As
filed pursuant to Rule 424(b)(5)
Registration
No. 333-267870
PROSPECTUS
SUPPLEMENT
(To
Prospectus dated October 20, 2022)

740,000
Shares of Common Stock
We
are offering 740,000 shares of our common stock, par value $0.001 per share, at a price of $4.35 per share, to certain investors pursuant
to this prospectus supplement and the accompanying prospectus.
In
a concurrent private placement, we are also issuing to the investors, for no additional consideration, a warrant to purchase one
share of common stock for each share of common stock purchased in this offering. The common warrants have an exercise price of $4.35
and, subject to certain ownership limitations described herein, are exercisable upon issuance and will expire on the date that is five
years following the date of issuance. The warrants and the shares of our common stock issuable upon the exercise of the warrants are
being offered pursuant to the exemptions provided in Section 4(a)(2) under the Securities Act of 1933, as amended (the “Securities
Act”) and Regulation D promulgated thereunder, and are not being offered pursuant to this prospectus supplement and the accompanying
prospectus. There is no established public trading market for the warrants and we do not expect a market to develop. In addition, we
do not intend to list the warrants on the any national securities exchange or any other nationally recognized trading system.
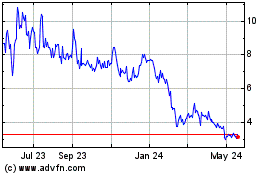
Our
common stock is listed on the Nasdaq Capital Market under the symbol “GTBP.” The last reported sales price of our common
stock on May 21, 2024 was $5.60 per share.
We
have retained Roth Capital Partners, LLC to act as our exclusive placement agent, or the placement agent, in connection with the securities
offered by this prospectus supplement. The placement agent is not purchasing or selling any of the securities we are offering and the
placement agent is not required to arrange the purchase or sale of any specific number of securities or dollar amount. We have agreed
to pay to the placement agent the placement agent fees set forth in the table below, which assumes that we sell all of the securities
offered by this prospectus supplement. See “Plan of Distribution” on page S-7 of this prospectus supplement for more
information regarding these arrangements.
As
of May 21, 2024, the aggregate market value of our common stock held by non-affiliates pursuant to General Instruction I.B.6. of Form
S-3 was $3,261,219, based on 1,254,315 outstanding shares of our common stock held by non-affiliates and a price of $7.80 per share,
the closing sale price of our common stock reported on the Nasdaq Capital Market on May 20, 2024 (a date within 60 days of the date hereof).
During the 12 calendar months prior to and including the date of this prospectus supplement (excluding this offering), we have sold an
aggregate of $0.00 of securities pursuant to General Instruction I.B.6 of Form S-3. As a result, we are eligible to offer and sell up
to an aggregate of $3,261,219 of our securities pursuant to General Instruction I.B.6. of Form S-3.
| | |
Per Share of
Common Stock | | |
Total | |
| Offering price | |
$ | 4.35 | | |
$ | 3,219,000 | |
| Placement agent’s fees(1) | |
$ | 0.261 | | |
$ | 193,140 | |
| Proceeds, before expenses, to us(2) | |
$ | 4.089 | | |
$ | 3,025,860 | |
| (1) |
Includes
a cash fee, equal to 6% of the gross proceeds raised in this offering, to be paid to the placement agent. We have also agreed
to reimburse the placement agent for certain of its offering-related expenses in an aggregate amount up to $50,000. In addition,
we have agreed to issue the placement agent or its designees warrants to purchase up to 88,800 shares of common stock (equal to
6% of the aggregate number of shares of common stock (i) sold in this offering and (ii) issuable upon exercise of the common warrants
sold in the concurrent private placement) at an exercise price of $5.4375 per share, which represents 125% of the offering price
per share of common stock. We refer to these warrants in this prospectus supplement as the “placement agent warrants.”
See “Plan of Distribution” for a complete description of the compensation to be received by the placement agent. |
| (2) |
The amount of the offering
proceeds to us presented in this table does not give effect to any exercise of the common warrants being issued in the concurrent
private placement. |
Investing
in our securities involves a high degree of risk. See “Risk Factors” beginning on page S-4 of this prospectus supplement
and in the documents incorporated by reference in this prospectus supplement and the accompanying prospectus.
None
of the Securities and Exchange Commission or any state securities commission has approved or disapproved of the securities being offered
by this prospectus supplement or accompanying prospectus, or determined if this prospectus supplement or accompanying prospectus is truthful
or complete. Any representation to the contrary is a criminal offense.
Delivery
of the securities offered hereby is expected to be made on or about May 23, 2024, subject to satisfaction of customary closing conditions.
Roth
Capital Partners
May
21, 2024
TABLE
OF CONTENTS
ABOUT
THIS PROSPECTUS SUPPLEMENT
This
prospectus supplement and the accompanying prospectus are part of a registration statement that we filed with the Securities and Exchange
Commission, or the SEC, utilizing a “shelf” registration process. The document is in two parts. The first part is the prospectus
supplement, which describes the specific terms of this offering. The second part is the prospectus, which provides more general information
about securities we may offer from time to time, some of which may not apply to this offering. Generally, when we refer to this “prospectus,”
we are referring to both parts of this document combined. We urge you to carefully read this prospectus supplement and the prospectus,
and the documents incorporated by reference herein and therein, before buying any of the securities being offered under this prospectus
supplement. This prospectus supplement may add or update information contained in the prospectus and the documents incorporated by reference
therein. As a result:
| |
● |
to
the extent that any statement we make in this prospectus supplement is inconsistent with statements made in the accompanying prospectus
or any documents incorporated by reference therein that were filed before the date of this prospectus supplement, the statements
made in this prospectus supplement will be deemed to modify or supersede those made in the accompanying prospectus and such documents
incorporated by reference therein; |
| |
|
|
| |
● |
any
statement contained in a document incorporated by reference in this prospectus supplement or the accompanying prospectus that is
inconsistent with information in a document incorporated by reference herein or therein that we filed on an earlier date will be
deemed to modify and supersede the statement in the document filed on the earlier date; and |
| |
|
|
| |
● |
any
information that we file with the SEC incorporated by reference into this prospectus supplement after the date hereof will automatically
update and supersede the information herein. |
You
should rely only on the information contained or incorporated by reference in this prospectus supplement and the accompanying prospectus,
or contained in any free writing prospectus prepared by or on our behalf. We have not, and the placement agent has not, authorized anyone
to provide you with different information. This prospectus supplement is an offer to sell only the securities offered hereby, and only
under circumstances and in jurisdictions where it is lawful to do so. The distribution of this prospectus supplement and sale of these
securities in certain jurisdictions may be restricted by law. Persons in possession of this prospectus supplement or the accompanying
prospectus are required to inform themselves about and observe any such restrictions. You should assume that the information appearing
in this prospectus supplement, the accompanying prospectus and the documents incorporated by reference in this prospectus supplement
and the accompanying prospectus, and in any free writing prospectus that we have authorized for use in connection with this offering,
is accurate only as of the date of those respective documents, regardless of the date of delivery of this prospectus supplement or the
accompanying prospectus, or the date of sale of any security.
This
prospectus supplement and the information incorporated herein by reference contain summaries of certain provisions contained in some
of the documents described herein, but reference is made to the actual documents for complete information. All of the summaries are qualified
in their entirety by the actual documents. Copies of some of the documents referred to herein have been filed, will be filed or will
be incorporated by reference as exhibits to the registration statement of which this prospectus supplement is a part, and you may obtain
copies of those documents as described below under the heading “Where You Can Find More Information.”
Throughout
this prospectus supplement, the terms “GT Biopharma,” “GTBP,” “we,” “us,” “our,”
“the company” and “our company” refer to GT Biopharma, Inc., a Delaware corporation formerly known as DDI Pharmaceuticals,
Inc., Diagnostic Data, Inc. and Oxis International, Inc., together with our subsidiaries.
CAUTIONARY
NOTICE REGARDING FORWARD-LOOKING STATEMENTS
Some
of the statements in this prospectus supplement are “forward-looking statements” within the meaning of the safe harbor from
liability established by the Private Securities Litigation Reform Act of 1995. Forward-looking statements include statements regarding
our current beliefs, goals and expectations about matters such as our expected financial performance and condition, operating results,
our business strategy and our financing plans. The forward-looking statements in this prospectus supplement are not based on historical
facts, but rather reflect the current expectations of our management concerning future results and events. The forward-looking statements
generally can be identified by the use of terms such as “believe,” “expect,” “anticipate,” “intend,”
“plan,” “foresee,” “may,” “guidance,” “estimate,” “potential,”
“outlook,” “target,” “forecast,” “likely” or other similar words or phrases. Similarly,
statements that describe our objectives, plans or goals are, or may be, forward-looking statements.
Forward-looking
statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements
to be different from any future results, performance and achievements expressed or implied by these statements. We cannot guarantee that
our forward-looking statements will turn out to be correct or that our beliefs and goals will not change. Our actual results could be
very different from and worse than our expectations for various reasons. The risk factors and cautionary language discussed in this prospectus
supplement provide examples of risks, uncertainties and events that may cause actual results to differ materially from the expectations
described by us in such forward-looking statements, including among other things:
| |
● |
our
ability to develop and advance our current product candidates and programs into, and successfully complete, clinical trials; |
| |
|
|
| |
● |
our
financial performance and our ability to effectively manage our anticipated growth; |
| |
|
|
| |
● |
the
period over which we estimate our existing cash and cash equivalents will be sufficient to fund our future operating expenses and
capital expenditure requirements; |
| |
|
|
| |
● |
the
impact of laws and regulations; |
| |
|
|
| |
● |
general
economic conditions; |
| |
|
|
| |
● |
the
effects of the coronavirus on the ongoing disruption of supply chains, the global economy, on the global financial markets and on
our business; |
| |
|
|
| |
● |
the
timing, scope and likelihood of regulatory filings and approvals; |
| |
|
|
| |
● |
our
ability to develop and advance our current product candidates and programs into, and successfully complete, clinical trials; |
| |
|
|
| |
● |
our
continued reliance on third parties to conduct additional clinical trials of our product candidates; |
| |
|
|
| |
● |
our
manufacturing, commercialization, and marketing capabilities and strategy; |
| |
|
|
| |
● |
our
intellectual property position, including the scope of protection we are able to establish and maintain for intellectual property
rights covering product candidates we may develop, including the validity of intellectual property rights held by third parties,
and our ability not to infringe, misappropriate or otherwise violate any third-party intellectual property rights; |
| |
|
|
| |
● |
the
rate and degree of market acceptance and clinical utility of our product candidates we may develop; |
| |
|
|
| |
● |
our
ability to hire additional qualified personnel and attract and retain key employees; and |
| |
|
|
| |
● |
the
result of any future financing efforts. |
These
risks, among others, could cause actual results to differ materially from those implied by the forward-looking statements contained in
this prospectus supplement. You should review carefully all information, including the discussion under “Risk Factors”
in this prospectus supplement and in the documents incorporated by reference herein. Any forward-looking statements in this prospectus
supplement are made only as of the date hereof and, except as may be required by law, we do not have any obligation to publicly update
any forward-looking statements contained in this prospectus supplement to reflect subsequent events or circumstances.
PROSPECTUS
SUPPLEMENT SUMMARY
This
summary is not complete and does not contain all of the information that you should consider before investing in the securities offered
by this prospectus supplement. You should read this summary together with the entire prospectus supplement and the accompanying prospectus
carefully, including “Risk Factors” and our consolidated financial statements and the related notes, before making an investment
decision. See the “Risk Factors” section of this prospectus supplement beginning on page S-4 for a discussion of the
risks involved in investing in our securities.
Overview
We
are a clinical stage biopharmaceutical company focused on the development and commercialization of novel immuno-oncology products based
on our proprietary Tri-specific Killer Engager (TriKE®) fusion protein immune cell engager technology platform. Our TriKE® platform
generates proprietary therapeutics designed to harness and enhance the cancer killing abilities of a patient’s own natural killer
cells, or NK cells. Once bound to an NK cell, our moieties are designed to enhance the NK cell, and precisely direct it to one or more
specifically targeted proteins expressed on a specific type of cancer cell or virus infected cell, resulting in the targeted cell’s
death. TriKE®s can be designed to target any number of tumor antigens on hematologic malignancies or solid tumors and do not require
patient-specific customization.
We
are using our TriKE® platform with the intent to bring to market immuno-oncology products that can treat a range of hematologic malignancies
and solid tumors. The platform is scalable, and we are putting processes in place to be able to produce investigational new drug (IND)
ready moieties in a timely manner after a specific TriKE® conceptual design. Specific drug candidates can then be advanced into the
clinic on our own or through potential collaborations with partnering companies. We believe our TriKE®s may have the ability, if
approved for marketing, to be used as both monotherapy and in combination with other standard-of-care therapies.
We
are also using our TriKE® platform to develop therapeutics useful for the treatment of infectious disease such as for the treatment
of patients infected by the human immunodeficiency virus (HIV). While the use of anti-retroviral drugs has substantially improved the
health and increased the longevity of individuals infected with HIV, these drugs are designed to suppress virus replication to help modulate
progression to acquired immunodeficiency syndrome (AIDS) and to limit further transmission of the virus. Despite the use of anti-retroviral
drugs, infected individuals retain reservoirs of latent HIV-infected cells that, upon cessation of anti-retroviral drug therapy, can
reactivate and re- establish an active HIV infection. For a curative therapy, destruction of these latent HIV infected cells must take
place. The HIV-TriKE® contains the antigen binding fragment (Fab) from a broadly neutralizing antibody targeting the HIV-Env
protein or a protein that binds infected CD4+ T cells. The HIV-TriKE® is designed to target HIV while redirecting NK cell
killing specifically to actively replicating HIV infected cells. The HIV-TriKE® induced NK cell proliferation and demonstrated
the ability in vitro to reactivate and kill HIV-infected T-cells. These findings indicate a potential role for the HIV-TriKE®
in the reactivation and elimination of the latently infected HIV reservoir cells by harnessing the NK cell’s ability to mediate
the antibody-directed cellular cytotoxicity (ADCC).
Our
initial work was conducted in collaboration with the Masonic Cancer Center at the University of Minnesota under a program led by Dr.
Jeffrey Miller, Professor of Medicine, and the Deputy Director at the Center. Dr. Miller is a recognized key opinion leader in the field
of NK cell and IL-15 biology and their therapeutic potential. We have exclusive rights to the TriKE® platform and are
generating additional intellectual property for specific moieties.
Corporate
Information
Our
principal executive offices are located at 8000 Marina Blvd, Suite 100, Brisbane, CA 94005, and our telephone number is (415) 919-4040.
We maintain a website at www.gtbiopharma.com. Information contained on or accessible through our website is not, and should not
be considered, part of, or incorporated by reference into, this prospectus.
The
Offering
| Common Stock offered in this offering |
740,000 shares
of our common stock |
| |
|
| Offering Price |
$4.35 per share and accompanying
common warrant |
| |
|
| Placement Agent Warrants |
We will also issue placement
agent warrants to purchase up to 88,800 shares of common stock (and the shares of common stock issuable upon the exercise of the
placement agent warrants) to the placement agent (or its designees) as part of the compensation payable to the placement agent in
connection with this offering. The placement agent warrants will be in substantially similar form to the common warrants except they
will have an exercise price of $5.4375 per share (which represents 125% of the offering price per share of common stock sold in this
offering) and will expire five years from the commencement of sales of the offering. Please refer to “Plan of Distribution”
for additional information with respect to the placement agent warrants. |
| |
|
| Common stock to be outstanding after this offering |
2,156,651 shares (assuming no exercise of any common warrants and based on 1,416,651
shares outstanding as of May 15, 2024). |
| |
|
| Use of proceeds |
We intend to use the net
proceeds from this offering for general corporate purposes. See “Use of Proceeds” in this prospectus supplement. |
| |
|
| Risk factors |
You should carefully consider
the risk factors described in the section of this prospectus supplement and the accompanying prospectus titled “Risk Factors,”
together with all of the other information included in or incorporated by reference into this prospectus supplement and the accompanying
prospectus, before deciding to purchase our securities. |
| |
|
| Concurrent private placement |
In
a concurrent private placement, we are issuing to purchasers of our common stock in this offering, for no additional consideration,
a common warrant to purchase one share of common stock for each share of common stock purchased in this offering. The common warrants
will be exercisable upon issuance at an exercise price of $4.35 per share and will expire on the date that is 5 years following the
original issuance date. The common warrants and the shares of our common stock issuable upon the exercise of the common warrants
are not being registered under the Securities Act pursuant to the registration statement of which this prospectus supplement and
the accompanying base prospectus form a part and are not being offered pursuant to this prospectus supplement and the accompanying
base prospectus. The common warrants are being offered pursuant to an exemption from the registration requirements of the Securities
Act provided in Section 4(a)(2) of the Securities Act and/or Rule 506(b) of Regulation D thereunder. See “Private Placement
Transaction and Common Warrants” on page S-10 of this prospectus supplement. |
| |
|
| Nasdaq Capital Market symbol |
Our common stock is listed
on the Nasdaq Capital Market under the symbol “GTBP.” |
Unless otherwise stated in this prospectus supplement, the number of shares of our common stock to be outstanding as of the date of this prospectus supplement and after this offering is based on 1,416,651 shares outstanding as of May 15, 2024, and excludes (a) placement agent warrants to purchase up to 88,800 shares of our common stock issuable upon the exercise of warrants to be issued to the placement agent in connection with the offering having an exercise price of $5.4375 per share and (b) the shares of common stock issuable upon exercise of the common warrants being offered by us in the concurrent private placement.
RISK
FACTORS
Investing
in our securities involves a high degree of risk. Our business, financial condition and results of operations could be materially and
adversely affected by any of these risks. If any of these risks occur, the value of the securities we are offering in this prospectus
supplement may decline and you may lose all or part of your investment. Before investing in our securities, you should consider carefully
the risk factors set forth in this prospectus supplement, the accompanying prospectus and in any free writing prospectus that we have
authorized for use in connection with this offering, along with the risk factors described in “Part I. Item 1A. Risk Factors”
in our Annual Report on Form 10-K for the fiscal year ended December 31, 2023 and other filings subsequently made by us with the United
States Securities and Exchange Commission, or the SEC, that are incorporated by reference into this prospectus supplement and the accompanying
prospectus.
Risks
Related to This Offering
Purchasers
of our common stock in this offering will experience immediate and substantial dilution in the book value of their investment.
The
offering price per share in this offering is substantially higher than the net tangible book value per share of our ordinary shares before
giving effect to this offering. Accordingly, purchasers of our securities in this offering will incur immediate dilution of approximately
$0.51 per share, representing the difference between the offering price per share and our as-adjusted net tangible book value
per share as of March 31, 2024. Furthermore, if outstanding options or warrants are exercised, purchasers could experience further
dilution. For more information, including how these amounts were calculated, see “Dilution.”
Our
management will have broad discretion as to the use of the proceeds from this offering, and may not use the proceeds effectively.
Our
management will have broad discretion as to the application of the net proceeds from this offering. Currently, we intend to use the net
proceeds from this offering for general corporate purposes. See “Use of Proceeds.” Purchasers will not have the opportunity,
as part of their investment decision, to assess whether these proceeds are being used appropriately. Our management may use the net proceeds
for corporate purposes that may not improve our financial condition or market value, which could cause the price of our securities to
decline.
We
will need additional capital to conduct our operations and develop our products, and our ability to obtain the necessary funding is uncertain.
We
have used a significant amount of cash since inception to finance the continued development and testing of our product candidates, and
we expect to need substantial additional capital resources to develop our product candidates going forward and launch and commercialize
any product candidates for which we receive regulatory approval.
We
may not be successful in generating and/or maintaining operating cash flow, and the timing of our capital expenditures and other expenditures
may not result in cash sufficient to sustain our operations through the commercialization of our product candidates. If financing is
not sufficient and additional financing is not available or available only on terms that are detrimental to our long-term survival, it
could have a material adverse effect on our ability to continue to function. The timing and degree of any future capital requirements
will depend on many factors, including:
| ● | the
accuracy of the assumptions underlying our estimates for capital needs in 2024 and beyond; |
| ● | scientific
and clinical progress in our research and development programs; |
| ● | the
magnitude and scope of our research and development programs and our ability to establish,
enforce and maintain strategic arrangements for research, development, clinical testing,
manufacturing and marketing; |
| ● | our
progress with pre-clinical development and clinical trials; |
| ● | the
time and costs involved in obtaining regulatory approvals; |
| ● | the
costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing patent
claims; and |
| ● | the
number and type of product candidates that we pursue. |
Additional
financing through strategic collaborations, public or private equity or debt financings or other financing sources may not be available
on acceptable terms, or at all. Additional equity financing could result in significant dilution to our stockholders, and any debt financings
will likely involve covenants restricting our business activities. Further, if we obtain additional funds through arrangements with collaborative
partners, these arrangements may require us to relinquish rights to some of our technologies, product candidates or products that we
would otherwise seek to develop and commercialize on our own.
If
sufficient capital is not available, we may be required to delay, reduce the scope of or eliminate one or more of our research or product
development initiatives, any of which could have a material adverse effect on our financial condition or business prospects.
DILUTION
If
you invest in our securities, your ownership interest will be immediately diluted to the extent of the difference between the offering
price per share and the as-adjusted net tangible book value per share after this offering. Net tangible book value per share represents
our total tangible assets less our total liabilities, divided by the number of shares outstanding. As of March 31, 2024, our net tangible
book value per share was $3.85.
As-adjusted
net tangible book value per share represents our net tangible book value per share after giving effect to the issuance and sale of all
shares offered hereby, and after deducting placement agent fees and estimated offering expenses payable by us in connection with this
offering.
We
determine dilution by subtracting the net tangible book value per share as of March 31, 2024, on an as-adjusted basis for this offering,
from the amount of cash that a new investor paid for a share of common stock. The resulting amount represents an immediate decrease
in net tangible book value of $0.01 per share to existing investors, and immediate dilution in net tangible book value of
$0.51 per share to new investors purchasing securities in this offering at the offering price.
The
following table illustrates this dilution, and is based on, as of March 31, 2024, 1,380,633 shares issued and outstanding and 2,120,633 shares issued and outstanding on an as-adjusted basis as described above.
| Offering price per share | |
| | | |
$ | 4.35 | |
| Historical net tangible book value per share as of March 31, 2024 | |
$ | 3.85 | | |
| | |
| Decrease in net tangible book value per share attributable to this offering | |
$ | 0.01 | | |
| | |
| As-adjusted net tangible book value per share after this offering | |
| | | |
$ | 3.84 | |
| Dilution per share to new investors in this offering | |
| | | |
$ | 0.51 | |
The
information above does not give effect to (i) shares issuable upon the exercise or vesting of various outstanding securities as of May
21, 2024 or (ii) shares issuable upon exercise of the common warrants or placement agent warrants being issued in this offering.
To
the extent that such additional convertible securities are exercised, there will be further dilution to new investors. In addition, we
may seek to raise additional capital due to market conditions or strategic considerations even if we believe we have sufficient funds
for our current or future operating plans. To the extent that additional capital is raised through the sale of equity or convertible
debt securities, the issuance of these securities could result in further dilution to our shareholders.
USE
OF PROCEEDS
We
estimate that the net proceeds from the sale of the common stock in this offering will be approximately $2.8 million, after deducting fees payable to the placement agent and estimated offering expenses
payable by us.
We
intend to use the net proceeds of this offering for general corporate purposes. Our expected use of net proceeds from the offering represents
our current intentions based upon our present plans and business condition. Investors are cautioned, however, that expenditures may vary
substantially from these uses. Investors will be relying on the judgment of our management, who will have broad discretion regarding
the application of the proceeds of this offering. The amounts and timing of our actual expenditures will depend upon numerous factors,
including the amount of cash generated by our operations, the amount of competition and other operational factors. We may find it necessary
or advisable to use portions of the proceeds from this offering for other purposes.
PLAN
OF DISTRIBUTION
Pursuant
to a placement agency agreement dated as of May 21, 2024, we have engaged Roth Capital Partners, LLC, or the placement
agent, to act as our exclusive placement agent in connection with this offering of securities pursuant to this prospectus supplement
and accompanying prospectus. The placement agent is not purchasing or selling any such securities offered by us under this prospectus
supplement, nor is it required to arrange for the purchase and sale of any specific number or dollar amount of such securities, other
than to use its “reasonable best efforts” to arrange for the sale of such securities by us. Therefore, we may not sell all
of the securities being offered. The placement agent may engage sub-agents or selected dealers to assist with this offering.
The
placement agent will have no authority to bind us by virtue of the placement agency agreement. We have entered into a securities
purchase agreement, dated as of May 21, 2024, directly with certain institutional investors, who have agreed to purchase our securities
in this offering. We will only sell to investors who have entered into this securities purchase agreement.
Delivery
of the securities offered hereby is expected to be made on or about May 23, 2024, subject to satisfaction of certain customary closing
conditions.
The
following table shows, both on a per-share and total basis, the offering price, placement agent fees and proceeds, before expenses to
us.
| | |
Per Share of
Common Stock | | |
Total | |
| Offering price | |
$ | 4.35 | | |
$ | 3,219,000 | |
| Placement agent’s fees(1) | |
$ | 0.261 | | |
$ | 193,140 | |
| Proceeds, before expenses, to us(2) | |
$ | 4.089 | | |
$ | 3,025,860 | |
| (1) |
Includes a cash fee, equal
to 6% of the gross proceeds raised in this offering, to be paid to the placement agent. |
| (2) |
The amount of the offering
proceeds to us presented in this table does not give effect to any exercise of the common warrants being issued in the concurrent
private placement. |
We
have agreed to pay the placement agent a cash fee equal to 6% of the gross proceeds raised in this offering. We have also agreed to reimburse
the placement agent at closing for legal and other non-accountable expenses incurred by them in connection with the offering in an aggregate
amount up to $50,000. We estimate the total expenses payable by us for this offering, excluding the placement agent fees and expenses,
will be approximately $200,000.
In
addition, we have agreed to issue to the placement agent as compensation warrants to purchase up to 88,800 shares (equal to 6% of the
aggregate number of shares of common stock (i) sold in this Offering and (ii) issuable upon exercise of the common warrants sold in the
concurrent private placement).
The
placement agent warrants will have substantially the same terms as the common warrants issued to the investors in the concurrent private
placement, except that the placement agent warrants have an exercise price of $5.4375 per share, which represents 125% of the offering
price per share of common stock and will have a term of five years from the commencement of sales of the offering.
Lock-up
Restrictions
Pursuant
to the securities purchase agreement with the purchasers of the shares and common warrants, we have agreed for a period of 60 days following
the closing of this offering not to issue, enter into an agreement to issue or announce the issuance or proposed issuance of shares of
our common stock or any other securities convertible into, or exercisable or exchangeable for, shares of common stock or file any registration
statement or amendment or supplement thereto, subject to limited exceptions. This agreement does not apply to, in addition to certain
customary exceptions, the issuance by us of equity or debt securities pursuant to acquisitions or strategic transactions approved by
a majority of our disinterested directors, where not for the purpose of raising capital, provided that, in each case, such securities
are issued as “restricted securities” (as defined in Rule 144 under the Securities Act), and carry no registration rights
that require or permit the filing of any registration statement in connection therewith during the 60-day lock-up period.
We
have also agreed for a period of one year following the closing date of this offering not to (i) issue or agree to issue equity or debt
securities convertible into, or exercisable or exchangeable for, shares at a conversion price, exercise price or exchange price which
floats with the trading price of our shares or which may be adjusted after issuance upon the occurrence of certain events or (ii) enter
into any agreement, including an equity line of credit, whereby we may issue securities at a future-determined price. This agreement
does not apply to the offer, issuance or sale by us of our shares in an at-the-market offering 120 days after the closing of this offering.
Our
directors and executive officers have also entered into lock-up agreements. Under these agreements, these individuals have agreed, subject
to specified exceptions, not to sell or transfer any shares of common stock or securities convertible, exchangeable or exercisable into,
shares of our common stock during a period ending 60 days after the date of this prospectus supplement.
Regulation
M
The
placement agent may be deemed to be an underwriter within the meaning of Section 2(a)(11) of the Securities Act, and any commissions
received by it and any profit realized on the resale of the securities sold by it while acting as principal might be deemed to be underwriting
discounts or commissions under the Securities Act. As an underwriter, the placement agent would be required to comply with the requirements
of the Securities Act and the Exchange Act, including, without limitation, Rule 415(a)(4) under the Securities Act and Rule 10b-5 and
Regulation M under the Exchange Act. These rules and regulations may limit the timing of purchases and sales of our securities by the
placement agent acting as principal. Under these rules and regulations, the placement agent:
| |
● |
may
not engage in any stabilization activity in connection with our securities; and |
| |
|
|
| |
● |
may
not bid for or purchase any of our securities or attempt to induce any person to purchase any of our securities, other than as permitted
under the Exchange Act, until it has completed its participation in the distribution. |
Indemnification
We
have agreed to indemnify the placement agent against certain liabilities, including certain liabilities arising under the Securities
Act, or to contribute to payments that the placement agent may be required to make for these liabilities.
Determination
of Offering Price
The
offering price of the securities we are offering was negotiated between us, the placement agent and the investors in the offering based
on the trading of our shares prior to the offering, among other things.
Electronic
Offer, Sale and Distribution of Securities
A
prospectus in electronic format may be made available on the websites maintained by the placement agent, if any, participating in this
offering and the placement agent may distribute prospectuses electronically. Other than the prospectus in electronic format, the information
on these websites is not part of this prospectus supplement or the registration statement of which the accompanying prospectus forms
a part, has not been approved or endorsed by us or the placement agent, and should not be relied upon by investors.
Other
Relationships
The
placement agent and its respective affiliates have from to time to time in the past engaged and may in the future engage in investment
banking and other commercial dealings in the ordinary course of business with us or our affiliates, for which they have received or may
receive customary fees and expenses.
Listing
Our
common stock is listed on the Nasdaq Capital Market under the symbol “GTBP.”
DESCRIPTION
OF COMMON STOCK
This
description is a summary and is qualified in its entirety by reference to our certificate of incorporation, as amended, a copy of which
is filed as Exhibit 3.1 to our Current Report on Form 8-K, filed on February 11, 2021, and our amended and restated bylaws, a copy of
which is filed as Exhibit 3.1 to our Current Report on Form 8-K, filed on November 9, 2022.
General
We
are authorized to issue 250,000,000 shares of common stock, par value $0.001 per share. As of May 15, 2024, we had 1,416,651 shares of
common stock outstanding.
Voting
Rights
Holders
of our common stock are entitled to one vote for each share of common stock held of record for the election of directors and on all matters
submitted to a vote of stockholders.
Dividends
Holders
of our common stock are entitled to receive dividends ratably, if any, as may be declared by the board of directors out of legally available
funds, subject to any preferential dividend rights of any preferred stock then outstanding. We do not anticipate that we will pay any
dividends on our common stock in the foreseeable future. Rather, we anticipate that we will retain earnings, if any, for use in the development
of our business.
Liquidation
In
the event of our dissolution, liquidation or winding up, holders of our common stock are entitled to share ratably in our net assets
legally available after the payment of all of our debts and other liabilities, subject to the liquidation preferences of any preferred
stock then outstanding.
Rights
and Preferences
Holders
of our common stock have no preemptive, subscription, redemption or conversion rights. The rights, preferences and privileges of holders
of common stock are subject to, and may be adversely affected by, the rights of the holders of shares of any series of preferred stock
currently outstanding or that we may designate and issue in the future.
PRIVATE
PLACEMENT TRANSACTION AND COMMON WARRANTS
Common
Warrant
In
a concurrent private placement, we are issuing to each of the investors in this offering, for no additional consideration, common
warrants to purchase one share of common stock for each share purchased in this offering.
The
common warrants and the shares of common stock issuable upon the exercise of the common warrants are not being registered under the Securities
Act, are not being offered pursuant to this prospectus supplement and the accompanying prospectus and are being offered pursuant to the
exemption provided in Section 4(a)(2) of the Securities Act and/or Rule 506(b) of Regulation D thereunder. Accordingly, the purchasers
may only sell the shares of common stock issued upon exercise of the common warrants pursuant to an effective registration statement
under the Securities Act covering the resale of those shares, an exemption under Rule 144 under the Securities Act or another applicable
exemption under the Securities Act.
The
following description of our common warrants we are offering is a summary and is qualified in its entirety by reference to the provisions
of the common warrant, which has been provided to the investors in this offering and which will be filed with the SEC as an exhibit to
a Current Report on Form 8-K in connection with this offering and incorporated by reference into the registration statement of which
this prospectus supplement and the accompanying prospectus form a part.
Common
warrants will be issued in certificated form only.
Duration
and Exercise Price
Each
common warrant offered hereby has an initial exercise price per share equal to $4.35. Each common warrant is exercisable for one share
of common stock. The common warrants are exercisable immediately following issuance, and have a term of exercise equal to five years
following the initial exercise date. The exercise price and number of shares issuable upon exercise is subject to appropriate adjustment
in the event of stock dividends, stock splits, reorganizations or similar events affecting our common stock and the exercise price. The
common warrants will be issued in certificated form only.
Exercisability
The
common warrants are exercisable immediately after issuance, at the option of each holder, in whole or in part, by delivering to us a
duly-executed exercise notice accompanied by payment in full for the number of shares purchased upon such exercise (except in the case
of a cashless exercise as discussed below). A holder (together with its affiliates) may not exercise any portion of such holder’s
common warrants to the extent that the holder would own more than 4.99% (or 9.99%, at the holder’s election) of our outstanding
common stock immediately after exercise, except that upon notice from the holder to us, the holder may decrease or increase the limitation
of ownership of outstanding stock after exercising the holder’s common warrants up to 9.99% of the number of shares of our common
stock outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the
terms of the common warrants, provided that any increase in such limitation shall not be effective until 61 days following notice to
us. No fractional shares will be issued in connection with the exercise of a common warrant. In lieu of fractional shares, we will either
pay the holder an amount in cash equal to the fractional amount multiplied by the exercise price or round up to the next whole share.
Cashless
Exercise
If,
at the time a holder exercises its common warrants, a registration statement registering the issuance of the shares of common stock underlying
the common warrants under the Securities Act, is not then effective or available for the resale of such shares, then in lieu of making
the cash payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the holder may
elect instead to receive upon such exercise (either in whole or in part) the net number of shares of common stock determined according
to a formula set forth in the common warrant.
Fundamental
Transactions
In
the event of any fundamental transaction, as described in the common warrants and generally including any merger with or into another
entity, sale of all or substantially all of our assets, tender offer or exchange offer, or reclassification of our shares of common stock,
then upon any subsequent exercise of a common warrant, the holder will have the right to receive as alternative consideration, for each
share of common stock that would have been issuable upon such exercise immediately prior to the occurrence of such fundamental transaction,
the number of shares of common stock of the successor or acquiring corporation or of our company, if it is the surviving corporation,
and any additional consideration receivable upon or as a result of such transaction by a holder of the number of shares of common stock
for which the common warrant is exercisable immediately prior to such event. In addition, upon a fundamental transaction, the holder
will have the right to require us to repurchase its common warrant at its fair value using the Black Scholes option pricing formula in
the common warrants; provided, however, that, if the fundamental transaction is not within our control, including not approved by our
board of directors, then the holder shall only be entitled to receive the same type or form of consideration (and in the same proportion),
at the Black Scholes value of the unexercised portion of the common warrant, that is being offered and paid to the holders of our common
stock in connection with the fundamental transaction.
Transferability
Subject
to applicable laws, a common warrant may be transferred at the option of the holder upon surrender of the common warrant to us together
with the appropriate instruments of transfer.
Exchange
Listing
There
is no trading market available for the common warrants on any securities exchange or nationally recognized trading system. We do not
intend to list the common warrants on any securities exchange or nationally recognized trading system. Without an active market, the
liquidity of the common warrants will be limited.
Right
as a Shareholder
Except
as otherwise provided in the common warrants or by virtue of such holder’s ownership of our common stock, the holders of the common
warrants do not have the rights or privileges of holders of our common stock, including any voting rights, until they exercise their
common warrants.
Placement
Agent Warrant
The
following description of our placement agent warrants we are offering is a summary and is qualified in its entirety by reference to the
provisions of the placement agent warrant, which will be filed with the SEC as an exhibit to a Current Report on Form 8-K in connection
with this offering and incorporated by reference into the registration statement of which this prospectus supplement and the accompanying
prospectus form a part. Prospective investors should carefully review the terms and provisions of the form of placement agent warrant
for a complete description of the terms and conditions of the placement agent warrants.
The
placement agent warrants are substantially similar to the common warrants, except that each placement agent warrant will have an exercise
price of $5.4375 per share (which represents 125% of the offering price per share of common stock sold in this offering) and will expire
five years from the commencement of sales of the offering.
MATERIAL
TAX CONSIDERATIONS
The
following is a description of the material U.S. federal income tax consequences relating to the acquisition, ownership and disposition
of our common stock and (our “shares”) by a U.S. Holder (as defined below). This description addresses only the U.S. federal
income tax consequences to U.S. Holders that will hold our securities as capital assets. This description does not address tax considerations
applicable to U.S. Holders that may be subject to special tax rules, including, without limitation:
| |
● |
banks,
financial institutions or insurance companies; |
| |
|
|
| |
● |
real
estate investment trusts, regulated investment companies or grantor trusts; |
| |
|
|
| |
● |
brokers,
dealers or traders in securities, commodities or currencies; |
| |
|
|
| |
● |
tax-exempt
entities or organizations, including an “individual retirement account” or “Roth IRA” as defined in Section
408 or 408A of the Code (as defined below), respectively; |
| |
|
|
| |
● |
U.S.
expatriates and certain former citizens or long-term residents of the United States; |
| |
|
|
| |
● |
persons that received our
securities as compensation for the performance of services; |
| |
|
|
| |
● |
persons that will hold
our securities as part of a “hedging,” “integrated” or “conversion” transaction or as a position
in a “straddle” for U.S. federal income tax purposes; |
| |
|
|
| |
● |
partnerships (including
entities classified as partnerships for U.S. federal income tax purposes) or other pass-through entities, or holders that will hold
our shares through such an entity; |
| |
|
|
| |
● |
S
corporations; |
| |
|
|
| |
● |
holders
that acquire shares of our common stock as a result of holding or owning our preferred shares; |
| |
|
|
| |
● |
holders
whose “functional currency” is not the U.S. Dollar; |
| |
|
|
| |
● |
persons
deemed to sell our shares under the constructive sale provisions of the Code (as defined below); or |
| |
|
|
| |
● |
persons
subject to special tax accounting rules as a result of any item of gross income with respect to the securities being taken into account
in an applicable financial statement; or. |
| |
|
|
| |
● |
holders that own directly,
indirectly or through attribution 10% or more of the voting power or value of our shares. |
Moreover,
this description does not address the U.S. federal estate, gift or alternative minimum tax consequences, or any state, local or non-U.S.
tax consequences, of the acquisition, ownership and disposition of our shares.
This
description is based on the U.S. Internal Revenue Code of 1986, as amended, or the Code, existing and proposed U.S. Treasury Regulations
and judicial and administrative interpretations thereof, in each case as in effect on the date hereof. All of the foregoing is subject
to change, which change could apply retroactively and could affect the tax consequences described below. There can be no assurance that
the U.S. Internal Revenue Service, or the IRS, will not take a different position concerning the tax consequences of the acquisition,
ownership and disposition of the securities or that such a position would not be sustained. Holders should consult their own tax advisors
concerning the U.S. federal, state, local and non-U.S. tax consequences of purchasing, owning and disposing of our shares in their particular
circumstances.
For
purposes of this description, a “U.S. Holder” is a beneficial owner of our shares that, for U.S. federal income tax purposes,
is:
| |
● |
An
individual citizen or resident of the United States; |
| |
|
|
| |
● |
a
corporation created or organized in or under the laws of the United States or any state thereof, including the District of Columbia; |
| |
|
|
| |
● |
an
estate, the income of which is subject to U.S. federal income taxation regardless of its source; or |
| |
|
|
| |
● |
a
trust if such trust has validly elected to be treated as a United States person for U.S. federal income tax purposes or if (1) a
court within the United States is able to exercise primary supervision over its administration and (2) one or more United States
persons have the authority to control all of the substantial decisions of such trust. |
If
a partnership (or any other entity treated as a partnership for U.S. federal income tax purposes) holds our shares, the tax treatment
of a partner in such partnership will generally depend on the status of the partner and the activities of the partnership. Such a partner
or partnership should consult its tax advisor as to the particular U.S. federal income tax consequences of acquiring, owning and disposing
of our shares in its particular circumstance.
You
should consult your tax advisor with respect to the U.S. federal, state, local and non-U.S. tax consequences of acquiring, owning and
disposing of our shares.
Medicare
Tax
Certain
U.S. Holders that are individuals, estates or trusts are subject to a 3.8% tax on all or a portion of their “net investment income,”
which may include all or a portion of their dividend income and net gains from the disposition of securities. Each U.S. Holder that is
an individual, estate or trust is urged to consult its tax advisors regarding the applicability of the Medicare tax to its income and
gains in respect of its investment in our shares.
Information
Reporting and Backup Withholding
Information
reporting requirements generally will apply to payments of dividends (including constructive dividends) on, and to proceeds from the
sale or other disposition of, securities paid by us to a U.S. Holder, unless such holder is an exempt recipient, such as a corporation.
Backup withholding will apply to those payments if the U.S. Holder fails to furnish its correct taxpayer identification number or otherwise
fails to comply with applicable requirements to establish an exemption.
Backup
withholding is not an additional tax. Rather, any amounts withheld under the backup withholding rules will be allowed as a credit against
the beneficial owner’s U.S. federal income tax liability, if any, and any excess amounts withheld under the backup withholding
rules may be refunded, provided that the required information is timely furnished to the IRS. U.S. Holders should consult their own tax
advisors regarding their qualification for exemption from information reporting and backup withholding and the procedure for obtaining
such exemption.
The
above description is not intended to constitute a complete analysis of all tax consequences relating to the acquisition, ownership and
disposition of our shares. You should consult your tax advisor concerning the tax consequences of the acquisition, ownership and disposition
of our securities in your particular situation.
LEGAL
MATTERS
The
validity of the shares being offered by this prospectus supplement and other legal matters concerning this offering and the validity
of the securities offered by this prospectus supplement, will be passed upon by Baker & McKenzie LLP, Dallas, Texas. Certain legal
matters in connection with this offering with respect to U.S. law will be passed upon for the placement agent by Ellenoff, Grossman &
Schole LLP, New York, New York.
EXPERTS
The
financial statements of GT Biopharma, Inc. incorporated by reference in this prospectus supplement have been audited by Weinberg &
Company, P.A., an independent registered public accounting firm, as stated in their report. Such financial statements are incorporated
by reference in reliance upon the report of such firm given, upon their authority as experts in accounting and auditing.
WHERE
YOU CAN FIND MORE INFORMATION
This
prospectus supplement is part of a registration statement we filed with the SEC. This prospectus supplement does not contain all of the
information set forth in the registration statement and the exhibits to the registration statement. For further information with respect
to us and the securities we are offering under this prospectus supplement, we refer you to the registration statement and the exhibits
and schedules filed as a part of the registration statement. You should rely only on the information contained in this prospectus supplement
or incorporated by reference in this prospectus supplement. We have not authorized anyone else to provide you with different information.
We are not making an offer of these securities in any state where the offer is not permitted. You should not assume that the information
in this prospectus supplement is accurate as of any date other than the date on the front page of this prospectus supplement, regardless
of the time of delivery of this prospectus supplement or any sale of the securities offered by this prospectus supplement.
We
file annual, quarterly and current reports, proxy statements and other information with the SEC. The SEC maintains a website at www.sec.gov
that contains reports, proxy and information statements, and other information regarding issuers, such as us, that file electronically
with the SEC.
Copies
of certain information filed by us with the SEC are also available on our website at www.gtbiopharma.com. Information contained
in or accessible through our website does not constitute a part of this prospectus and is not incorporated by reference in this prospectus
supplement. We have included our website address as an inactive textual reference only.
INCORPORATION
OF CERTAIN INFORMATION BY REFERENCE
The
SEC allows us to “incorporate by reference” information into this prospectus supplement, which means that we can disclose
important information to you by referring you to another document filed separately with the SEC. The information that we incorporate
by reference is an important part of this prospectus supplement and information that we file later with the SEC will automatically update
and supersede this information.
The
following documents are incorporated by reference into this document:
| |
● |
our
Annual Report on Form 10-K for the fiscal year ended December 31, 2023, filed with the SEC on March 26, 2024; |
| |
|
|
| |
● |
our
Definitive Proxy Statement on Schedule 14A (to the extent incorporated by reference into our Annual Report on Form 10-K), filed with
the SEC on April 29, 2024; |
| |
|
|
| |
● |
Our
Quarterly Report on Form 10-Q for the quarter ended March 31, 2024, filed with the SEC on May 15, 2024; |
| |
|
|
| |
● |
our
Current Reports on Form 8-K filed with the SEC on February 1, 2024 and April 30, 2024, to the extent the information in such reports
is filed and not furnished; and |
| |
|
|
| |
● |
The
description of our common stock contained in our Registration Statement on Form 8A-12B filed with the SEC on February 8, 2021. |
We
also incorporate by reference into this prospectus supplement all documents (other than current reports furnished under Item 2.02 or
Item 7.01 of Form 8-K and exhibits filed on such form that are related to such items) that are filed by us with the SEC pursuant to Sections
13(a), 13(c), 14 or 15(d) of the Exchange Act (i) after the date of the initial filing of the registration statement of which this prospectus
supplement forms a part and prior to effectiveness of the registration statement, or (ii) after the date of this prospectus supplement
but prior to the termination of the offering. These documents include periodic reports, such as Annual Reports on Form 10-K, Quarterly
Reports on Form 10-Q and Current Reports on Form 8-K, as well as proxy statements.
We
will provide to each person, including any beneficial owner, to whom a prospectus is delivered, without charge upon written or oral request,
a copy of any or all of the documents that are incorporated by reference into this prospectus supplement but not delivered with the prospectus
supplement, including exhibits that are specifically incorporated by reference into such documents. You should direct any requests for
documents to GT Biopharma, Inc., Attn: Corporate Secretary, 8000 Marina Blvd, Suite 100, Brisbane, CA 94005, and our telephone number
is (415) 919-4040.
Any
statement contained in this prospectus supplement or contained in a document incorporated or deemed to be incorporated by reference into
this prospectus supplement will be deemed to be modified or superseded to the extent that a statement contained in this prospectus supplement
or any document deemed to be incorporated by reference into this prospectus supplement, modifies or supersedes such statement.
PROSPECTUS

$150,000,000
Common
Stock
Warrants
From
time to time, we may offer and sell up to $150,000,000 of any combination of the securities described in this prospectus in one
or more offerings. We may also offer securities as may be issuable upon conversion, redemption, repurchase, exchange or exercise of any
securities registered hereunder, including any applicable anti-dilution provisions.
This
prospectus provides a general description of the securities we may offer. Each time we offer securities, we will provide specific terms
of the securities offered in a supplement to this prospectus. We may also authorize one or more free writing prospectuses to be provided
to you in connection with these offerings. The prospectus supplement and any related free writing prospectus may also add, update or
change information contained in this prospectus. You should carefully read this prospectus, the applicable prospectus supplement and
any related free writing prospectus, as well as any documents incorporated by reference, before you invest in any of the securities being
offered.
We
are a “smaller reporting company” under the federal securities laws and, as such, are subject to reduced public company reporting
requirements.
This
prospectus may not be used to consummate a sale of any securities unless accompanied by a prospectus supplement.
We
will sell these securities directly to investors, through agents designated from time to time or to or through underwriters or dealers,
on a continuous or delayed basis. For additional information on the methods of sale, you should refer to the section entitled “Plan
of Distribution” in this prospectus and in the applicable prospectus supplement. If any agents or underwriters are involved in
the sale of any securities with respect to which this prospectus is being delivered, the names of such agents or underwriters and any
applicable fees, commissions, discounts or over-allotment options will be set forth in a prospectus supplement. The price to the public
of such securities and the net proceeds we expect to receive from such sale will also be
set
forth in a prospectus supplement.
Our
common stock is traded on Nasdaq Capital Market under the symbol “GTBP.” On October 12, 2022, the last reported sale
price of our common stock was $1.77 per share. The applicable prospectus supplement will contain information, where applicable,
as to any other listing on Nasdaq Capital Market or any securities market or other exchange of the securities, if any, covered by the
prospectus supplement.
Investing
in our securities involves a high degree of risk. You should review carefully the risks and uncertainties described under the heading
“Risk Factors” on page 3 of this prospectus and in any applicable prospectus supplement and related free writing prospectus
we have authorized for use in connection with a specific offering, and under similar headings in the other documents that are incorporated
by reference into this prospectus.
NEITHER
THE SECURITIES AND EXCHANGE COMMISSION NOR ANY STATE SECURITIES COMMISSION HAS APPROVED OR DISAPPROVED OF THESE SECURITIES OR DETERMINED
IF THIS PROSPECTUS IS TRUTHFUL OR COMPLETE. ANY REPRESENTATION TO THE CONTRARY IS A CRIMINAL OFFENSE.
The
date of this prospectus is October 20, 2022.
TABLE
OF CONTENTS
ABOUT
THIS PROSPECTUS
This
prospectus is a part of a registration statement on Form S-3 that we filed with the Securities and Exchange Commission, or SEC, utilizing
a “shelf” registration process. Under this shelf registration statement, we may sell any combination of the securities described
in this prospectus in one or more offerings up to a total aggregate offering price of $150,000,000. This prospectus provides you with a
general description of the securities we may offer.
Each
time we sell securities under this prospectus, we will provide a prospectus supplement that will contain specific information about the
terms of that offering. We may also authorize one or more free writing prospectuses to be provided to you that may contain material information
relating to these offerings. The prospectus supplement and any related free writing prospectus that we may authorize to be provided to
you may also add, update or change information contained in this prospectus or in any documents that we have incorporated by reference
into this prospectus. You should read this prospectus, any applicable prospectus supplement and any related free writing prospectus,
together with the information incorporated herein by reference as described under the heading “Incorporation of Certain Information
by Reference,” before investing in any of the securities offered.
THIS
PROSPECTUS MAY NOT BE USED TO CONSUMMATE A SALE OF SECURITIES UNLESS IT IS ACCOMPANIED BY A PROSPECTUS SUPPLEMENT.
Neither
we, nor any agent, underwriter or dealer has authorized any person to give any information or to make any representation other than those
contained or incorporated by reference in this prospectus, any applicable prospectus supplement or any related free writing prospectus
prepared by or on behalf of us or to which we have referred you. This prospectus, any applicable supplement to this prospectus or any
related free writing prospectus do not constitute an offer to sell or the solicitation of an offer to buy any securities other than the
registered securities to which they relate, nor do this prospectus, any applicable supplement to this prospectus or any related free
writing prospectus constitute an offer to sell or the solicitation of an offer to buy securities in any jurisdiction to any person to
whom it is unlawful to make such offer or solicitation in such jurisdiction.
You
should not assume that the information contained in this prospectus, any applicable prospectus supplement or any related free writing
prospectus is accurate on any date subsequent to the date set forth on the front of the document or that any information we have incorporated
by reference is correct on any date subsequent to the date of the document incorporated by reference, even though this prospectus, any
applicable prospectus supplement or any related free writing prospectus is delivered, or securities are sold, on a later date.
This
prospectus and the information incorporated herein by reference contain summaries of certain provisions contained in some of the documents
described herein, but reference is made to the actual documents for complete information. All of the summaries are qualified in their
entirety by the actual documents. Copies of some of the documents referred to herein have been filed, will be filed or will be incorporated
by reference as exhibits to the registration statement of which this prospectus is a part, and you may obtain copies of those documents
as described below under the heading “Where You Can Find More Information.”
Throughout
this prospectus, the terms “GT Biopharma,” “GTBP,” “we,” “us,” “our,” “the
company” and “our company” refer to GT Biopharma, Inc., a Delaware corporation formerly known as DDI Pharmaceuticals,
Inc., Diagnostic Data, Inc. and Oxis International, Inc., together with our subsidiaries.
PROSPECTUS
SUMMARY
This
summary highlights selected information from this prospectus and does not contain all of the information that you need to consider in
making your investment decision. You should carefully read the entire prospectus, the applicable prospectus supplement and any related
free writing prospectus, including the risks of investing in our securities discussed under the heading “Risk Factors” contained
in the applicable prospectus supplement and any related free writing prospectus, and under similar headings in the other documents that
are incorporated by reference into this prospectus. You should also carefully read the information incorporated by reference into this
prospectus, including our financial statements and related notes, and the exhibits to the registration statement of which this prospectus
is a part, before making your investment decision.
Overview
We
are a clinical stage biopharmaceutical company focused on the development and commercialization of novel immuno-oncology products based
on our proprietary Tri-specific Killer Engager (TriKE®) fusion protein immune cell engager technology platform. Our TriKE® platform
generates proprietary therapeutics designed to harness and enhance the cancer killing abilities of a patient’s own natural killer
cells, or NK cells. Once bound to an NK cell, our moieties are designed to enhance the NK cell, and precisely direct it to one or more
specifically targeted proteins expressed on a specific type of cancer cell or virus infected cell, resulting in the targeted cell’s
death. TriKE®s can be designed to target any number of tumor antigens on hematologic malignancies or solid tumors and do not require
patient-specific customization.
We
are using our TriKE® platform with the intent to bring to market immuno-oncology products that can treat a range of hematologic malignancies,
and solid tumors. The platform is scalable, and we are putting processes in place to be able to produce investigational new drug (IND)
ready moieties in a timely manner after a specific TriKE® conceptual design. Specific drug candidates can then be advanced into the
clinic on our own or through potential collaborations with partnering companies. We believe our TriKE®s may have the ability, if
approved for marketing, to be used as both monotherapy and in combination with other standard-of-care therapies.
We
are also using our TriKE® platform to develop therapeutics useful for the treatment of infectious disease such as for the treatment
of patients infected by the human immunodeficiency virus (HIV). While the use of anti-retroviral drugs has substantially improved the
health and increased the longevity of individuals infected with HIV, these drugs are designed to suppress virus replication to help modulate
progression to acquired immunodeficiency syndrome (AIDS) and to limit further transmission of the virus. Despite the use of anti-retroviral
drugs, infected individuals retain reservoirs of latent HIV-infected cells that, upon cessation of anti-retroviral drug therapy, can
reactivate and re-establish an active HIV infection. For a curative therapy, destruction of these latent HIV infected cells must take
place. The HIV-TriKE® contains the antigen binding fragment (Fab) from a broadly neutralizing antibody targeting the HIV-Env protein.
The HIV-TriKE® is designed to target HIV while redirecting NK cell killing specifically to actively replicating HIV infected cells.
The HIV-TriKE® induced NK cell proliferation and demonstrated the ability in vitro to reactivate and kill HIV-infected T-cells. These
findings indicate a potential role for the HIV-TriKE® in the reactivation and elimination of the latently infected HIV reservoir
cells by harnessing the NK cell’s ability to mediate the antibody-directed cellular cytotoxicity (ADCC).
Our
initial work has been conducted in collaboration with the Masonic Cancer Center at the University of Minnesota under a program led by
Dr. Jeffrey Miller, the Deputy Director. Dr. Miller is a recognized leader in the field of NK cell and IL-15 biology and their therapeutic
potential. We have exclusive rights to the TriKE® platform and are generating additional intellectual property for specific moieties.
Corporate
Information
Our
principal executive offices are located at 8000 Marina Blvd, Suite 100, Brisbane, CA 94005, and our telephone number is (415) 919-4040.
We maintain a website at www.gtbiopharma.com. Information contained on or accessible through our website is not, and should not
be considered, part of, or incorporated by reference into, this prospectus.
The
Securities We May Offer
Under
this prospectus, we may offer shares of our common stock and/or warrants or rights to purchase such securities, either individually or
in units, from time to time at prices and on terms to be determined by market conditions at the time of the relevant offering, up to
a total aggregate offering price of $150,000,000. This prospectus provides you with a general description of the securities we may offer.
Each time we offer a type or series of securities under this prospectus, we will provide a prospectus supplement that will describe the
specific amounts, prices and other important terms of the securities, including, to the extent applicable:
| |
● |
designation
or classification; |
| |
|
|
| |
● |
aggregate
principal amount or aggregate offering price; |
| |
|
|
| |
● |
maturity,
if applicable; |
| |
|
|
| |
● |
rates
and times of payment of interest or dividends, if any; |
| |
|
|
| |
● |
redemption,
conversion or sinking fund terms, if any; |
| |
|
|
| |
● |
voting
or other rights, if any; and |
| |
|
|
| |
● |
conversion
or exercise prices, if any. |
The
prospectus supplement also may add, update or change information contained in this prospectus or in documents we have incorporated by
reference into this prospectus. However, no prospectus supplement will fundamentally change the terms that are set forth in this prospectus
or offer a security that is not registered and described in this prospectus at the time of its effectiveness.
We
may sell the securities directly to investors or to or through agents, underwriters or dealers. We, and our agents or underwriters, reserve
the right to accept or reject all or part of any proposed purchase of securities. If we offer securities through agents or underwriters,
we will include in the applicable prospectus supplement:
| |
● |
the
names of those agents or underwriters; |
| |
|
|
| |
● |
applicable
fees, discounts and commissions to be paid to them; |
| |
|
|
| |
● |
details
regarding over-allotment options, if any; and |
| |
|
|
| |
● |
the
net proceeds to us. |
This
prospectus may not be used to consummate a sale of any securities unless it is accompanied by a prospectus supplement.
RISK
FACTORS
Investing
in our common stock involves a high degree of risk. You should carefully consider the risks, uncertainties and assumptions discussed
under the heading “Risk Factors” in our most recent annual report on Form 10-K (together with any material changes
thereto contained in subsequently filed quarterly reports on Form 10-Q, current reports on Form 8-K or other documents that we file with
the Securities and Exchange Commission, which are incorporated herein by reference as described under the heading “Where You
Can Find More Information”) and any prospectus supplement. The risks and uncertainties we have described in such documents
are not the only risks that we face. Additional risks and uncertainties not presently known to us or that we currently deem immaterial
may also impair our business, financial condition and operating results and adversely affect the market price of our common stock.
Prior
to making a decision about investing in our securities, you should carefully consider the specific factors discussed under the caption
“Risk Factors” in the applicable prospectus supplement, together with all of the other information contained in the prospectus
supplement or appearing or incorporated by reference in this prospectus. These risks could materially affect our business, results of
operations or financial condition and cause the value of our securities to decline. You could lose all or part of your investment.
CAUTIONARY
NOTICE REGARDING FORWARD-LOOKING STATEMENTS
Some
of the statements in this prospectus are “forward-looking statements” within the meaning of the safe harbor from liability
established by the Private Securities Litigation Reform Act of 1995. Forward-looking statements include statements regarding our current
beliefs, goals and expectations about matters such as our expected financial performance and condition, operating results, our business
strategy and our financing plans. The forward-looking statements in this prospectus are not based on historical facts, but rather reflect
the current expectations of our management concerning future results and events. The forward-looking statements generally can be identified
by the use of terms such as “believe,” “expect,” “anticipate,” “intend,” “plan,”
“foresee,” “may,” “guidance,” “estimate,” “potential,” “outlook,”
“target,” “forecast,” “likely” or other similar words or phrases. Similarly, statements that describe
our objectives, plans or goals are, or may be, forward-looking statements.
Forward-looking
statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements
to be different from any future results, performance and achievements expressed or implied by these statements. We cannot guarantee that
our forward-looking statements will turn out to be correct or that our beliefs and goals will not change. Our actual results could be
very different from and worse than our expectations for various reasons. The risk factors and cautionary language discussed in this prospectus
provide examples of risks, uncertainties and events that may cause actual results to differ materially from the expectations described
by us in such forward-looking statements, including among other things:
| |
● |
our
ability to develop and advance our current product candidates and programs into, and successfully complete, clinical trials; |
| |
|
|
| |
● |
our
financial performance and our ability to effectively manage our anticipated growth; |
| |
|
|
| |
● |
the
period over which we estimate our existing cash and cash equivalents will be sufficient to fund our future operating expenses and
capital expenditure requirements; |
| |
|
|
| |
● |
the
impact of laws and regulations;; |
| |
|
|
| |
● |
general
economic conditions; |
| |
|
|
| |
● |
the
effects of the coronavirus on the ongoing disruption of supply chains, the global economy, on the global financial markets and on
our business; |
| |
|
|
| |
● |
the
timing, scope and likelihood of regulatory filings and approvals; |
| |
|
|
| |
● |
our
ability to develop and advance our current product candidates and programs into, and successfully complete, clinical trials; |
| |
|
|
| |
● |
our
continued reliance on third parties to conduct additional clinical trials of our product candidates; |
| |
|
|
| |
● |
our
manufacturing, commercialization, and marketing capabilities and strategy; |
| |
|
|
| |
● |
our
intellectual property position, including the scope of protection we are able to establish and maintain for intellectual property
rights covering product candidates we may develop, including the validity of intellectual property rights held by third parties,
and our ability not to infringe, misappropriate or otherwise violate any third-party intellectual property rights; |
| |
|
|
| |
● |
the
rate and degree of market acceptance and clinical utility of our product candidates we may develop; |
| |
|
|
| |
● |
our
ability to hire additional qualified personnel and attract and retain key employees; and |
| |
|
|
| |
● |
the
result of any future financing efforts. |
These
risks, among others, could cause actual results to differ materially from those implied by the forward-looking statements contained in
this prospectus. You should review carefully all information, including the discussion under “Risk Factors” and “Management’s
Discussion and Analysis of Financial Condition and Results of Operations” in this prospectus or under similar headings in any
accompanying prospectus supplement or contained in the documents incorporated by reference herein. Any forward-looking statements in
this prospectus are made only as of the date hereof and, except as may be required by law, we do not have any obligation to publicly
update any forward-looking statements contained in this prospectus to reflect subsequent events or circumstances.
DILUTION
We
will set forth in a prospectus supplement the following information, as required, regarding any dilution of the equity interests of investors
purchasing securities in an offering under this prospectus:
| |
● |
the
net tangible book value per share of our equity securities before and after the offering; |
| |
|
|
| |
● |
the
amount of the change in such net tangible book value per share attributable to the cash payments made by purchasers in the offering;
and |
| |
|
|
| |
● |
the
amount of immediate dilution from the public offering price that will be absorbed by such purchasers. |
USE
OF PROCEEDS
Unless
otherwise indicated in the applicable prospectus supplement, we intend to use the net proceeds from the sale of securities for general
corporate purposes.
PLAN
OF DISTRIBUTION
We
may sell the securities offered by this prospectus to one or more underwriters or dealers for public offering, through agents, directly
to one or more purchasers or through a combination of any such methods of sale. The name of any such underwriters, dealers or agents
involved in the offer and sale of the securities, the amounts underwritten and the nature of their obligation to take the securities
will be specified in the applicable prospectus supplement. We have reserved the right to sell the securities directly to investors on
our own behalf in those jurisdictions where we are authorized to do so. The sale of the securities may be effected from time to time
in one or more transactions:
| |
● |
on
any national or international securities exchange or quotation service on which the securities may be listed or quoted at the time
of sale; |
| |
|
|
| |
● |
in
the over-the-counter market; |
| |
|
|
| |
● |
in
transactions otherwise than on such exchanges or in the over-the-counter market; or |
| |
|
|
| |
● |
through
the writing of options. |
Each
time that we sell securities covered by this prospectus, we will provide a prospectus supplement or supplements that will describe the
method of distribution and set forth the terms and conditions of the offering of such securities, including the offering price of the
securities and the proceeds to us, if applicable.
We
and our agents and underwriters may offer and sell the securities at a fixed price or prices that may be changed, at market prices prevailing
at the time of sale, at prices related to such prevailing market prices or at negotiated prices. The securities may be offered on an
exchange, which will be disclosed in the applicable prospectus supplement. We may, from time to time, authorize dealers, acting as our
agents, to offer and sell the securities upon such terms and conditions as set forth in the applicable prospectus supplement. We may
also sell the securities offered by any applicable prospectus supplement in “at-the-market offerings” within the meaning
of Rule 415 of the Securities Act, to or through a market maker or into an existing trading market, on an exchange or otherwise.
If
we use underwriters to sell securities, we will enter into an underwriting agreement with them at the time of the sale to them. In connection
with the sale of the securities, underwriters or agents may receive compensation from us in the form of underwriting discounts or commissions
and may also receive commissions from purchasers of the securities for whom they may act as agent. The names of any underwriters, any
underwriting compensation paid by us to underwriters or agents in connection with the offering of the securities, and any discounts,
concessions or commissions allowed by underwriters to participating dealers, will be set forth in the applicable prospectus supplement
to the extent required by applicable law. Underwriters may sell the securities to or through dealers, and such dealers may receive compensation
in the form of discounts, concessions or commissions from the underwriters or commissions (which may be changed from time to time) from
the purchasers for whom they may act as agents. If a dealer is utilized in the sale of the securities being offered by this prospectus,
the securities will be sold to the dealer, as principal. The dealer may then resell the securities to the public at varying prices to
be determined by the dealer at the time of resale.
Dealers
and agents participating in the distribution of the securities may be deemed to be underwriters, and any discounts and commissions received
by them and any profit realized by them on resale of the securities may be deemed to be underwriting discounts and commissions under
the Securities Act. Unless otherwise indicated in the applicable prospectus supplement, an agent will be acting on a best efforts basis.
If
so indicated in the prospectus supplement, we will authorize underwriters, dealers or agents to solicit offers by certain specified institutions
to purchase offered securities from us at the public offering price set forth in the prospectus supplement pursuant to delayed delivery
contracts providing for payment and delivery on a specified date in the future. Such contracts will be subject to any conditions set
forth in the applicable prospectus supplement and the prospectus supplement will set forth the commission payable for solicitation of
such contracts. The underwriters and other persons soliciting such contracts will have no responsibility for the validity or performance
of any such contracts. Offers to purchase the securities being offered by this prospectus may also be solicited directly. Underwriters,
dealers and agents may be entitled, under agreements entered into with us, to indemnification against and contribution towards certain
civil liabilities, including any liabilities under the Securities Act.
DESCRIPTION
OF COMMON STOCK
The
following description of our securities, together with any additional information we include in any applicable prospectus supplement
or any related free writing prospectus, summarizes the material terms and provisions of our capital stock. For the complete terms of
our capital stock, please refer to our certificate of incorporation and bylaws that are incorporated by reference into the registration
statement of which this prospectus is a part or may be incorporated by reference in this prospectus or any applicable prospectus supplement.
The terms of these securities may also be affected by the Delaware General Corporation Law (“DGCL”). The summary below and
that contained in any applicable prospectus supplement or any related free writing prospectus are qualified in their entirety by reference
to our certificate of incorporation and bylaws.
General
We
are authorized to issue 250,000,000 shares of common stock, par value $0.001 per share. As of August 15, 2022, we had approximately
31,092,498 shares of common stock outstanding.
Voting
Rights
Holders
of our common stock are entitled to one vote for each share of common stock held of record for the election of directors and on all matters
submitted to a vote of stockholders.
Dividends
Holders
of our common stock are entitled to receive dividends ratably, if any, as may be declared by the board of directors out of legally available
funds, subject to any preferential dividend rights of any preferred stock then outstanding.
Liquidation
In
the event of our dissolution, liquidation or winding up, holders of our common stock are entitled to share ratably in our net assets
legally available after the payment of all of our debts and other liabilities, subject to the liquidation preferences of any preferred
stock then outstanding.
Rights
and Preferences
Holders
of our common stock have no preemptive, subscription, redemption or conversion rights. The rights, preferences and privileges of holders
of common stock are subject to, and may be adversely affected by, the rights of the holders of shares of any series of preferred stock
currently outstanding or that we may designate and issue in the future.
DESCRIPTION
OF WARRANTS
The
following description, together with the additional information we may include in any applicable prospectus supplement and in any related
free writing prospectuses, summarizes the material terms and provisions of the warrants that we may offer under this prospectus, which
may consist of warrants to purchase common stock and may be issued in one or more series. Warrants may be offered independently or together
with common stock offered by any prospectus supplement, and may be attached to or separate from those securities. While the terms we
have summarized below will apply generally to any warrants that we may offer under this prospectus, we will describe the particular terms
of any series of warrants that we may offer in more detail in the applicable prospectus supplement and any applicable free writing prospectus.
The terms of any warrants offered under a prospectus supplement may differ from the terms described below. However, no prospectus supplement
will fundamentally change the terms that are set forth in this prospectus or offer a security that is not registered and described in
this prospectus at the time of its effectiveness.
We
have filed forms of the warrant agreements and forms of warrant certificates containing the terms of the warrants that may be offered
as exhibits to the registration statement of which this prospectus is a part. We will file as exhibits to the registration statement
of which this prospectus is a part, or will incorporate by reference from reports that we file with the SEC, the form of warrant agreement,
if any, including a form of warrant certificate, that describes the terms of the particular series of warrants we are offering. The following
summaries of material provisions of the warrants and the warrant agreements are subject to, and qualified in their entirety by reference
to, all the provisions of the warrant agreement and warrant certificate applicable to the particular series of warrants that we may offer
under this prospectus. We urge you to read the applicable prospectus supplements related to the particular series of warrants that we
may offer under this prospectus, as well as any related free writing prospectuses, and the complete warrant agreements and warrant certificates
that contain the terms of the warrants.
General
We
will describe in the applicable prospectus supplement the terms relating to a series of warrants being offered, including, to the extent
applicable:
| |
● |
the
title of such securities; |
| |
|
|
| |
● |
the
offering price or prices and aggregate number of warrants offered; |
| |
|
|
| |
● |
the
currency or currencies for which the warrants may be purchased; |
| |
|
|
| |
● |
if
applicable, the designation and terms of the securities with which the warrants are issued and the number of warrants issued with
each |
| |
|
|
| |
● |
such
security or each principal amount of such security; |
| |
|
|
| |
● |
if
applicable, the date on and after which the warrants and the related securities will be separately transferable; |
| |
|
|
| |
● |
if
applicable, the minimum or maximum amount of such warrants which may be exercised at any one time; |
| |
|
|
| |
● |
the
number of shares of common stock purchasable upon the exercise of one warrant and the price at which, and the currency in which,
these shares may be purchased upon such exercise; |
| |
|
|
| |
● |
the
effect of any merger, consolidation, sale or other disposition of our business on the warrant agreements and the warrants; |
| |
|
|
| |
● |
the
terms of any rights to redeem or call the warrants; |
| |
|
|
| |
● |
the
terms of any rights to force the exercise of the warrants; |
| |
|
|
| |
● |
any
provisions for changes to or adjustments in the exercise price or number of securities issuable upon exercise of the warrants; |
| |
|
|
| |
● |
the
dates on which the right to exercise the warrants will commence and expire; |
| |
|
|
| |
● |
the
manner in which the warrant agreements and warrants may be modified; |
| |
|
|
| |
● |
a
discussion of any material or special U.S. federal income tax consequences of holding or exercising the warrants; |
| |
|
|
| |
● |
the
terms of the securities issuable upon exercise of the warrants; and |
| |
|
|
| |
● |
any
other specific terms, preferences, rights or limitations of or restrictions on the warrants; |
| |
|
|
| |
● |
if
applicable, the minimum or maximum amount of the warrants that may be exercised at any one time; |
| |
|
|
| |
● |
information
with respect to book-entry procedures, if any; |
| |
|
|
| |
● |
the
anti-dilution provisions of the warrants, if any; |
| |
|
|
| |
● |
any
redemption or call provisions; |
| |
|
|
| |
● |
whether
the warrants may be sold separately or with other securities as parts of units; and |
| |
|
|
| |
● |
any
additional terms of the warrants, including terms, procedures and limitations relating to the exchange and exercise of the warrants. |
LEGAL
MATTERS
Unless
otherwise indicated in the applicable prospectus supplement, certain legal matters in connection with the offering and the validity of
the securities offered by this prospectus, and any supplement thereto, will be passed upon by Baker & McKenzie LLP, Dallas, Texas.
Additional legal matters may be passed upon for us or any underwriters, dealers or agents, by counsel that we will name in the applicable
prospectus supplement.
EXPERTS
The
financial statements of GT Biopharma, Inc. incorporated by reference in this Prospectus have been audited by Weinberg & Company,
P.A., an independent registered public accounting firm, as stated in their report. Such financial statements are incorporated by reference
in reliance upon the report of such firm given, upon their authority as experts in accounting and auditing.
WHERE
YOU CAN FIND MORE INFORMATION
This
prospectus is part of a registration statement we filed with the SEC. This prospectus does not contain all of the information set forth
in the registration statement and the exhibits to the registration statement. For further information with respect to us and the securities
we are offering under this prospectus, we refer you to the registration statement and the exhibits and schedules filed as a part of the
registration statement. You should rely only on the information contained in this prospectus or incorporated by reference in this prospectus.
We have not authorized anyone else to provide you with different information. We are not making an offer of these securities in any state
where the offer is not permitted. You should not assume that the information in this prospectus is accurate as of any date other than
the date on the front page of this prospectus, regardless of the time of delivery of this prospectus or any sale of the securities offered
by this prospectus.
We
file annual, quarterly and current reports, proxy statements and other information with the SEC. The SEC maintains a website at www.sec.gov
that contains reports, proxy and information statements, and other information regarding issuers, such as us, that file electronically
with the SEC.
Copies
of certain information filed by us with the SEC are also available on our website at www.gtbiopharma.com. Information contained
in or accessible through our website does not constitute a part of this prospectus and is not incorporated by reference in this prospectus.
We have included our website address as an inactive textual reference only.
INCORPORATION
OF CERTAIN INFORMATION BY REFERENCE
The
SEC allows us to “incorporate by reference” information into this prospectus, which means that we can disclose important
information to you by referring you to another document filed separately with the SEC. The information that we incorporate by reference
is an important part of this prospectus and information that we file later with the SEC will automatically update and supersede this
information.
The
following documents are incorporated by reference into this document:
| |
● |
our
Annual Report on Form
10-K for the fiscal year ended December 31, 2021, filed with the SEC on March 28, 2022; |
| |
|
|
| |
● |
our
Definitive
Proxy Statement on Schedule 14A (to the extent incorporated by reference into our Annual
Report on Form 10-K), filed with the SEC on April 29, 2022;
|
| |
● |
Our
Quarterly Report on Form 10-Q for the quarters ended March
31, 2022 and June
30, 2002, filed with the SEC on May 16, 2022 and August 15, 2022, respectively;
|
| |
● |
our
Current Reports on Form 8-K filed with the SEC on February
18, 2022, March
8, 2022, June
10, 2022, June
14, 2022 and August
30, 2022, and our Current Report on Form
8-K/A filed with the SEC on August 17, 2022, to the extent the information in such reports is filed and not furnished; and |
| |
● |
The
description of our common stock contained in our Registration Statement on Form
8A-12B filed with the SEC on February 8, 2021. |
We
also incorporate by reference into this prospectus all documents (other than current reports furnished under Item 2.02 or Item 7.01 of
Form 8-K and exhibits filed on such form that are related to such items) that are filed by us with the SEC pursuant to Sections 13(a),
13(c), 14 or 15(d) of the Exchange Act (i) after the date of the initial filing of the registration statement of which this prospectus
forms a part and prior to effectiveness of the registration statement, or (ii) after the date of this prospectus but prior to the termination
of the offering. These documents include periodic reports, such as Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current
Reports on Form 8-K, as well as proxy statements.
We
will provide to each person, including any beneficial owner, to whom a prospectus is delivered, without charge upon written or oral request,
a copy of any or all of the documents that are incorporated by reference into this prospectus but not delivered with the prospectus,
including exhibits that are specifically incorporated by reference into such documents. You should direct any requests for documents
to GT Biopharma, Inc., Attn: Corporate Secretary, 8000 Marina Blvd, Suite 100, Brisbane, CA 94005, and our telephone number is (415)
919-4040.
Any
statement contained in this prospectus or contained in a document incorporated or deemed to be incorporated by reference into this prospectus
will be deemed to be modified or superseded to the extent that a statement contained in this prospectus or any subsequently filed supplement
to this prospectus, or document deemed to be incorporated by reference into this prospectus, modifies or supersedes such statement.
740,000
Shares of Common Stock

GT
BIOPHARMA, INC.
ROTH
CAPITAL PARTNERS
May
21, 2024
GT Biopharma (NASDAQ:GTBP)
Historical Stock Chart
From May 2024 to Jun 2024
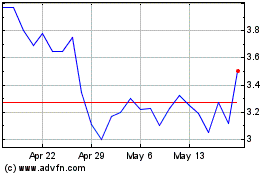
GT Biopharma (NASDAQ:GTBP)
Historical Stock Chart
From Jun 2023 to Jun 2024