Apellis Pharmaceuticals Expands Leadership Team, Appoints Adam Townsend as Chief Commercial Officer
November 20 2018 - 7:00AM

Apellis Pharmaceuticals, Inc., (Nasdaq:APLS) a clinical-stage
biopharmaceutical company focused on the development of novel
therapeutic compounds to treat disease through the inhibition of
the complement system, announced today the appointment of Adam
Townsend as Chief Commercial Officer. Prior to joining Apellis,
Townsend worked at Biogen in Cambridge, MA., with progressive
leadership roles in commercial and corporate development.
“We are incredibly pleased to welcome Adam to Apellis’
leadership team. Adam has a wealth of knowledge and expertise in
bringing novel therapies to market and has worked with a number of
leading organizations in the US, Europe and the UK,” said
Cedric Francois, MD, PhD, founder and chief executive officer of
Apellis. “His experience leading the recent launches of Tecfidera®
and Spinraza® while at Biogen, and his proven success in marketing
and commercializing innovative therapies, will be invaluable
as we prepare to build our commercial franchises in paroxysmal
nocturnal hemoglobinuria (PNH) and geographic atrophy (GA),
followed by other complement-mediated conditions in the
future.”
In his new role, Townsend will be responsible for overseeing the
strategy, development, and execution of all aspects of Apellis’
commercial policies, objectives, and initiatives as the company
expands operations with two clinical programs in Phase 3: DERBY
& OAKS for the treatment of GA and PEGASUS for the treatment of
PNH.
"Apellis’ promising science, dynamic leadership team and
impressive growth trajectory were all very enticing qualities as I
considered the next step in my career,” said Townsend. “I’m
thrilled to join this exciting company and assist in advancing
Apellis’ mission to treat patients impacted with
complement-mediated diseases.”
Prior to Biogen, Townsend held numerous leadership positions in
Europe at EUSA Pharma as General Manager of the UK, Ireland and
Nordic Region and as the Head of Commercial. In addition, Townsend
worked for Amgen in Switzerland as part of its European Marketing
team, driving significant and consistent growth across multiple
geographies and also in the UK where he progressed to be the Head
of Marketing. Adam received a B.Sc. (Hons) in Medical Biochemistry
from the University of London, UK.
About Apellis Apellis Pharmaceuticals, Inc. is
a clinical-stage biopharmaceutical company focused on the
development of novel therapeutic compounds for the treatment of a
broad range of life-threatening or debilitating autoimmune diseases
based upon complement immunotherapy through the inhibition of the
complement system at the level of C3. Apellis is the first company
to advance chronic therapy with a C3 inhibitor into clinical
trials. For additional information about Apellis and APL-2, please
visit http://www.apellis.com. For additional information
regarding our clinical trials,
visit www.apellis.com/clinical-trials.html.
Forward-Looking Statements
Statements in this press release about future expectations, plans
and prospects, as well as any other statements regarding matters
that are not historical facts, may constitute “forward-looking
statements” within the meaning of The Private Securities Litigation
Reform Act of 1995. These statements include, but are not limited
to, statements relating to the implications of preliminary clinical
data. The words “anticipate,” “believe,” “continue,” “could,”
“estimate,” “expect,” “intend,” “may,” “plan,” “potential,”
“predict,” “project,” “should,” “target,” “will,” “would” and
similar expressions are intended to identify forward-looking
statements, although not all forward-looking statements contain
these identifying words. Actual results may differ materially from
those indicated by such forward-looking statements as a result of
various important factors, including: whether dosing in the Phase 3
GA program will resume when anticipated; whether preliminary or
interim results from a clinical trial will be predictive of the
final results of the trial; whether results obtained in preclinical
studies and clinical trials will be indicative of results that will
be generated in future clinical trials; whether APL-2 will
successfully advance through the clinical trial process on a timely
basis, or at all; whether the results of such clinical trials will
warrant regulatory submissions and whether APL-2 will receive
approval from the United States Food and Drug Administration or
equivalent foreign regulatory agencies for GA, PNH or any other
indication; whether, if Apellis’ products receive approval, they
will be successfully distributed and marketed; and other factors
discussed in the “Risk Factors” section of Apellis’ Quarterly
Report on Form 10-Q filed with the Securities and Exchange
Commission on November 13, 2018 and the risks described in other
filings that Apellis may make with the Securities and Exchange
Commission. Any forward-looking statements contained in this press
release speak only as of the date hereof, and Apellis specifically
disclaims any obligation to update any forward-looking statement,
whether as a result of new information, future events or
otherwise.
Media Contact:Nicole
Cliffordnclifford@denterlein.com 617.482.0042
(office)949.533.9295 (mobile)
Investor Contact: Alex Kane
akane@w2ogroup.com 212.301.7218 (office) 929.400.2691 (mobile)
A photo accompanying this announcement is available at
http://www.globenewswire.com/NewsRoom/AttachmentNg/fa700b07-9e50-4e6c-a91d-64832bc58121
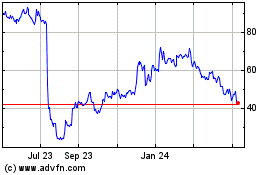
Apellis Pharmaceuticals (NASDAQ:APLS)
Historical Stock Chart
From Aug 2024 to Sep 2024
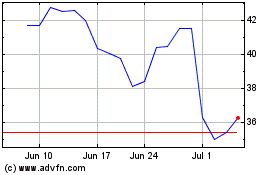
Apellis Pharmaceuticals (NASDAQ:APLS)
Historical Stock Chart
From Sep 2023 to Sep 2024