SWIFTWATER, Pa., July 26, 2016 /PRNewswire/ -- Sanofi
Pasteur, the vaccines division of Sanofi (EURONEXT: SAN and NYSE:
SNY), announced today that its first doses of Fluzone®
(Influenza Vaccine) for the 2016-2017 influenza ("flu") season have
been released by the U.S. Food and Drug Administration (FDA) for
shipment. This represents the first of more than 65 million total
doses of seasonal influenza vaccine manufactured by Sanofi Pasteur
that will be delivered to U.S. health care providers and pharmacies
beginning in July and continuing throughout the remainder of the
year. Sanofi Pasteur plans to increase its supply to respond to the
shifting pediatric public health needs.
Experience the interactive Multimedia News Release here:
http://www.multivu.com/players/English/7842051-sanofi-pasteur-influenza-vaccine/
Seasonal influenza activity typically occurs between October and
May and peaks between December and February. However, influenza
activity peaked noticeably late last season occurring in early
March 2016.1 Influenza
seasons are always unpredictable as new influenza strains emerge
and strain activity fluctuates throughout the year, making timely
vaccination even more important to help protect against the virus,
especially for seniors, young children and infants six months of
age and older.
The Centers for Disease Control and Prevention (CDC) recommends
annual influenza vaccination for everyone six months of age and
older, with rare exception, and recommends receiving the vaccine as
soon as it is available to help with prevention even before the
season begins. In the 2015-2016 influenza season, 50 percent of
seniors who were vaccinated received Fluzone High-Dose vaccine, and
this number is expected to rise to 60 percent in the coming
season.
"There is general awareness of influenza among the public
given its widespread prevalence," said David P. Greenberg, M.D., Associate Vice
President and Regional Medical Head North America, Sanofi Pasteur.
"What is not well known is that influenza can be
life-threatening and have a lasting health impact, especially for
the most vulnerable populations. In the 2014-2015 season, the CDC
estimated 40 million flu illnesses, 19 million flu-associated
medical visits and almost 1 million flu-associated
hospitalizations."2
As the single largest provider of influenza vaccine to
the United States, Sanofi Pasteur
provides a wide range of influenza vaccine options. The Fluzone
vaccine portfolio meets immunization needs across the entire
lifespan, from children as young as six months of age through
adults 65 years of age and older, and includes the following
products:
- Fluzone High-Dose vaccine, the first influenza vaccine
designed specifically for people 65 years of age and older, is an
inactivated influenza vaccine.3 Fluzone High-Dose
vaccine is the only influenza vaccine for seniors that the U.S.
Food and Drug Administration (FDA) has approved as having
demonstrated superior efficacy against influenza compared with
Fluzone vaccine. It contains four times the antigen content of
Fluzone vaccine to help promote a stronger immune response to
influenza.4 Clinical data published in The New
England Journal of Medicine demonstrated that Fluzone High-Dose
vaccine was 24.2 percent more effective than Fluzone vaccine in
preventing laboratory-confirmed influenza caused by any influenza
viral type or subtype in association with influenza-like illness,
in adults 65 years of age and older.4 A secondary
endpoint demonstrated Fluzone High-Dose vaccine was 51.1 percent
more effective than Fluzone vaccine against influenza caused by
viral strains antigenically similar to those contained in the
vaccine using a modified CDC influenza-like illness
definition.4
- Fluzone Quadrivalent vaccine helps protect against four
influenza strains (two A strains and two B strains). The influenza
B strain is associated with substantial hospitalization and
mortality, especially in children and young adults. In fact, on
average, over multiple recent seasons, 34 percent of
influenza-related deaths in children up to 18 years of age were due
to influenza B.5 Fluzone Quadrivalent vaccine is the
first four-strain influenza vaccine licensed for use in people six
months of age and older.
- Fluzone Intradermal Quadrivalent vaccine was licensed by
the U.S. Food and Drug Administration (FDA) in 2014 for adults 18
through 64 years of age. Fluzone Intradermal Quadrivalent vaccine
is administered directly into the skin using a 90 percent smaller,
1.5 mm microneedle and offers four-strain protection that is
convenient, efficient, and easy to use, allowing for streamlined
administration by health care providers.
"Sanofi Pasteur is at the forefront of influenza vaccine
innovation," said Dr. Greenberg. "Our legacy for researching
and manufacturing influenza vaccines has existed for nearly 70
years at our Swiftwater-based
facility, demonstrating our experience and commitment to this
important public health need. We are dedicated to continuously
improving our influenza vaccine options to ultimately help protect
against influenza strains, including drifted strains that may occur
in the future."
To address recommendations by the Advisory Committee on
Immunization Practices (ACIP), Sanofi Pasteur has increased
production of Fluzone Quadrivalent vaccine where possible. Sanofi
Pasteur is the sole provider of injectable influenza vaccine for
pediatric patients six months through 35 months of age.
Health care providers who placed reservations with Sanofi
Pasteur should expect to receive initial shipments between late
July and early August to support fall immunization campaigns.
Health care providers wishing to reserve vaccine can do so by
visiting www.vaccineshoppe.com or by calling 1-800-VACCINE
(1-800-822-2463). Members of the public seeking a specific vaccine
option, such as Fluzone High-Dose vaccine, Fluzone Intradermal
Quadrivalent vaccine, or Fluzone Quadrivalent vaccine, can search
for local providers at www.Fluzone.com.
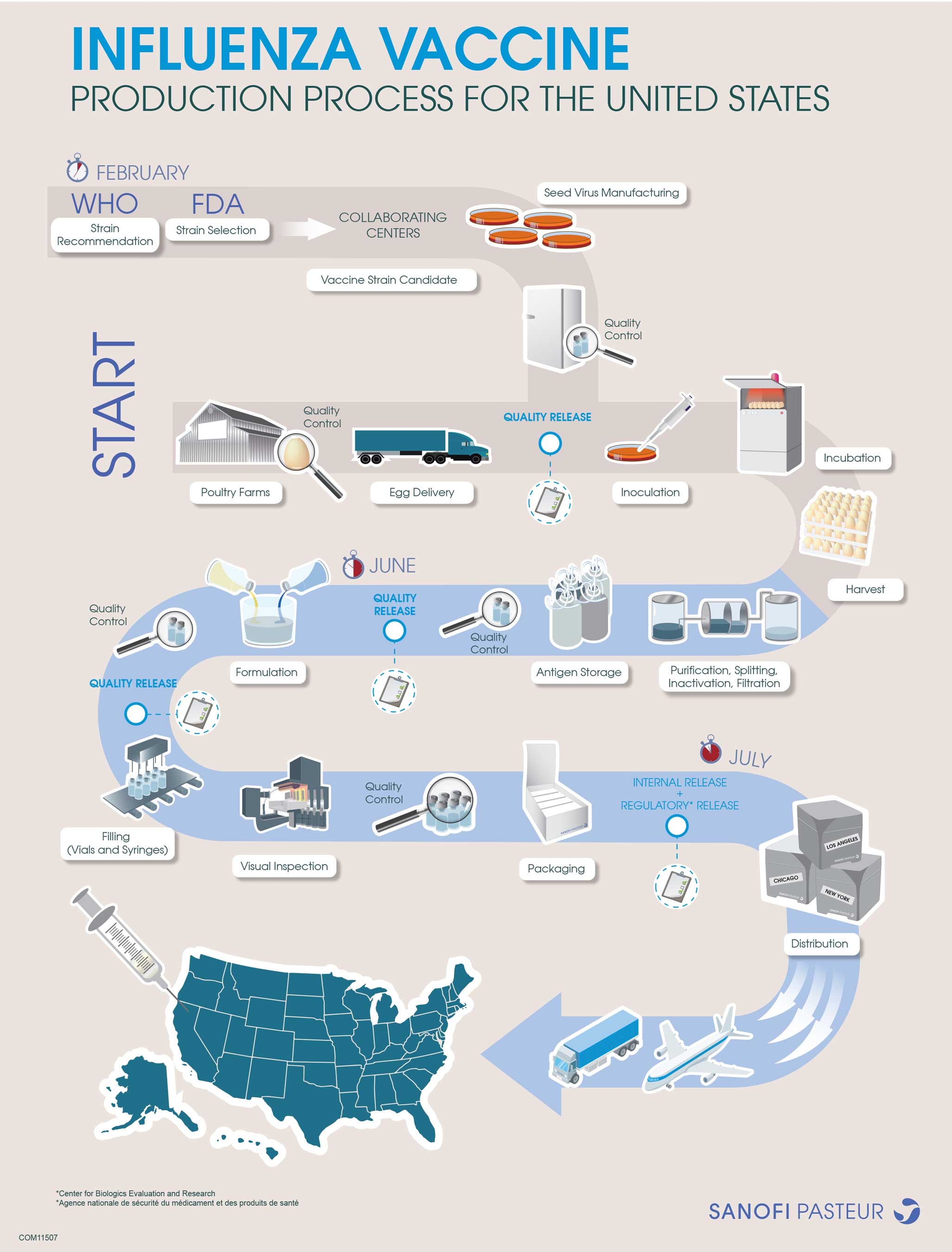
About Sanofi Pasteur Influenza Vaccine Manufacturing
As the world leader in the research, development and manufacturing
of seasonal influenza vaccines, Sanofi Pasteur uses the most
innovative technology and processes. For more information about
Sanofi Pasteur's influenza vaccine manufacturing process, please
view the following infographic ("Production Process for the United
States Infographic") and watch a video that provides a glimpse into
the Sanofi Pasteur production facilities.
About Influenza
Influenza is a serious respiratory
illness that is easily spread and can lead to severe complications,
even death. Each year, one out of every five Americans gets the flu
and, on average, more than 200,000 people are hospitalized from
influenza-related complications.6 Influenza seasons are
unpredictable with flu-related deaths ranging from 3000 to as high
as 49,000 people.7 Combined with pneumonia, influenza is
one of the top ten leading causes of death in the United States.8 Vaccination is
safe and effective and the best way to help prevent influenza and
its complications.
Children six months through eight years of age may require two
doses of vaccine for the 2016-2017 influenza season. Parents should
consult their health care provider about the number of doses of
influenza vaccine their child should receive.
Individuals who are not immunized early in the season will still
have time do so as the influenza vaccination is beneficial
throughout the season, even into the spring, as long as influenza
viruses are still in circulation.9
About Fluzone Vaccines
Indication
Fluzone Quadrivalent, Fluzone Intradermal
Quadrivalent, and Fluzone High-Dose vaccines are given to help
prevent influenza disease caused by influenza A and B strains
contained in each vaccine.
Fluzone Quadrivalent vaccine is given to people 6 months of age
and older. Fluzone Intradermal Quadrivalent vaccine is given to
people 18 through 64 years of age. Fluzone High-Dose vaccine is
given to people 65 years of age and older.
Safety Information
Side effects to Fluzone
Quadrivalent, Fluzone Intradermal Quadrivalent, and Fluzone
High-Dose vaccines include pain and swelling at the injection site
(also itching in adults receiving Fluzone Intradermal Quadrivalent
vaccine); muscle aches, fatigue, headache, and fever (also
irritability, abnormal crying, drowsiness, appetite loss, and
vomiting in young children receiving Fluzone Quadrivalent vaccine).
Itching, redness, swelling, and firmness at the injection site
occurred more frequently with Fluzone Intradermal vaccine
(containing 3 influenza strains) than with Fluzone vaccine. Other
side effects may occur.
Fluzone Quadrivalent, Fluzone Intradermal Quadrivalent, and
Fluzone High-Dose, vaccines should not be administered to anyone
with a severe allergic reaction (e.g., anaphylaxis) to any vaccine
component, including eggs, egg products, or thimerosal (the
multidose vial is the only presentation containing thimerosal), or
to a previous dose of any influenza vaccine.
Tell the doctor if you/your child has ever experienced
Guillain-Barré syndrome (severe muscle weakness) after a previous
dose of influenza vaccine. If you notice any other problems or
symptoms following vaccination, please contact your health care
professional immediately. Vaccination with Fluzone Quadrivalent,
Fluzone Intradermal Quadrivalent, or Fluzone High-Dose vaccine may
not protect all individuals.
For more information about Fluzone Quadrivalent, Fluzone
Intradermal Quadrivalent, or Fluzone High-Dose vaccine, talk to
your health care professional and see complete Patient
Information.
About Sanofi
Sanofi, a global healthcare leader,
discovers, develops and distributes therapeutic solutions focused
on patients' needs. Sanofi has core strengths in diabetes
solutions, human vaccines, innovative drugs, consumer healthcare,
emerging markets, animal health and Genzyme. Sanofi is listed in
Paris (EURONEXT: SAN) and in
New York (NYSE: SNY).
Sanofi Pasteur, the vaccines division of Sanofi, provides more
than 1 billion doses of vaccine each year, making it possible to
immunize more than 500 million people across the globe. A world
leader in the vaccine industry, Sanofi Pasteur produces a portfolio
of high quality vaccines that matches its areas of expertise and
meets public-health demand. The company's heritage, to create
vaccines that protect life, dates back more than a century. Sanofi
Pasteur is the largest company entirely dedicated to vaccines.
Every day, the company invests more than EUR
1 million in research and development. For more information,
please visit: www.sanofipasteur.com or www.sanofipasteur.us
Forward Looking Statements
This press
release contains forward-looking statements as defined in the
Private Securities Litigation Reform Act of 1995, as amended.
Forward-looking statements are statements that are not historical
facts. These statements include projections and estimates and their
underlying assumptions, statements regarding plans, objectives,
intentions and expectations with respect to future financial
results, events, operations, services, product development and
potential, and statements regarding future performance.
Forward-looking statements are generally identified by the words
"expects", "anticipates", "believes", "intends", "estimates",
"plans" and similar expressions. Although Sanofi's management
believes that the expectations reflected in such forward-looking
statements are reasonable, investors are cautioned that
forward-looking information and statements are subject to various
risks and uncertainties, many of which are difficult to predict and
generally beyond the control of Sanofi, that could cause actual
results and developments to differ materially from those expressed
in, or implied or projected by, the forward-looking information and
statements. These risks and uncertainties include among other
things, the uncertainties inherent in research and development,
future clinical data and analysis, including post marketing,
decisions by regulatory authorities, such as the FDA or the EMA,
regarding whether and when to approve any drug, device or
biological application that may be filed for any such product
candidates as well as their decisions regarding labelling and other
matters that could affect the availability or commercial potential
of such product candidates, the absence of guarantee that the
product candidates if approved will be commercially successful, the
future approval and commercial success of therapeutic alternatives,
the Group's ability to benefit from external growth opportunities,
trends in exchange rates and prevailing interest rates, the impact
of cost containment initiatives and subsequent changes thereto, the
average number of shares outstanding as well as those discussed or
identified in the public filings with the SEC and the AMF made by
Sanofi, including those listed under "Risk Factors" and "Cautionary
Statement Regarding Forward-Looking Statements" in Sanofi's annual
report on Form 20-F for the year ended December 31, 2015. Other than as required by
applicable law, Sanofi does not undertake any obligation to update
or revise any forward-looking information or
statements.
1 Centers for Disease Control and Prevention
(CDC). What You Should Know for the 2015-2016 Influenza Season.
http://www.cdc.gov/flu/about/season/flu-season-2015-2016.htm.
Accessed May 9, 2016.
2 CDC. What You Should Know for the 2014-2015 Influenza
Season. http://www.cdc.gov/flu/pastseasons/1415season.htm. Accessed
May 12, 2016.
3 Fluzone High-Dose vaccine [Prescribing Information].
Swiftwater, PA: Sanofi Pasteur
Inc.; 2015.
4 DiazGranados CA, Dunning AJ, Kimmel M, et al. Efficacy
of high-dose versus standard-dose influenza vaccine in older
adults. N Engl J Med. 2014;371:635-645.
5 Ambrose CS, Levin MJ. The rationale for quadrivalent
influenza vaccine. Hum Vaccin Immunother. 2012; 8:81-88
6 CDC. Seasonal Influenza-Associated Hospitalizations in
the United States.
http://www.cdc.gov/flu/about/qa/hospital.htm. Accessed May 12, 2016.
7 CDC. Estimating Seasonal Influenza-Associated Deaths
in the United States: CDC Study
Confirms Variability of Flu.
http://www.cdc.gov/flu/about/disease/us_flu-related_deaths.htm.
Accessed May 12, 2016.
8 Bastian AB, Kochanek DK, Murphy SL, et al. National
Vital Statistics Reports. Pg 1.
http://www.cdc.gov/nchs/data/nvsr/nvsr64/nvsr64_02.pdf. Accessed
May 12, 2016.
9 CDC. Key Facts About Seasonal Flu Vaccine.
http://www.cdc.gov/flu/protect/keyfacts.htm. Accessed May 12, 2016.


To view the original version on PR Newswire,
visit:http://www.prnewswire.com/news-releases/sanofi-pasteur-ships-first-of-its-2016-2017-seasonal-influenza-vaccine-doses-in-united-states-300303562.html
SOURCE Sanofi Pasteur