Sanofi to invest in a leading-edge production site in Singapore; continues to strengthen its vaccines manufacturing capacitie...
April 12 2021 - 1:00AM

Sanofi to invest in a leading-edge production site in Singapore;
continues to strengthen its vaccines manufacturing capacities
Sanofi to invest in a leading-edge production site in
Singapore; continues to strengthen its vaccines manufacturing
capacities
- Sanofi will invest €400 million over five years to create a
unique vaccine production site in Singapore, pushing the boundaries
of operations through cutting edge manufacturing and digital
technologies.
- The new site will provide Sanofi with the ability to produce
innovative vaccines on a massive scale for Asia, and quickly
respond to future pandemic risks.
- The Singapore site will complement Sanofi’s existing
manufacturing capacities in Europe and North America and will
become a regional center of excellence for vaccines production in
Asia.
PARIS – April 12, 2021 - Sanofi
today announced a €400 million investment over five years to create
a unique vaccine production center in Singapore, pushing the
boundaries of operations through cutting edge manufacturing and
digital technologies. In partnership with the Singapore Economic
Development Board (EDB), the new site will mainly supply the Asian
region and complement existing manufacturing capacities in Europe
and North America.
The investment furthers Sanofi’s commitment to
driving the future of vaccines by expanding manufacturing
capabilities to ensure quality and scale, while responding to the
risk of future pandemics. The project is expected to create up to
200 local jobs and enhance Singapore’s position as a regional
innovation hub for the healthcare industry.
“As a major healthcare player, it’s our
responsibility to act and to meet the unprecedented growing demands
for vaccines. By investing in a new production site in Singapore,
Sanofi is aiming to strengthen production capacity to meet
ever-growing global demands on vaccines, and answer more rapidly to
future pandemics,” said Thomas Triomphe, Executive Vice President
and Global Head of Sanofi Pasteur. “We are very pleased by the
strong collaboration with the Singapore Economic Development Board
to achieve this exciting milestone.”
“Sanofi’s decision to locate its first-in-Asia
digitally-enabled vaccine production center in Singapore, to supply
markets in the region and beyond, is an endorsement of Singapore’s
position as a leading centre for advanced manufacturing” said Dr
Beh Swan Gin, Chairman, EDB. “EDB will continue to promote
digitalization, automation and innovation to transform the
manufacturing sector and create good jobs for Singaporeans.”
This factory will be designed around a central
unit housing several fully digitalized modules that allow
production of three to four vaccines simultaneously, versus only
one in current industrial sites. In addition, the factory will have
the flexibility to leverage multiple vaccine manufacturing
technology platforms based on different cell types This modularity
and flexibility will allow the production of a specific vaccine to
be prioritized in a faster timeframe depending on public health
needs.
The project is currently entering its design
phase with construction expected to begin in Q3 2021. The five-year
project will see the site fully operational in Q1 2026 once all
qualifications and validations of the first manufactured vaccine
have been completed.
In the context of Sanofi’s environmental
strategy, the site is designed to be carbon-neutral, and to
drastically reduce both resource consumption and waste
production.
Editor’s Note: This
investment in a new vaccine manufacturing facility further
demonstrates Sanofi’s overall growth strategy, with vaccines
contributing as a key growth driver through differentiated
products, market expansion and new launches. It follows an
announcement made in March to build a new facility in Canada to
increase the Company’s global availability of high-dose influenza
vaccine.
A world leader in vaccines
Sanofi, with its global vaccine entity Sanofi
Pasteur, is a leading vaccine developer and manufacturer. Sanofi
offers a portfolio of vaccines to protect children, adolescents,
and adults from diseases such as influenza, infectious diseases,
and endemic diseases.
With 16,000 employees and an international
network of 12 industrial sites dedicated to vaccine manufacturing,
Sanofi Pasteur’s vaccines protect more than half a billion people
in the world.
|
About Sanofi Sanofi is dedicated to
supporting people through their health challenges. We are a global
biopharmaceutical company focused on human health. We prevent
illness with vaccines, provide innovative treatments to fight pain
and ease suffering. We stand by the few who suffer from rare
diseases and the millions with long-term chronic
conditions. With more than 100,000 people in 100 countries,
Sanofi is transforming scientific innovation into healthcare
solutions around the globe. Sanofi, Empowering Life |
|
Media Relations Contact Nicolas
Kressmann Tel.: +1 732-532-5318
Nicolas.Kressmann@sanofi.com |
Investor Relations Contacts
Paris Eva Schaefer-Jansen Arnaud
Delepine Investor Relations
Contacts North America Felix
LauscherFara BerkowitzSuzanne Greco IR main line:Tel.: +33
(0)1 53 77 45 45
investor.relations@sanofi.com https://www.sanofi.com/en/investors/contact |
|
Sanofi Forward-Looking StatementsThis press
release contains forward-looking statements as defined in the
Private Securities Litigation Reform Act of 1995, as amended.
Forward-looking statements are statements that are not historical
facts. These statements include projections and estimates and their
underlying assumptions, statements regarding plans, objectives,
intentions and expectations with respect to future financial
results, events, operations, services, product development and
potential, and statements regarding future performance.
Forward-looking statements are generally identified by the words
“expects”, “anticipates”, “believes”, “intends”, “estimates”,
“plans” and similar expressions. Although Sanofi’s management
believes that the expectations reflected in such forward-looking
statements are reasonable, investors are cautioned that
forward-looking information and statements are subject to various
risks and uncertainties, many of which are difficult to predict and
generally beyond the control of Sanofi, that could cause actual
results and developments to differ materially from those expressed
in, or implied or projected by, the forward-looking information and
statements. These risks and uncertainties include among other
things, the uncertainties inherent in research and development,
future clinical data and analysis, including post marketing,
decisions by regulatory authorities, such as the FDA or the EMA,
regarding whether and when to approve any drug, device or
biological application that may be filed for any such product
candidates as well as their decisions regarding labelling and other
matters that could affect the availability or commercial potential
of such product candidates, the fact that product candidates if
approved may not be commercially successful, the future approval
and commercial success of therapeutic alternatives, Sanofi’s
ability to benefit from external growth opportunities, to complete
related transactions and/or obtain regulatory clearances, risks
associated with intellectual property and any related pending or
future litigation and the ultimate outcome of such
litigation, trends in exchange rates and prevailing interest
rates, volatile economic and market conditions, cost containment
initiatives and subsequent changes thereto, and the impact that
COVID-19 will have on us, our customers, suppliers, vendors, and
other business partners, and the financial condition of any one of
them, as well as on our employees and on the global economy as a
whole. Any material effect of COVID-19 on any of the
foregoing could also adversely impact us. This situation is
changing rapidly, and additional impacts may arise of which we are
not currently aware and may exacerbate other previously identified
risks. The risks and uncertainties also include the uncertainties
discussed or identified in the public filings with the SEC and the
AMF made by Sanofi, including those listed under “Risk Factors” and
“Cautionary Statement Regarding Forward-Looking Statements” in
Sanofi’s annual report on Form 20-F for the year ended December 31,
2020. Other than as required by applicable law, Sanofi does not
undertake any obligation to update or revise any forward-looking
information or statements. |
- PDF
- Picture: new Sanofi site in Singapore
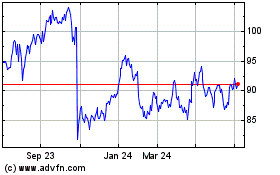
Sanofi (BIT:1SAN)
Historical Stock Chart
From Mar 2024 to Apr 2024
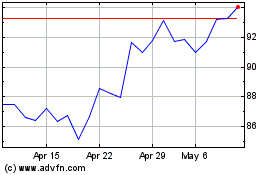
Sanofi (BIT:1SAN)
Historical Stock Chart
From Apr 2023 to Apr 2024