FDA Warns Abbott, Infinant Health About Selling Preterm Infant Probiotic Products
October 26 2023 - 12:55PM
Dow Jones News
By Dean Seal
The Food and Drug Administration has warned Abbott Laboratories
and Infinant Health they are illegally selling probiotic products
that aren't approved for preterm infants.
The regulator said Thursday it is advising healthcare providers
about the possible risks that probiotic products, or ones
containing live bacteria or yeast, post to preterm infants in
hospital settings.
These products can be dangerous for preterm infants, the FDA
said, noting that there has been one infant death reported this
year from a probiotic product being used in a hospital setting to
prevent necrotizing enterocolitis.
The agency said it hasn't approved any probiotic product for use
as a drug or biological product in infants of any age, and is
therefore reaching out to Abbott and Infinant about some of their
products.
The letter to Abbott, sent Tuesday, targets the company's
Similac Probiotic Tri-Blend. The FDA said Abbott has agreed to
discontinue sales of the product and work with the agency on
further corrective actions.
The FDA is warning Infinant about its probiotic product, Evivo
with MCT Oil, being sold to treat or prevent disease in preterm
infants. The product has since been voluntarily recalled and isn't
available in the U.S. anymore.
Write to Dean Seal at dean.seal@wsj.com
(END) Dow Jones Newswires
October 26, 2023 12:40 ET (16:40 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
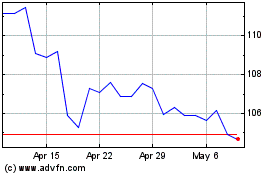
Abbott Laboratories (NYSE:ABT)
Historical Stock Chart
From Jun 2024 to Jul 2024
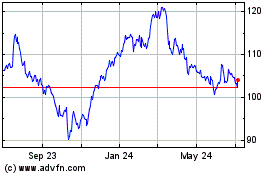
Abbott Laboratories (NYSE:ABT)
Historical Stock Chart
From Jul 2023 to Jul 2024