Unitaid Awards Medincell up to $6 million Extension Grant to Fight Malaria
April 08 2024 - 1:09PM
Business Wire
Global health agency Unitaid has awarded Medincell
(Paris:MEDCL) an extension grant of up to US$ 6 million over three
years to fund the clinical phase 1 activities of long-acting
injectable mdc-STM. If proven safe, effective, and acceptable,
mdc-STM could have a significant impact on transmission of malaria
among vulnerable populations in high-transmission areas.
mdc-STM is an investigational three-month active injectable
formulation of ivermectin using Medincell’s BEPO® technology to
fight malaria transmission. A previous grant of $6.4 million was
awarded in March 2020 by Unitaid to fund the formulation research
phase and preclinical activities of the program conducted by
Medincell and the project consortium members, IRD, IRSS and
CIRDES.
Medincell is committed to the fight against the major global
health threats, such as malaria that remains endemic in 85
countries representing 50% of the world's population. According to
WHO estimates, 249 million people were infected worldwide in 2022,
94% of them in Africa, leading to 580,000 deaths in the
area.
Download the complete press release
About Medincell
Medincell is a clinical- and commercial-stage biopharmaceutical
licensing company developing long-acting injectable drugs in many
therapeutic areas. Our innovative treatments aim to guarantee
compliance with medical prescriptions, to improve the effectiveness
and accessibility of medicines, and to reduce their environmental
footprint. They combine active pharmaceutical ingredients with our
proprietary BEPO® technology which controls the delivery of a drug
at a therapeutic level for several days, weeks or months from the
subcutaneous or local injection of a simple deposit of a few
millimeters, entirely bioresorbable. The first treatment based on
BEPO® technology, intended for the treatment of schizophrenia, was
approved by the FDA in April 2023, and is now distributed in the
United States by Teva under the name UZEDY™ (BEPO® technology is
licensed to Teva under the name SteadyTeq™). We collaborate with
leading pharmaceutical companies and foundations to improve global
health through new treatment options. Based in Montpellier,
Medincell currently employs more than 140 people representing more
than 25 different nationalities.
UZEDY™ and SteadyTeq™ are trademarks of Teva Pharmaceuticals
www.medincell.com
About Unitaid
Unitaid saves lives by making critical health products available
and affordable for people in low- and middle-income countries. We
work with partners to identify innovative treatments, tests and
tools, help tackle the market barriers that are holding them back
and get them to the people who need them most – fast. Since its
creation in 2006, Unitaid has facilitated access to more than 100
groundbreaking health products to help address the world’s biggest
health challenges, including HIV, TB, and malaria; women’s and
children’s health; and pandemic prevention, preparedness and
response. Every year, more than 170 million people benefit from the
products whose deployment Unitaid has supported.
unitaid.org
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240408604143/en/
David Heuzé Head of Corporate and Financial
Communications, and ESG david.heuze@medincell.com / +33 (0)6 83 25
21 86
Grace Kim Head of US Financial Strategy & IR
grace.kim@medincell.com / +1 (646) 991-4023
Nicolas Mérigeau Media Relations medincell@newcap.eu /
+33 (0)1 44 71 94 94
Louis-Victor Delouvrier/Alban Dufumier Investor Relations
France medincell@newcap.eu / +33 (0)1 44 71 94 94
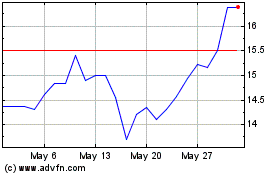
Medincell (EU:MEDCL)
Historical Stock Chart
From Apr 2024 to May 2024
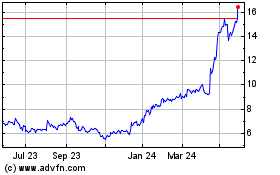
Medincell (EU:MEDCL)
Historical Stock Chart
From May 2023 to May 2024