FDA Advisory Panel Meetings Will Address Moderna and J&J Boosters
October 01 2021 - 4:17PM
Dow Jones News
By Josh Beckerman
The U.S. Food and Drug Administration said an advisory committee
will discuss Moderna Inc. and Johnson & Johnson Covid-19
vaccine booster doses on Oct. 14 and Oct. 15.
The Vaccines and Related Biological Products Advisory Committee
will also discuss the use of "mix and match" boosters in which the
booster dose is a different vaccine than the one used for primary
vaccination.
On Sept. 22, the FDA approved a booster developed by Pfizer Inc.
and BioNTech SE for people 65 and older and certain other adults at
high risk of severe illness.
Write to Josh Beckerman at josh.beckerman@wsj.com
(END) Dow Jones Newswires
October 01, 2021 16:02 ET (20:02 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
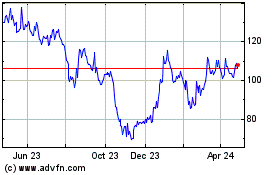
Moderna (NASDAQ:MRNA)
Historical Stock Chart
From Mar 2024 to Apr 2024
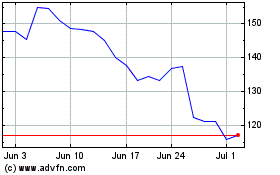
Moderna (NASDAQ:MRNA)
Historical Stock Chart
From Apr 2023 to Apr 2024