AVITA Medical (ASX: AVH, OTCQX: AVMXY), a global regenerative
medicine company, today announced that PM360, a leading trade
magazine for marketing decision makers in the pharmaceutical,
biotech, medical device, and diagnostics industries, has named
AVITA as one of the most innovative companies of 2018.
AVITA Medical’s RECELL® Autologous Cell Harvesting System
(RECELL System) was selected as part of PM360’s 7th Annual
Innovations Issue, which is published in December each year. The
RECELL System was approved by the U.S. Food and Drug Administration
(FDA) in September 2018 for the treatment of acute thermal burns in
patients 18 years and older. The RECELL System is used to prepare
Spray-On Skin™ Cells using a small amount of a patient’s own skin,
providing a new way to treat severe burns, while significantly
reducing the amount of donor skin required. PM360 selected the
RECELL System as the product is expected to make an immediate
impact within a market that has not seen major innovation in
decades.
“Our selections represent the companies, offerings, and
strategies that demonstrated what we believe is unique in its
ability to impact the industry,” says Anna Stashower, CEO/Publisher
of PM360. “We hope others in the industry are able to work with
these innovators or use these innovations to better serve patients,
physicians, and other healthcare stakeholders.”
PM360 received hundreds of submissions from across the
healthcare and life sciences industries. The editorial staff of
PM360 evaluated each submission and selected their picks for the
most innovative, regardless of category. All of this year’s
selections can be found at:
www.pm360online.com/pm360-presents-the-2018-innovators.
ABOUT PM360
PM360 is the premier, must-read magazine for marketing decision
makers in the pharmaceutical, biotech, medical device, and
diagnostics industries. Published monthly, PM360 is the only
journal that focuses on delivering the full spectrum of practical
information necessary for product managers and pharmaceutical
marketing professionals to succeed in the complex and highly
regulated healthcare environment.
The journal’s targeted and insightful editorial focuses on
issues that directly impact critical decision making, including:
Planning and implementation of cutting-edge strategies, trends, the
latest technological advances, branding/marketing,
advertising/promotion, patient/professional education, sales,
market research, PR, and leadership. Additionally, the “360” in the
title signifies the span of this critical, how-to info with
personal and career insights for an enjoyable and thought-provoking
read.
By providing the full circle of enriching content, PM360 is
truly an indispensable tool for busy and productive marketing
professionals to stay at the top of their game.
ABOUT AVITA MEDICAL LIMITED
AVITA Medical is a regenerative medicine company with a
technology platform positioned to address unmet medical needs in
burns, chronic wounds, and aesthetics indications. AVITA Medical’s
patented and proprietary collection and application technology
provides innovative treatment solutions derived from the
regenerative properties of a patient’s own skin. The medical
devices work by preparing a REGENERATIVE EPIDERMAL SUSPENSION™
(RES™), an autologous suspension comprised of the patient’s skin
cells necessary to regenerate natural healthy epidermis. This
autologous suspension is then sprayed onto the areas of the patient
requiring treatment.
AVITA Medical’s first U.S. product, the RECELL® System, was
approved by the U.S. Food and Drug Administration (FDA) in
September 2018. The RECELL System is indicated for use in the
treatment of acute thermal burns in patients 18 years and older.
The RECELL System is used to prepare Spray-On Skin™ Cells using a
small amount of a patient’s own skin, providing a new way to treat
severe burns, while significantly reducing the amount of donor skin
required. The RECELL System is designed to be used at the point of
care alone or in combination with autografts depending on the depth
of the burn injury. Compelling data from randomized, controlled
clinical trials conducted at major U.S. burn centers and real-world
use in more than 7,000 patients globally, reinforce that the RECELL
System is a significant advancement over the current standard of
care for burn patients and offers benefits in clinical outcomes and
cost savings. Healthcare professionals should read the INSTRUCTIONS
FOR USE - RECELL® Autologous Cell Harvesting Device for a full
description of important safety information including
contraindications, warnings, and precautions.
In international markets outside of Europe, our products are
marketed under the RECELL System brand to promote skin healing in a
wide range of applications including burns, chronic wounds, and
aesthetics. The RECELL System is TGA-registered in Australia,
CFDA-cleared in China, and received CE-mark approval in Europe.
To learn more, visit www.avitamedical.com.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This letter includes forward-looking statements. These
forward-looking statements generally can be identified by the use
of words such as “anticipate,” “expect,” “intend,” “could,” “may,”
“will,” “believe,” “estimate,” “look forward,” “forecast,” “goal,”
“target,” “project,” “continue,” “outlook,” “guidance,” “future,”
other words of similar meaning and the use of future dates.
Forward-looking statements in this letter include, but are not
limited to, statements concerning, among other things, our ongoing
clinical trials and product development activities, regulatory
approval of our products, the potential for future growth in our
business, and our ability to achieve our key strategic, operational
and financial goal. Forward-looking statements by their nature
address matters that are, to different degrees, uncertain. Each
forward- looking statement contained in this letter is subject to
risks and uncertainties that could cause actual results to differ
materially from those expressed or implied by such statement.
Applicable risks and uncertainties include, among others, the
timing of regulatory approvals of our products; physician
acceptance, endorsement, and use of our products; failure to
achieve the anticipated benefits from approval of our products; the
effect of regulatory actions; product liability claims; risks
associated with international operations and expansion; and other
business effects, including the effects of industry, economic or
political conditions outside of the company’s control. Investors
should not place considerable reliance on the forward-looking
statements contained in this letter. Investors are encouraged to
read our publicly available filings for a discussion of these and
other risks and uncertainties. The forward-looking statements in
this letter speak only as of the date of this release, and we
undertake no obligation to update or revise any of these
statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20181219005936/en/
Media: Monsoon Communications Sarah Kemter Phone:
+61 (0)3 9620 3333 Mobile: +61 (0)407 162 530
sarahk@monsoon.com.au
Investors: Westwicke Partners Caroline Corner
Phone: +1-415-202-5678 caroline.corner@westwicke.com
AVITA Medical Ltd Dale A. Sander Chief Financial Officer
Phone: +1-661-367-9178 dsander@avitamedical.com
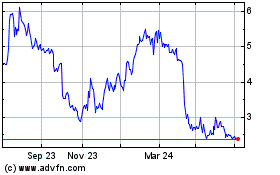
AVITA Medical (ASX:AVH)
Historical Stock Chart
From Mar 2024 to Apr 2024
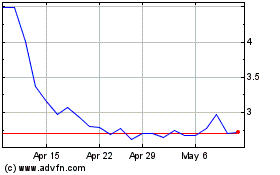
AVITA Medical (ASX:AVH)
Historical Stock Chart
From Apr 2023 to Apr 2024