InMed Expands Patent Portfolio with Novel Cannabinoid Analogs and Advances Collaboration Agreement with Leading Cannabinoid Research Expert
April 28 2022 - 7:30AM

InMed Pharmaceuticals Inc. (“InMed” or the
“Company”) (Nasdaq: INM), a leader in the research, development,
manufacturing and commercialization of rare cannabinoids, is
pleased to announce the publication of a patent application in
North America for several cannabinoid analogs. This patent
application has broad claims directed to their molecular structure,
uses and methods of manufacturing.
Entitled “Cannabinoid Analogs and Methods for
their Preparation”, the patent application describes several new
cannabinoid-related chemical compounds that have not been
previously described. If granted, the broad patent application
allows for the creation of several variations of novel cannabinoid
compounds, producing a robust library of proprietary new chemical
entities (‘NCEs’).
Scientists at BayMedica, LLC, InMed’s subsidiary
in the US, designed and produced these cannabinoid analogs. These
NCEs are expected to offer similar or improved therapeutic effects
compared to their parent (naturally occurring) cannabinoid with
modifications that may make them preferred candidates to treat
specific diseases.
Eric Hsu, SVP, Pre-Clinical Research and
Development for InMed commented; “We believe rare cannabinoids hold
tremendous therapeutic potential that may address a number of unmet
medical needs. This patent covers the selective modification of
naturally occurring cannabinoids to target specific properties for
pharmaceutical development. Such patent protection is an important
component to ensure long-term commercial exclusivity as we continue
to invest in R&D.”
InMed has initiated a research collaboration
with the Department of Biotechnological and Applied Clinical
Sciences, University of L’Aquila (Italy) in the laboratory of Dr.
Mauro Maccarrone, an international expert in cannabinoid research
and founding member of the European Cannabinoid Research Alliance.
He is also the 2007 recipient of the International Association for
Cannabinoid Medicines’s prestigious Ester Fride Award for Basic
Research, and the 2016 recipient of the International Cannabinoid
Research Society’s prestigious Mechoulam Award. Dr. Maccarrone’s
lab will be screening the Company’s novel cannabinoid analogs to
investigate pharmacological properties and potential therapeutic
uses.
Creating a robust patent library of
NCEsThe patent application covers technology that allows
for the creation of libraries of NCEs that can be compared to other
drugs in order to select candidates for advancement. The
cannabinoid analogs were manufactured in a biosynthetic process
using the yeast S. cerevisiae, which is commonly used to produce
therapeutic biologicals and vaccines.
Filing this patent application in a number of
global jurisdicitions further emphasizes the Company’s commitment
towards developing NCEs and enhancing rare cannabinoids R&D
opportunities.
Why pursue cannabinoid
analogs?Unlike natural cannabinoids isolated from the
plant which are not patentable, these cannabinoid analogs are
patentable. Benefits may include:
- Targeting certain physiological
outcomes in specific diseases;
- Improving upon the safety profile
of the natural cannabinoids;
- Enhancing the ability to integrate
with specific delivery technologies; and
- Protecting the long-term research
investment and commercial opportunities.
Learn more about InMed’s Cannabinoid
Analogs:Novel Patentable Cannabinoid Analogs | InMed
Pharmaceuticals
Learn more about InMed’s Cannabinoids in Development:
https://www.inmedpharma.com/pharmaceutical/cannabinoids-in-development/
About InMed: InMed
Pharmaceuticals is a global leader in the research, development,
manufacturing and commercialization of rare cannabinoids. Together
with its subsidiary BayMedica, LLC, the Company has unparalleled
cannabinoid manufacturing capabilities to serve a spectrum of
consumer markets, including pharmaceutical and health and wellness.
InMed is also a clinical-stage company developing a pipeline of
rare cannabinoid therapeutics and dedicated to delivering new
treatment alternatives to patients that may benefit from
cannabinoid-based pharmaceutical drugs. For more information,
visit www.inmedpharma.com and www.baymedica.com.
Investor Contact: Colin ClancySenior Director,
Investor RelationsT: +1.604.416.0999E: cclancy@inmedpharma.com
Cautionary Note Regarding Forward-Looking
Information:
This news release contains "forward-looking
information" and "forward-looking statements" (collectively,
"forward-looking information") within the meaning of applicable
securities laws. Forward-looking information is based on
management's current expectations and beliefs and is subject to a
number of risks and uncertainties that could cause actual results
to differ materially from those described in the forward-looking
statements. Forward-looking information in this news release
includes statements about: expanding patent portfolio with novel
cannabinoid analogs; advancing collaboration agreement with leading
cannabinoid research expert; screening analogs for therapeutic
uses;publication of patent application in North America for several
cannabinoids analogs; the patent has broad claims directed to
molecular structure, uses and methods of manufacturing; NCEs are
expected to offer similar or improved therapeutic effects compared
to their parent (naturally occurring) cannabinoid with
modifications that may make them preferred candidates to treat
specific diseases; cannabinoids hold tremendous therapeutic
potential that may address a number of unmet medical needs; patent
application covering technology that allows for the creation of
libraries of NCEs that can be compared to other drugs in order to
select candidates for advancement; screening the Company’s novel
cannabinoid analogs to investigate pharmacological properties and
potential therapeutic uses; benefits of analogs that may include
targeting certain physiological outcomes in specific diseases,
improving upon the safety profile of the natural cannabinoids;
enhancing the ability to integrate with specific delivery
technologies; protecting the long-term research investment and
commercial opportunities; being a global leader in the research,
development, manufacturing and development of rare cannabinoids;
and delivering new treatment alternatives to patients that may
benefit from cannabinoid-based pharmaceutical drugs.
With respect to the forward-looking information
contained in this news release, InMed has made numerous
assumptions. While InMed considers these assumptions to be
reasonable, these assumptions are inherently subject to significant
business, economic, competitive, market and social uncertainties
and contingencies.
Additionally, there are known and unknown risk
factors which could cause InMed's actual results, performance or
achievements to be materially different from any future results,
performance or achievements expressed or implied by the
forward-looking information contained herein. A complete discussion
of the risks and uncertainties facing InMed’s stand-alone business
is disclosed in InMed’s Annual Report on Form 10-K and other
filings with the Security and Exchange Commission on
www.sec.gov.
All forward-looking information herein is
qualified in its entirety by this cautionary statement, and InMed
disclaims any obligation to revise or update any such
forward-looking information or to publicly announce the result of
any revisions to any of the forward-looking information contained
herein to reflect future results, events or developments, except as
required by law.
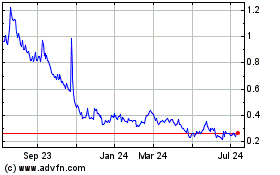
InMed Pharmaceuticals (NASDAQ:INM)
Historical Stock Chart
From Mar 2024 to Apr 2024
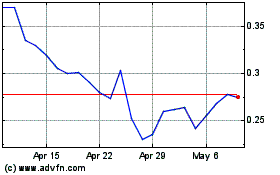
InMed Pharmaceuticals (NASDAQ:INM)
Historical Stock Chart
From Apr 2023 to Apr 2024