ABBOTT PARK, Ill., Jan. 28, 2016
/PRNewswire/ -- Abbott (NYSE: ABT) today announced
financial results for the fourth quarter ended Dec. 31, 2015.
- Adjusted diluted EPS from continuing operations, which excludes
specified items, was $0.62 in the
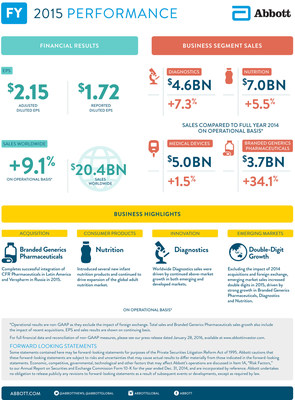
fourth quarter and $2.15 for the full
year, at the midpoint of Abbott's previous guidance range and
reflecting 8.6 percent growth for the full year. Reported diluted
EPS from continuing operations under GAAP was $0.46 in the fourth quarter and $1.72 for the full year.
- Fourth-quarter worldwide sales of $5.2
billion increased 4.9 percent on an operational basis and
decreased 3.1 percent on a reported basis.
- Full-year sales increased 9.1 percent on an operational basis
and 0.8 percent on a reported basis. Full-year sales in emerging
markets increased double digits excluding the impact of 2014
acquisitions and foreign exchange.
- For the full year 2015, Abbott expanded its adjusted gross and
operating margin ratios by 260 and 90 basis points over the prior
year, respectively. The gross and operating margin ratios under
GAAP improved by 250 and 120 basis points over the prior year,
respectively.
- Abbott issues full-year 2016 adjusted EPS guidance range of
$2.10 to $2.20. Projected full-year
2016 EPS under GAAP is $1.55 to
$1.65. Abbott's 2016 forecast assumes a significantly lower
contribution from Venezuelan operations as a result of challenging
market conditions in that country. Excluding the impact of foreign
exchange and Venezuela, the
midpoint of Abbott's 2016 adjusted EPS guidance range would reflect
strong double-digit growth.
"In 2015, we achieved top-tier sales and earnings growth despite
a challenging currency environment," said Miles D. White, chairman and chief executive
officer, Abbott. "Our underlying performance continues to be
strong."
FOURTH-QUARTER BUSINESS OVERVIEW
Note: Prior year financial results have been
adjusted to exclude the sales from Abbott's
developed markets branded generics pharmaceuticals and animal
health businesses that were sold to Mylan and Zoetis,
respectively, in the first quarter 2015. Therefore, sales
and growth rates shown in the following charts represent continuing
operations.
Following are sales by business segment and commentary for
the fourth quarter and the full year:
Total Company
($ in millions)
|
|
|
|
|
|
|
% Change vs.
4Q14
|
|
Sales
4Q15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
*
|
1,603
|
|
3,585
|
|
5,188
|
|
3.2
|
|
5.6
|
|
(5.7)
|
|
4.9
|
|
(3.1)
|
|
Nutrition
|
725
|
|
1,075
|
|
1,800
|
|
5.5
|
|
5.4
|
|
(3.7)
|
|
5.5
|
|
(0.2)
|
|
Diagnostics
|
367
|
|
853
|
|
1,220
|
|
5.1
|
|
7.7
|
|
(3.6)
|
|
7.0
|
|
(1.1)
|
|
Established
Pharmaceuticals
|
--
|
|
885
|
|
885
|
|
n/a
|
|
10.9
|
|
(4.0)
|
|
10.9
|
|
(4.0)
|
|
Medical
Devices
|
503
|
|
794
|
|
1,297
|
|
(0.7)
|
|
1.5
|
|
(9.0)
|
|
0.7
|
|
(5.9)
|
|
|
* Total Abbott Sales
from continuing operations include adjustments in Other Sales of
$14 million.
|
|
|
|
|
|
|
|
% Change vs.
12M14
|
|
Sales
12M15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
*
|
6,271
|
|
14,134
|
|
20,405
|
|
2.2
|
|
12.1
|
|
0.2
|
|
9.1
|
|
0.8
|
|
Nutrition
|
2,868
|
|
4,107
|
|
6,975
|
|
1.2
|
|
8.5
|
|
(0.3)
|
|
5.5
|
|
0.3
|
|
Diagnostics
|
1,392
|
|
3,254
|
|
4,646
|
|
6.2
|
|
7.8
|
|
(4.6)
|
|
7.3
|
|
(1.6)
|
|
Established
Pharmaceuticals
|
--
|
|
3,720
|
|
3,720
|
|
n/a
|
|
34.1
|
|
19.3
|
|
34.1
|
|
19.3
|
|
Medical
Devices
|
1,982
|
|
3,060
|
|
5,042
|
|
1.2
|
|
1.7
|
|
(10.9)
|
|
1.5
|
|
(6.5)
|
|
|
* Total Abbott Sales
from continuing operations include Other Sales of $22
million.
|
|
n/a = Not
Applicable.
|
|
Note: Operational
growth reflects percentage change over the prior year excluding the
impact of exchange rates.
|
Fourth-quarter 2015 worldwide sales of $5.2 billion increased 4.9 percent on an
operational basis and decreased 3.1 percent on a reported basis,
including an unfavorable 8.0 percent effect of foreign
exchange.
International sales increased 5.6 percent on an operational
basis and decreased 5.7 percent on a reported basis in the fourth
quarter.
Full-year 2015 worldwide sales of $20.4
billion increased 9.1 percent on an operational basis and
0.8 percent on a reported basis, including an unfavorable 8.3
percent effect of foreign exchange. Full-year sales in emerging
markets increased double digits excluding the impact of 2014
acquisitions and foreign exchange, driven by double-digit growth in
Established Pharmaceuticals, Diagnostics and Nutrition.
Nutrition
($ in
millions)
|
|
|
|
|
|
|
% Change vs.
4Q14
|
|
Sales
4Q15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
|
725
|
|
1,075
|
|
1,800
|
|
5.5
|
|
5.4
|
|
(3.7)
|
|
5.5
|
|
(0.2)
|
|
Pediatric
|
409
|
|
625
|
|
1,034
|
|
6.0
|
|
2.3
|
|
(5.6)
|
|
3.7
|
|
(1.3)
|
|
Adult
|
316
|
|
450
|
|
766
|
|
4.8
|
|
9.9
|
|
(1.1)
|
|
7.9
|
|
1.3
|
|
|
|
|
|
|
|
% Change vs.
12M14
|
|
Sales
12M15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
|
2,868
|
|
4,107
|
|
6,975
|
|
1.2
|
|
8.5
|
|
(0.3)
|
|
5.5
|
|
0.3
|
|
Pediatric
|
1,592
|
|
2,378
|
|
3,970
|
|
3.9
|
|
8.1
|
|
0.7
|
|
6.4
|
|
2.0
|
|
Adult
|
1,276
|
|
1,729
|
|
3,005
|
|
(2.1)
|
|
9.1
|
|
(1.5)
|
|
4.3
|
|
(1.8)
|
Worldwide Nutrition sales increased 5.5 percent in the fourth
quarter on an operational basis and decreased 0.2 percent on a
reported basis, including an unfavorable 5.7 percent effect of
foreign exchange.
Worldwide Pediatric Nutrition sales increased 3.7 percent on an
operational basis and decreased 1.3 percent on a reported basis in
the quarter, including an unfavorable 5.0 percent effect of foreign
exchange. In the U.S., sales growth in the quarter was led by
continued consumer uptake of several recently launched non-GMO
products. International growth was led by share expansion of
ElevaTM in the premium segment of the Chinese market and
double-digit operational growth across several Latin American
countries, partially offset by a difficult comparison to the fourth
quarter 2014, when sales increased strong double digits.
Worldwide Adult Nutrition sales increased 7.9 percent on an
operational basis and 1.3 percent on a reported basis in the
quarter, including an unfavorable 6.6 percent effect of foreign
exchange. International growth was led by continued category
expansion in Latin America and
other priority international markets. U.S. adult nutrition sales
increased 4.8 percent led by growth of Ensure® in the
retail and institutional segments of the market.
Diagnostics
($ in millions)
|
|
|
|
|
|
|
% Change vs.
4Q14
|
|
Sales
4Q15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
|
367
|
|
853
|
|
1,220
|
|
5.1
|
|
7.7
|
|
(3.6)
|
|
7.0
|
|
(1.1)
|
|
Core
Laboratory
|
217
|
|
752
|
|
969
|
|
6.3
|
|
7.7
|
|
(3.9)
|
|
7.4
|
|
(1.8)
|
|
Molecular
|
46
|
|
80
|
|
126
|
|
(7.1)
|
|
8.6
|
|
(1.8)
|
|
2.6
|
|
(3.9)
|
|
Point of
Care
|
104
|
|
21
|
|
125
|
|
9.0
|
|
6.5
|
|
(0.4)
|
|
8.6
|
|
7.3
|
|
|
|
|
|
|
|
% Change vs.
12M14
|
|
Sales
12M15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
|
1,392
|
|
3,254
|
|
4,646
|
|
6.2
|
|
7.8
|
|
(4.6)
|
|
7.3
|
|
(1.6)
|
|
Core
Laboratory
|
813
|
|
2,894
|
|
3,707
|
|
6.3
|
|
7.4
|
|
(5.1)
|
|
7.2
|
|
(2.8)
|
|
Molecular
|
191
|
|
275
|
|
466
|
|
(3.9)
|
|
11.6
|
|
(1.1)
|
|
5.2
|
|
(2.2)
|
|
Point of
Care
|
388
|
|
85
|
|
473
|
|
12.0
|
|
10.7
|
|
2.6
|
|
11.7
|
|
10.2
|
Worldwide Diagnostics sales increased 7.0 percent in the fourth
quarter on an operational basis, driven by continued above-market
growth in both emerging and developed markets. Sales decreased 1.1
percent on a reported basis, including an unfavorable 8.1 percent
effect of foreign exchange.
Core Laboratory Diagnostics sales increased 7.4 percent in the
quarter on an operational basis and decreased 1.8 percent on a
reported basis, including an unfavorable 9.2 percent effect of
foreign exchange. Operational sales growth in the quarter was
driven by double-digit growth in emerging markets and continued
share gains in the U.S. and internationally.
Molecular Diagnostics sales increased 2.6 percent in the quarter
on an operational basis and decreased 3.9 percent on a reported
basis, including an unfavorable 6.5 percent effect of foreign
exchange. Strong growth in the infectious disease testing business,
which remains Abbott's core focus area in Molecular Diagnostics,
led operational growth in the quarter. As expected, U.S. growth was
impacted by the planned scale down of the genetics business.
Point of Care Diagnostics sales increased 8.6 percent in the
quarter on an operational basis as this business continues to
expand its product offering and geographic presence in targeted
developed and emerging markets. Sales increased 7.3 percent on
a reported basis, including an unfavorable 1.3 percent effect of
foreign exchange.
Established Pharmaceuticals
($ in millions)
Note: Prior year financial results have been
adjusted to exclude the sales from the
developed markets branded generics pharmaceuticals business,
which was sold to Mylan on Feb. 27,
2015. Therefore, sales and growth rates shown in the
following charts represent continuing operations.
|
|
|
|
|
|
|
% Change vs.
4Q14
|
|
Sales
4Q15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
|
--
|
|
885
|
|
885
|
|
n/a
|
|
10.9
|
|
(4.0)
|
|
10.9
|
|
(4.0)
|
|
Key Emerging
Markets
|
--
|
|
703
|
|
703
|
|
n/a
|
|
12.1
|
|
(4.3)
|
|
12.1
|
|
(4.3)
|
|
Other
|
--
|
|
182
|
|
182
|
|
n/a
|
|
6.2
|
|
(3.1)
|
|
6.2
|
|
(3.1)
|
|
|
|
|
|
|
|
% Change vs.
12M14
|
|
Sales
12M15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
|
--
|
|
3,720
|
|
3,720
|
|
n/a
|
|
34.1
|
|
19.3
|
|
34.1
|
|
19.3
|
|
Key Emerging
Markets
|
--
|
|
2,781
|
|
2,781
|
|
n/a
|
|
32.4
|
|
16.7
|
|
32.4
|
|
16.7
|
|
Other
|
--
|
|
939
|
|
939
|
|
n/a
|
|
39.6
|
|
27.8
|
|
39.6
|
|
27.8
|
Established Pharmaceuticals sales increased 10.9 percent in the
fourth quarter on an operational basis and decreased 4.0 percent on
a reported basis, including an unfavorable 14.9 percent effect of
foreign exchange.
Sales in Key Emerging Markets increased 12.1 percent on an
operational basis, driven by continued double-digit growth in
India, China, and Russia. Sales decreased 4.3 percent on a
reported basis, including an unfavorable 16.4 percent effect of
foreign exchange.
For the full year 2015, Established Pharmaceutical sales
increased double digits excluding the impact of acquisitions and
foreign exchange. Growth was driven by commercial initiatives and
locally-relevant portfolio expansion in key geographies.
During the year, Abbott successfully integrated the acquisitions
of CFR Pharmaceuticals in Latin
America and Veropharm in Russia. These businesses
strengthened Abbott's capabilities, relevant product portfolios,
and leadership in key geographies.
Medical Devices
($ in millions)
|
|
|
|
|
|
|
% Change vs.
4Q14
|
|
Sales
4Q15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
|
503
|
|
794
|
|
1,297
|
|
(0.7)
|
|
1.5
|
|
(9.0)
|
|
0.7
|
|
(5.9)
|
|
Vascular
|
286
|
|
414
|
|
700
|
|
(0.8)
|
|
(0.3)
|
|
(10.8)
|
|
(0.5)
|
|
(7.0)
|
|
Diabetes
Care
|
101
|
|
196
|
|
297
|
|
(7.1)
|
|
7.5
|
|
(4.3)
|
|
2.4
|
|
(5.2)
|
|
Medical
Optics
|
116
|
|
184
|
|
300
|
|
5.7
|
|
(0.2)
|
|
(9.4)
|
|
1.9
|
|
(4.1)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Vascular Product
Lines:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Coronary
Devicesa)
|
197
|
|
353
|
|
550
|
|
3.5
|
|
(1.4)
|
|
(11.6)
|
|
0.2
|
|
(6.8)
|
|
Endovascularb)
|
72
|
|
60
|
|
132
|
|
1.3
|
|
5.8
|
|
(6.2)
|
|
3.4
|
|
(2.3)
|
|
|
a)Includes
DES / BVS product portfolio, structural heart, guidewires, balloon
catheters, and other coronary products.
|
|
b)Includes
vessel closure, carotid stents and other peripheral
products.
|
|
|
|
|
|
|
|
% Change vs.
12M14
|
|
Sales
12M15
|
|
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Int'l
|
|
Total
|
|
U.S.
|
|
Operational
|
|
Reported
|
|
Operational
|
|
Reported
|
|
Total
|
1,982
|
|
3,060
|
|
5,042
|
|
1.2
|
|
1.7
|
|
(10.9)
|
|
1.5
|
|
(6.5)
|
|
Vascular
|
1,145
|
|
1,647
|
|
2,792
|
|
2.6
|
|
0.6
|
|
(11.9)
|
|
1.3
|
|
(6.5)
|
|
Diabetes
Care
|
394
|
|
723
|
|
1,117
|
|
(2.7)
|
|
5.8
|
|
(7.9)
|
|
2.9
|
|
(6.1)
|
|
Medical
Optics
|
443
|
|
690
|
|
1,133
|
|
1.3
|
|
0.2
|
|
(11.5)
|
|
0.6
|
|
(6.9)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Vascular Product
Lines:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Coronary
Devicesa)
|
768
|
|
1,408
|
|
2,176
|
|
4.7
|
|
(0.3)
|
|
(12.5)
|
|
1.3
|
|
(7.1)
|
|
Endovascularb)
|
282
|
|
238
|
|
520
|
|
5.7
|
|
5.5
|
|
(8.3)
|
|
5.6
|
|
(1.2)
|
|
|
|
a)Includes
DES / BVS product portfolio, structural heart, guidewires, balloon
catheters, and other coronary products.
|
|
b)Includes
vessel closure, carotid stents and other peripheral
products.
|
Worldwide Medical Devices sales increased 0.7 percent in the
fourth quarter on an operational basis and decreased 5.9 percent on
a reported basis, including an unfavorable 6.6 percent effect of
foreign exchange.
Worldwide sales of Vascular products decreased 0.5 percent in
the quarter on an operational basis and 7.0 percent on a reported
basis, including an unfavorable 6.5 percent effect of foreign
exchange. Sales of Abbott's MitraClip® device for the
treatment of mitral regurgitation increased double digits globally,
as Abbott continues to build the market for this first-in-class
device. In October, at the Transcatheter Cardiovascular
Therapeutics conference, Abbott presented trial results for
AbsorbTM, its first-of-its-kind fully dissolving stent,
showing comparable outcomes to XIENCETM, its highly
successful drug-eluting stent. In 2015, Abbott completed
submissions for regulatory approval of Absorb in Japan and the U.S.
Worldwide Diabetes Care sales increased 2.4 percent in the
quarter on an operational basis and decreased 5.2 percent on a
reported basis, including an unfavorable 7.6 percent effect of
foreign exchange. Internationally, operational sales growth
was driven by continued uptake of Abbott's FreeStyle®
Libre Flash Glucose Monitoring System. Abbott recently completed a
significant expansion of its manufacturing capacity for FreeStyle
Libre to meet strong customer demand.
Worldwide Medical Optics sales increased 1.9 percent in the
quarter on an operational basis and decreased 4.1 percent on a
reported basis, including an unfavorable 6.0 percent effect of
foreign exchange. Operational sales growth was driven by uptake of
new cataract products in the premium intraocular lens
segment. This growth was partially offset by market dynamics
in the refractive business.
ABBOTT ISSUES EARNINGS-PER-SHARE OUTLOOK FOR 2016
Abbott is issuing full-year 2016 guidance for earnings per
share, excluding specified items, of $2.10
to $2.20. Abbott's 2016 forecast assumes a significantly
lower contribution from Venezuelan operations as a result of
challenging market conditions in that country. Excluding the impact
of foreign exchange and Venezuela,
the midpoint of Abbott's 2016 adjusted EPS guidance range would
reflect strong double-digit growth.
Abbott forecasts net specified items for the full year 2016 of
approximately $0.55 per share.
Specified items include intangible amortization expense, charges
associated with cost reduction initiatives and deal and other
expenses.
Including net specified items, projected earnings per share
under Generally Accepted Accounting Principles (GAAP) would be
$1.55 to $1.65 for the full year
2016.
Abbott is issuing first-quarter 2016 guidance for earnings per
share, excluding specified items, of $0.38
to $0.40. Abbott forecasts specified items for the
first quarter 2016 of $0.15 related
to the same items discussed above for the full year 2016.
Including specified items, projected earnings per share under GAAP
would be $0.23 to $0.25 for the first
quarter.
ABBOTT ANNOUNCES INCREASE IN QUARTERLY DIVIDEND
On Dec. 11, 2015, the board of
directors of Abbott increased the company's quarterly dividend to
$0.26 per share from $0.24 per share. Abbott's cash dividend is
payable Feb. 16, 2016, to
shareholders of record at the close of business on Jan. 15, 2016. This marks the 368th
consecutive quarterly dividend paid by Abbott.
Abbott is a member of the S&P 500 Dividend Aristocrats
Index, which tracks companies that have annually increased their
dividend for 25 consecutive years.
About Abbott:
Abbott is a global healthcare company devoted to improving life
through the development of products and technologies that span the
breadth of healthcare. With a portfolio of leading, science-based
offerings in diagnostics, medical devices, nutritionals and branded
generic pharmaceuticals, Abbott serves people in more than 150
countries and employs approximately 74,000 people.
Visit Abbott at www.abbott.com and connect with us on Twitter at
@AbbottNews.
Abbott will webcast its live fourth-quarter earnings conference
call through its Investor Relations website at
www.abbottinvestor.com at 8 a.m. Central
time today. An archived edition of the call will be
available after 11 a.m. Central
time.
- Private Securities Litigation Reform
Act of 1995 -
A Caution Concerning Forward-Looking
Statements
Some statements in this news release may be forward-looking
statements for purposes of the Private Securities Litigation Reform
Act of 1995. Abbott cautions that these forward-looking statements
are subject to risks and uncertainties that may cause actual
results to differ materially from those indicated in the
forward-looking statements. Economic, competitive, governmental,
technological and other factors that may affect Abbott's operations
are discussed in Item 1A, "Risk Factors,'' to our Annual Report on
Securities and Exchange Commission Form 10-K for the year ended
Dec. 31, 2014, and are incorporated
by reference. Abbott undertakes no obligation to release publicly
any revisions to forward-looking statements as a result of
subsequent events or developments, except as required by
law.
|
Abbott Laboratories
and Subsidiaries
|
|
Consolidated
Statement of Earnings
|
|
Fourth Quarter Ended
December 31, 2015 and 2014
|
|
(in millions, except
per share data)
|
|
(unaudited)
|
|
|
4Q15
|
|
4Q14
|
|
%
Change
|
|
|
Net Sales
|
$5,188
|
|
$5,356
|
|
(3.1)
|
|
|
|
|
|
|
|
|
|
Cost of products
sold, excluding amortization expense
|
2,206
|
|
2,337
|
|
(5.6)
|
|
|
Amortization of
intangible assets
|
143
|
|
163
|
|
(12.7)
|
|
|
Research and
development
|
369
|
|
361
|
|
2.4
|
|
|
Selling, general, and
administrative
|
1,655
|
|
1,666
|
|
(0.6)
|
|
|
Total Operating Cost
and Expenses
|
4,373
|
|
4,527
|
|
(3.4)
|
|
|
|
|
|
|
|
|
|
Operating
earnings
|
815
|
|
829
|
|
(1.7)
|
|
|
|
|
|
|
|
|
|
Interest expense,
net
|
9
|
|
19
|
|
(54.7)
|
|
|
Net loss on
extinguishment of debt
|
--
|
|
18
|
|
n/m
|
|
|
Net foreign exchange
(gain) loss
|
(30)
|
|
(24)
|
|
21.6
|
|
|
Other (income)
expense, net
|
6
|
|
12
|
|
(55.0)
|
|
|
Earnings from
Continuing Operations before taxes
|
830
|
|
804
|
|
3.4
|
|
|
|
|
|
|
|
|
|
Taxes on Earnings
from Continuing Operations
|
135
|
|
170
|
|
(20.6)
|
|
|
Earnings from
Continuing Operations
|
695
|
|
634
|
|
9.8
|
|
|
|
|
|
|
|
|
|
Earnings from
Discontinued Operations, net of taxes
|
72
|
|
271
|
|
(73.5)
|
1)
|
|
Gain on Sale of
Discontinued Operations, net of taxes
|
--
|
|
--
|
|
n/m
|
|
|
Net Earnings from
Discontinued Operations, net of taxes
|
72
|
|
271
|
|
(73.5)
|
1)
|
|
|
|
|
|
|
|
|
Net
Earnings
|
$767
|
|
$905
|
|
(15.2)
|
|
|
|
|
|
|
|
|
|
Net Earnings from
Continuing Operations, excluding
|
|
|
|
|
|
|
|
Specified Items, as
described below
|
$932
|
|
$943
|
|
(1.2)
|
2)
|
|
|
|
|
|
|
|
|
Diluted Earnings per
Common Share from:
|
|
|
|
|
|
|
|
Continuing
Operations
|
$0.46
|
|
$0.41
|
|
12.2
|
|
|
Discontinued
Operations
|
0.05
|
|
0.18
|
|
(72.2)
|
1)
|
|
Total
|
$0.51
|
|
$0.59
|
|
(13.6)
|
|
|
|
|
|
|
|
|
|
Diluted Earnings per
Common Share from Continuing Operations, excluding Specified Items, as described
below
|
$0.62
|
|
$0.62
|
|
--
|
2)
|
|
|
|
|
|
|
|
|
Average Number of
Common Shares Outstanding Plus
Dilutive Common Stock Options
|
1,498
|
|
1,522
|
|
|
|
|
|
NOTES:
|
|
|
See tables below for
an explanation of certain non-GAAP financial
information.
|
|
n/m = Percent change
is not meaningful.
|
|
See footnotes
below.
|
|
|
|
1)
|
2015 Earnings and
Diluted Earnings per Common Share from Discontinued Operations
primarily reflect net favorable adjustments to tax expense as a
result of the resolution of various tax positions from previous
years related to discontinued operations.
|
|
|
|
2014 Earnings and
Diluted Earnings per Common Share from Discontinued Operations
reflect financial results from the developed markets branded
generics pharmaceuticals and animal health businesses, and a net
favorable adjustment to tax expense as a result of the resolution
of various tax positions from previous years related to AbbVie
operations.
|
|
|
|
2)
|
2015 Net Earnings
from Continuing Operations, excluding Specified Items, excludes net
after-tax charges of $237 million, or $0.16 per share, for
intangible amortization expense, expenses associated with cost
reduction initiatives and other expenses related to
acquisitions.
|
|
|
|
2014 Net Earnings
from Continuing Operations, excluding Specified Items, excludes net
after-tax charges of $309 million, or $0.21 per share, for
intangible amortization expense, expenses associated with cost
reduction initiatives and deal and other expenses related to the
acquisitions, as well as the tax expense associated with a one-time
repatriation of 2014 ex-U.S. earnings, partially offset by
favorability as a result of the resolution of various tax positions
and adjustment of tax uncertainties from prior years.
|
|
Abbott Laboratories
and Subsidiaries
|
|
Consolidated
Statement of Earnings
|
|
Fiscal Year Ended
December 31, 2015 and 2014
|
|
(in millions, except
per share data)
|
|
(unaudited)
|
|
|
12M15
|
|
12M14
|
|
%
Change
|
|
|
Net Sales
|
$20,405
|
|
$20,247
|
|
0.8
|
|
|
|
|
|
|
|
|
|
Cost of products
sold, excluding amortization expense
|
8,747
|
|
9,218
|
|
(5.1)
|
|
|
Amortization of
intangible assets
|
601
|
|
555
|
|
8.2
|
|
|
Research and
development
|
1,405
|
|
1,345
|
|
4.5
|
|
|
Selling, general, and
administrative
|
6,785
|
|
6,530
|
|
3.9
|
|
|
Total Operating Cost
and Expenses
|
17,538
|
|
17,648
|
|
(0.6)
|
|
|
|
|
|
|
|
|
|
Operating
earnings
|
2,867
|
|
2,599
|
|
10.3
|
|
|
|
|
|
|
|
|
|
Interest expense,
net
|
58
|
|
73
|
|
(21.2)
|
|
|
Net loss on
extinguishment of debt
|
--
|
|
18
|
|
n/m
|
|
|
Net foreign exchange
(gain) loss
|
(93)
|
|
(24)
|
|
n/m
|
|
|
Other (income)
expense, net
|
(281)
|
|
14
|
|
n/m
|
1)
|
|
Earnings from
Continuing Operations before taxes
|
3,183
|
|
2,518
|
|
26.4
|
|
|
|
|
|
|
|
|
|
Taxes on Earnings
from Continuing Operations
|
577
|
|
797
|
|
(27.6)
|
|
|
Earnings from
Continuing Operations
|
2,606
|
|
1,721
|
|
51.4
|
|
|
|
|
|
|
|
|
|
Earnings from
Discontinued Operations, net of taxes
|
65
|
|
563
|
|
(88.5)
|
|
|
Gain on Sale of
Discontinued Operations, net of taxes
|
1,752
|
|
--
|
|
n/m
|
|
|
Net Earnings from
Discontinued Operations, net of taxes
|
1,817
|
|
563
|
|
n/m
|
2)
|
|
|
|
|
|
|
|
|
Net
Earnings
|
$4,423
|
|
$2,284
|
|
93.6
|
|
|
|
|
|
|
|
|
|
Net Earnings from
Continuing Operations, excluding
|
|
|
|
|
|
|
|
Specified Items, as
described below
|
$3,258
|
|
$3,038
|
|
7.2
|
3)
|
|
|
|
|
|
|
|
|
Diluted Earnings per
Common Share from:
|
|
|
|
|
|
|
|
Continuing
Operations
|
$1.72
|
|
$1.12
|
|
53.6
|
|
|
Discontinued
Operations
|
1.20
|
|
0.37
|
|
n/m
|
2)
|
|
Total
|
$2.92
|
|
$1.49
|
|
96.0
|
|
|
|
|
|
|
|
|
|
Diluted Earnings per
Common Share from Continuing Operations, excluding Specified Items, as described
below
|
$2.15
|
|
$1.98
|
|
8.6
|
3)
|
|
|
|
|
|
|
|
|
Average Number of
Common Shares Outstanding Plus
Dilutive Common Stock Options
|
1,506
|
|
1,527
|
|
|
|
|
|
NOTES:
|
|
|
See tables below for
an explanation of certain non-GAAP financial
information.
|
|
n/m = Percent change
is not meaningful.
|
|
See footnotes
below.
|
|
|
1)
|
2015 Other (income)
expense includes a gain on the sale of a portion of Abbott's
position in Mylan stock and a decrease in the fair value of
contingent consideration related to a business acquisition, both
reported as specified items.
|
|
|
|
2)
|
2015 Earnings and
Diluted Earnings per Common Share from Discontinued Operations
reflect the after-tax gain of $1.752 billion on the sale of the
developed markets branded generics pharmaceuticals and animal
health businesses to Mylan on Feb. 27, 2015 and Zoetis on Feb. 10,
2015, respectively; the first-quarter financial results from these
businesses up to the date of sale; and a favorable adjustment to
tax expense as a result of the resolution of various tax positions
from previous years related to discontinued operations.
|
|
|
|
2014 Earnings and
Diluted Earnings per Common Share from Discontinued Operations
reflect financial results from the developed markets branded
generics pharmaceuticals and animal health businesses, and a net
favorable adjustment to tax expense as a result of the resolution
of various tax positions from previous years related to AbbVie
operations.
|
|
|
|
3)
|
2015 Net Earnings
from Continuing Operations, excluding Specified Items, excludes net
after-tax charges of $652 million, or $0.43 per share, for
intangible amortization expense, expenses associated with cost
reduction initiatives and other expenses related to acquisitions,
partially offset by a gain on the sale of a portion of Abbott's
position in Mylan stock and a decrease in the fair value of
contingent consideration related to a business
acquisition.
|
|
|
|
2014 Net Earnings
from Continuing Operations, excluding Specified Items, excludes net
after-tax charges of $1.317 billion, or $0.86 per share, for
intangible amortization expense, expenses associated with cost
reduction initiatives and deal and other expenses related to
acquisitions, as well as the tax expense associated with a one-time
repatriation of 2014 ex-U.S. earnings, partially offset by
favorability as a result of the resolution of various tax positions
and adjustment of tax uncertainties from prior years.
|
|
NON-GAAP
RECONCILIATION OF FINANCIAL INFORMATION FROM CONTINUING
OPERATIONS
|
|
Abbott Laboratories
and Subsidiaries
|
|
Non-GAAP
Reconciliation of Financial Information From Continuing
Operations
|
|
Fourth Quarter Ended
December 31, 2015 and 2014
|
|
(in millions, except
per share data)
|
|
(unaudited)
|
|
|
4Q15
|
|
As
Reported
(GAAP)
|
|
Specified
Items
|
|
As
Adjusted
|
|
% to
Sales
|
|
|
|
|
|
|
|
|
|
Intangible
Amortization
|
$143
|
|
($143)
|
|
--
|
|
|
|
Gross
Margin
|
2,839
|
|
178
|
|
$3,017
|
|
58.2%
|
|
R&D
|
369
|
|
(4)
|
|
365
|
|
7.0%
|
|
SG&A
|
1,655
|
|
(117)
|
|
1,538
|
|
29.6%
|
|
Other (Income)
Expense, Net
|
6
|
|
(6)
|
|
--
|
|
|
|
Earnings from
Continuing Operations before taxes
|
830
|
|
305
|
|
1,135
|
|
|
|
Taxes on Earnings
from Continuing Operations
|
135
|
|
68
|
|
203
|
|
|
|
Net Earnings from
Continuing Operations
|
695
|
|
237
|
|
932
|
|
|
|
Diluted Earnings per
Share from Continuing Operations
|
$0.46
|
|
$0.16
|
|
$0.62
|
|
|
Specified items reflect intangible amortization expense of
$143 million and other expenses of
$162 million, primarily associated
with cost reduction initiatives and acquisitions.
|
4Q14
|
|
As
Reported
(GAAP)
|
|
Specified
Items
|
|
As
Adjusted
|
|
%
to
Sales
|
|
|
|
|
|
|
|
|
|
Intangible
Amortization
|
$163
|
|
($163)
|
|
--
|
|
|
|
Gross
Margin
|
2,856
|
|
191
|
|
$3,047
|
|
56.9%
|
|
R&D
|
361
|
|
(19)
|
|
342
|
|
6.4%
|
|
SG&A
|
1,666
|
|
(86)
|
|
1,580
|
|
29.5%
|
|
Net loss on
extinguishment of debt
|
18
|
|
(18)
|
|
--
|
|
|
|
Other (Income)
Expense, Net
|
12
|
|
(1)
|
|
11
|
|
|
|
Earnings from
Continuing Operations before taxes
|
804
|
|
315
|
|
1,119
|
|
|
|
Taxes on Earnings
from Continuing Operations
|
170
|
|
6
|
|
176
|
|
|
|
Net Earnings from
Continuing Operations
|
634
|
|
309
|
|
943
|
|
|
|
Diluted Earnings per
Share from Continuing Operations
|
$0.41
|
|
$0.21
|
|
$0.62
|
|
|
Specified items reflect intangible amortization expense of
$163 million and other expenses of
$152 million, primarily associated
with cost reduction initiatives and deal and other expenses related
to acquisitions, as well as tax expense of $175 million associated with a one-time
repatriation of 2014 ex-U.S. earnings, partially offset by
favorability as a result of the resolution of various tax positions
and adjustment of tax uncertainties from prior years.
|
Abbott Laboratories
and Subsidiaries
|
|
Non-GAAP
Reconciliation of Financial Information From Continuing
Operations
|
|
Fiscal Year Ended
December 31, 2015 and 2014
|
|
(in millions, except
per share data)
|
|
(unaudited)
|
|
|
12M15
|
|
As
Reported
(GAAP)
|
|
Specified
Items
|
|
As
Adjusted
|
|
% to
Sales
|
|
|
|
|
|
|
|
|
|
Intangible
Amortization
|
$601
|
|
($601)
|
|
--
|
|
|
|
Gross
Margin
|
11,057
|
|
755
|
|
$11,812
|
|
57.9%
|
|
R&D
|
1,405
|
|
(85)
|
|
1,320
|
|
6.5%
|
|
SG&A
|
6,785
|
|
(272)
|
|
6,513
|
|
31.9%
|
|
Other (Income)
Expense, Net
|
(281)
|
|
288
|
|
7
|
|
|
|
Earnings from
Continuing Operations before taxes
|
3,183
|
|
824
|
|
4,007
|
|
|
|
Taxes on Earnings
from Continuing Operations
|
577
|
|
172
|
|
749
|
|
|
|
Net Earnings from
Continuing Operations
|
2,606
|
|
652
|
|
3,258
|
|
|
|
Diluted Earnings per
Share from Continuing Operations
|
$1.72
|
|
$0.43
|
|
$2.15
|
|
|
Specified items reflect intangible amortization expense of
$601 million and other expenses of
$510 million, primarily associated
with cost reduction initiatives and acquisitions, partially offset
by a gain on the sale of a portion of Abbott's position in Mylan
stock of $207 million and a decrease
in the fair value of contingent consideration related to a business
acquisition.
|
12M14
|
|
As
Reported
(GAAP)
|
|
Specified
Items
|
|
As
Adjusted
|
|
% to
Sales
|
|
|
|
|
|
|
|
|
|
Intangible
Amortization
|
$555
|
|
($555)
|
|
--
|
|
|
|
Gross
Margin
|
10,474
|
|
721
|
|
$11,195
|
|
55.3%
|
|
R&D
|
1,345
|
|
(72)
|
|
1,273
|
|
6.3%
|
|
SG&A
|
6,530
|
|
(367)
|
|
6,163
|
|
30.4%
|
|
Net loss on
extinguishment of debt
|
18
|
|
(18)
|
|
--
|
|
|
|
Other (Income)
Expense, Net
|
14
|
|
(9)
|
|
5
|
|
|
|
Earnings from
Continuing Operations before taxes
|
2,518
|
|
1,187
|
|
3,705
|
|
|
|
Taxes on Earnings
from Continuing Operations
|
797
|
|
(130)
|
|
667
|
|
|
|
Net Earnings from
Continuing Operations
|
1,721
|
|
1,317
|
|
3,038
|
|
|
|
Diluted Earnings per
Share from Continuing Operations
|
$1.12
|
|
$0.86
|
|
$1.98
|
|
|
Specified items reflect intangible amortization expense of
$555 million and other expenses of
$632 million, primarily associated
with cost reduction initiatives and deal and other expenses related
to acquisitions, as well as tax expense of $440 million associated with a one-time
repatriation of 2014 ex-U.S. earnings, partially offset by
favorability as a result of the resolution of various tax positions
and adjustment of tax uncertainties from prior years.
RECONCILIATION OF TAX RATE FOR CONTINUING OPERATIONS
A reconciliation of the fourth-quarter tax rates for continuing
operations for 2015 and 2014 is shown below:
|
4Q15
|
|
|
($ in
millions)
|
Pre-Tax
Income
|
|
Taxes on
Earnings
|
|
Tax
Rate
|
|
|
As reported
(GAAP)
|
$830
|
|
$135
|
|
16.3%
|
|
|
Specified
items
|
305
|
|
68
|
|
|
|
|
Excluding
specified items
|
$1,135
|
|
$203
|
|
17.9%
|
1)
|
|
|
|
|
|
|
|
|
4Q14
|
|
|
($ in
millions)
|
Pre-Tax
Income
|
|
Taxes on
Earnings
|
|
Tax
Rate
|
|
|
As reported
(GAAP)
|
$804
|
|
$170
|
|
21.1%
|
2)
|
|
Specified
items
|
315
|
|
6
|
|
|
|
|
Excluding
specified items
|
$1,119
|
|
$176
|
|
15.7%
|
1)
|
|
|
|
1)
|
Fourth-quarter 2014
and 2015 tax rates include the year-to-date impact of U.S. tax
legislation passed in Dec. 2014 and 2015, respectively, including
the R&D tax credit.
|
|
|
|
2)
|
Reported tax rate on
a GAAP basis includes the impact of tax expense of $175 million
associated with a one-time repatriation of 2014 ex-U.S. earnings,
partially offset by favorability of $133 million as a result of the
resolution of various tax positions and adjustment of tax
uncertainties from prior years.
|
A reconciliation of the full-year tax rates for continuing
operations for 2015 and 2014 is shown below:
|
12M15
|
|
|
($ in
millions)
|
Pre-Tax
Income
|
|
Taxes on
Earnings
|
|
Tax
Rate
|
|
|
As reported
(GAAP)
|
$3,183
|
|
$577
|
|
18.1%
|
|
|
Specified
items
|
824
|
|
172
|
|
|
|
|
Excluding
specified items
|
$4,007
|
|
$749
|
|
18.7%
|
3)
|
|
|
|
|
|
|
|
|
12M14
|
|
|
($ in
millions)
|
Pre-Tax
Income
|
|
Taxes on
Earnings
|
|
Tax
Rate
|
|
|
As reported
(GAAP)
|
$2,518
|
|
$797
|
|
31.7%
|
4)
|
|
Specified
items
|
1,187
|
|
(130)
|
|
|
|
|
Excluding
specified items
|
$3,705
|
|
$667
|
|
18.0%
|
3)
|
|
|
|
3)
|
Full-year 2014 and
2015 tax rates include the impact of U.S. tax legislation passed in
Dec. 2014 and 2015, respectively, including the R&D tax
credit.
|
|
|
|
4)
|
Reported tax rate on
a GAAP basis includes the impact of tax expense of $440 million
associated with a one-time repatriation of 2014 ex-U.S. earnings,
partially offset by favorability of $126 million as a result of the
resolution of various tax positions and adjustment of tax
uncertainties from prior years.
|
Photo -
http://photos.prnewswire.com/prnh/20160127/326626-INFO
Logo - http://photos.prnewswire.com/prnh/20150928/271488LOGO
To view the original version on PR Newswire,
visit:http://www.prnewswire.com/news-releases/abbott-reports-fourth-quarter-2015-results-300211338.html
SOURCE Abbott