Biota Commences Dosing in Vapendavir SPIRITUS Phase 2b Trial
March 03 2015 - 7:00AM

Biota Pharmaceuticals, Inc. (Nasdaq:BOTA) announced today that it
has commenced dosing of patients in its Phase 2b SPIRITUS trial of
vapendavir. The goal of the study is to enroll approximately 150
laboratory-confirmed human rhinovirus (HRV) infected patients with
moderate-to-severe asthma from the United States and multiple
European countries over the next 12 months and to report top-line
data in mid-2016.
"There are no antivirals currently approved for the treatment of
HRV infection, which is a major cause of disease exacerbation among
patients with asthma and COPD. Therefore, the initiation of this
important trial of vapendavir in moderate-to-severe asthmatics is
truly exciting, and based on the positive outcome from the Phase 2
study in mild asthmatics, I am looking forward to the data from
this trial next year," stated Dr. Jonathan Matz,
allergist-immunologist and Principal Investigator of the SPIRITUS
trial.
"The commencement of dosing in our SPIRITUS trial represents a
significant achievement in our ongoing effort to further define the
efficacy and safety profile of vapendavir in patient populations
with respiratory disease, whose disease control is at risk due to
viral respiratory infection," commented Anna Novotney-Barry, Vice
President of Clinical Development at Biota.
The primary endpoint of this multi-center, randomized,
double-blind, placebo-controlled dose-ranging study is the change
from baseline to study day 14 measured by an asthma control
questionnaire (ACQ)-6 total score. The secondary endpoints are
focused on safety and tolerability, lung function assessments such
as forced expiratory volume in one second (FEV1), incidence of
asthma exacerbations, assessments of the severity and duration of
cold symptoms measured by the Wisconsin Upper Respiratory Symptom
Survey-21 (WURSS-21) and virological assessments such as changes in
viral load.
About Human Rhinovirus (HRV) and Respiratory
Disease
While several different viruses are known to cause upper
respiratory tract infections, it is estimated that HRV causes more
than 50% of all cases. Although such infections can be
self-limiting colds in healthy subjects, patients with chronic lung
diseases such as asthma and chronic obstructive pulmonary disease
(COPD) are especially vulnerable to viral upper respiratory
infections, and HRV infections have been closely associated with
acute exacerbations of asthma, emphysema and COPD in susceptible
individuals. Level of disease control is predictive for future
adverse pulmonary risk and significant healthcare costs are
associated with asthma and COPD exacerbations.
About Biota Pharmaceuticals, Inc.
Biota Pharmaceuticals, Inc. is a company focused on the
discovery and development of products to treat serious viral
respiratory infectious diseases. The Company currently has two
late-stage product candidates: (i) laninamivir octanoate, which is
being developed as a one-time, inhaled treatment for influenza A
and B infections; and (ii) vapendavir, a potent, broad spectrum
capsid inhibitor of enteroviruses in development for the treatment
of human rhinovirus infected patients with underlying respiratory
illnesses, such as moderate-to-severe asthma and chronic
obstructive pulmonary disease (COPD). The Company is also
conducting IND-enabling studies with BTA-C585, an orally
bioavailable F protein inhibitor, in development for the treatment
of respiratory syncytial virus infections. For additional
information about the Company, please visit
www.biotapharma.com.
Safe Harbor Statement
This press release contains forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of 1995
that involve known and unknown risks and uncertainties concerning
Biota's business, operations and financial performance. Any
statements that are not of historical facts may be deemed to be
forward-looking statements, including; enrollment of 150
laboratory-confirmed HRV infected asthma patients; the time frame
in which the Company may fully enroll the Phase 2 SPIRITUS clinical
trial of vapendavir; the timing of top-line data from the SPIRITUS
trial, and other cautionary statements contained elsewhere in this
press release and in the Company's Annual Report on Form 10-K for
the year ended June 30, 2014, as filed with the U.S. Securities and
Exchange Commission on September 30, 2014, and the Company's
Quarterly Reports on Form 10-Q for the quarter ended September 30,
2014 and December 31, 2014, as filed with the U.S. Securities and
Exchange Commission on November 7, 2014 and February 6, 2015.
There may be events in the future that the Company is unable to
predict, or over which it has no control, and the Company's
business, financial condition, results of operations and prospects
may change in the future. The Company may not update these
forward-looking statements more frequently than quarterly unless it
has an obligation under U.S. Federal securities laws to do so.
Biota is a registered trademark of Biota Pharmaceuticals,
Inc.
CONTACT: Joseph M. Patti, PhD
President and Chief Executive Officer of Biota
(678) 221-3352
j.patti@biotapharma.com
Sarah McCabe
Stern Investor Relations, Inc.
(267) 909-9237
sarah@sternir.com
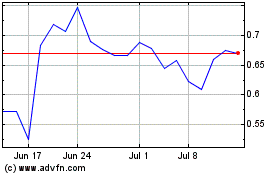
Vaxart (NASDAQ:VXRT)
Historical Stock Chart
From Aug 2024 to Sep 2024
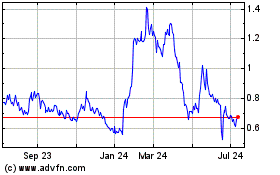
Vaxart (NASDAQ:VXRT)
Historical Stock Chart
From Sep 2023 to Sep 2024