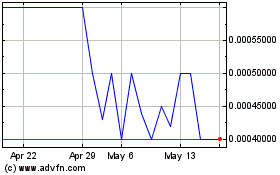
Itonis (PK) (USOTC:ITNS)
Historical Stock Chart
From Mar 2024 to Apr 2024
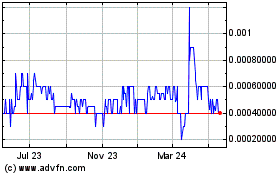
Itonis (PK) (USOTC:ITNS)
Historical Stock Chart
From Apr 2023 to Apr 2024