Immuron’s Technology Platform of Treatments in the Age of the Coronavirus Outbreak
February 03 2020 - 7:30AM

Immuron Limited (ASX: IMC; NASDAQ: IMRN), an Australian
biopharmaceutical company focused on developing and commercializing
oral immunotherapeutics for the treatment of gut mediated diseases,
announced that its commercially available and over-the-counter
gastrointestinal and digestive health supplement Travelan® has
potential to address the ongoing risks of travel to sites endemic
for travelers’ diarrhea, as the focus turns to the ensuing health
crises brought on by the Wuhan coronavirus outbreak and its rapid
spread.
The current crisis highlights the risks
associated with modern day international travel. Parasites,
viruses, food and water contaminated with pathogenic bacteria such
as enterotoxigenic Escherichia coli (E.coli), enteroaggregative
E.coli, Campylobacter, Salmonella, and Shigella are a constant
threat to all international travellers. Over 68 million Americans
traveled abroad in 20141, and approximately 65% of these trips were
to high risk destinations. Among travelers from developed countries
who visit developing countries, 30 to 70% may experience traveler’s
diarrhea. The annual number of international tourist arrivals
worldwide has reached more than 1 billion2.
“We are seeing the dramatic effects that global
travel is having on the potential spread of the newly identified
Wuhan coronavirus,” said Dr. Gary S. Jacob, CEO of Immuron Ltd.
“While the major danger is due to the respiratory repercussions
with this highly infectious agent, there are now reports coming
forth regarding patients presenting with gastrointestinal symptoms
as well. Immuron has been hard at work for a number of years
to develop our hyperimmune bovine colostrum technology to treat
enteric gastrointestinal pathogens responsible for travelers’
diarrhea, and believe that our product has great potential in
further reducing its incidence as increased global travel continues
to expand in the years to come."
|
COMPANY CONTACT:
Gary S. Jacob, Ph.D.Chief Executive OfficerPh: +61
(0)3 9824 5254info@immuron.com |
AUS INVESTOR RELATIONS:
Peter TaylorNWR CommunicationsPh: +61 (0)4 1203
6231peter@nwrcommunications.com.au |
USA INVESTOR RELATIONS:
Dave Gentry - CEO RedChip Companies, Inc. US Ph:
+1 (407) 491 4498 dave@redchip.com |
About
Travelan®Travelan® is an orally administered
passive immunotherapy that prophylactically reduces the likelihood
of contracting travelers’ diarrhea. Travelan® is a highly purified
tabletized preparation of hyper immune bovine antibodies and other
factors, which when taken with meals bind to diarrhea-causing
bacteria and prevent colonization and the pathology associated with
travelers’ diarrhea. In Australia, Travelan® is a listed medicine
on the Australian Register for Therapeutic Goods (AUST L 106709)
and is indicated to reduce the risk of Travelers’ Diarrhea, reduce
the risk of minor gastro-intestinal disorders and is antimicrobial.
In Canada, Travelan® is a licensed natural health product (NPN
80046016) and is indicated to reduce the risk of Travelers’
Diarrhea. In the U.S., Travelan® is sold as a dietary supplement
for digestive tract protection.
About Travelers’
diarrheaTravelers’ Diarrhea is a gastrointestinal
infection with symptoms that include loose, watery (and
occasionally bloody) stools, abdominal cramping, bloating, and
fever, Enteropathogenic bacteria are responsible for most cases,
with enterotoxigenic Escherichia coli (ETEC) playing a dominant
causative role. Campylobacter spp. are also responsible for a
significant proportion of cases. The more serious infections with
Salmonella spp. the bacillary dysentery organisms belonging to
Shigella spp. and Vibrio spp. (the causative agent of cholera) are
often confused with travelers’ diarrhea as they may be contracted
while travelling and initial symptoms are often
indistinguishable.
About ImmuronImmuron Limited
(ASX: IMC, NASDAQ: IMRN), is an Australian biopharmaceutical
company focused on developing and commercializing orally delivered
targeted polyclonal antibodies for the treatment of inflammatory
mediated and infectious diseases. Immuron has a novel and safe
technology platform with one commercial asset generating revenue.
In Australia, Travelan® is a listed medicine on the Australian
Register of Therapeutic Goods (AUST L 106709) and is indicated to
reduce the risk of Travelers’ Diarrhea, reduce the risk of minor
gastro-intestinal disorders and is antimicrobial. In Canada,
Travelan® is a licenced natural health product (NPN 80046016) and
is indicated to reduce the risk of Travelers’ Diarrhea. In the
U.S., Travelan® is sold as a dietary supplement for digestive tract
protection in accordance with section 403 (r)(6) of the Federal
Drug Administration (FDA). Immuron’s lead clinical candidate,
IMM-124E, contains the same active pharmaceutical ingredient as
Travelan®. The company now has plans to develop a U.S. registration
dossier for IMM-124E for Travelers’ Diarrhea. Immuron’s second
clinical-stage asset, IMM-529, targets Clostridium difficile
Infections (CDI), and is in clinical trial development in CDI
patients. These products together with the Company’s other
preclinical immunotherapy pipeline products currently under
development targeting immune-related and infectious diseases are
anticipated to meet pressing needs in the global immunotherapy
market. For more information visit: http://www.immuron.com
FORWARD-LOOKING STATEMENTS:
This press release may contain “forward-looking
statements” within the meaning of Section 27A of the Securities Act
of 1933 and Section 21E of the Securities Exchange Act of 1934,
each as amended. Such statements include, but are not limited
to, any statements relating to our growth strategy and product
development programs and any other statements that are not
historical facts. Forward-looking statements are based on
management’s current expectations and are subject to risks and
uncertainties that could negatively affect our business, operating
results, financial condition and stock value. Factors that could
cause actual results to differ materially from those currently
anticipated include: risks relating to our growth strategy; our
ability to obtain, perform under and maintain financing and
strategic agreements and relationships; risks relating to the
results of research and development activities; risks relating to
the timing of starting and completing clinical trials;
uncertainties relating to preclinical and clinical testing; our
dependence on third-party suppliers; our ability to attract,
integrate and retain key personnel; the early stage of products
under development; our need for substantial additional funds;
government regulation; patent and intellectual property matters;
competition; as well as other risks described in our SEC filings.
We expressly disclaim any obligation or undertaking to release
publicly any updates or revisions to any forward-looking statements
contained herein to reflect any change in our expectations or any
changes in events, conditions or circumstances on which any such
statement is based, except as required by law.
________________________
1 U.S. Department of Commerce, International
Trade Administration, National Travel and Tourism Office. U.S.
Citizen Traffic to Overseas Regions, Canada & Mexico 2014.
Monthly Statistics, U.S. Outbound Travel by World Regions. 2014.
Available at: http://travel.trade.gov/view/m-2014-O-001/index.html.
Accessed June 26, 20152 The World Bank. World Development
Indicators. online tables, global links, 6.14 Travel and Tourism
Available at http://wdi.worldbank.org/table/6.14. Accessed July
1,2015
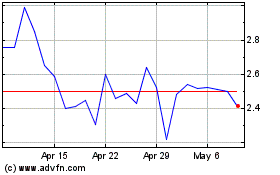
Immuron (NASDAQ:IMRN)
Historical Stock Chart
From Aug 2024 to Sep 2024
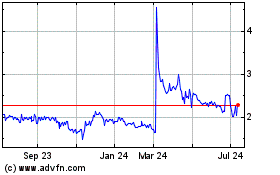
Immuron (NASDAQ:IMRN)
Historical Stock Chart
From Sep 2023 to Sep 2024