New FDA Clearance Illustrates the Utility of the
i:X to Reliably Detect
Clinically Significant Bacteria that Impedes Wound Healing
TORONTO, June 29, 2022 /PRNewswire/ -- MolecuLight
Inc., the leader in point-of-care fluorescence imaging for
real-time detection of wounds containing elevated bacterial loads,
announces that it has received an expansion to its FDA 510(k)
clearance for the MolecuLight i:X® imaging
device's ability to detect the location of elevated bacterial loads
(>104 CFU/g) in wounds. The expanded labelling also
includes the device's ability to identify areas of wounds
containing more bacterial species, including key target pathogens
of interest to the CDC that are major causes of antimicrobial
resistance1. Detectable species include gram-negative
and gram-positive species, aerobes and anaerobes. This expanded
labeling is based on a detailed retrospective statistical analysis
of over 350 patients.
"We are thrilled with the FDA's new clearance for MolecuLight'
ability to determine the location of elevated bacterial loads in
wounds, in addition to the ability to identify regions with more
bacterial species of interest", says Anil Amlani, MolecuLight's
CEO. "Clinicians worldwide are using the MolecuLight device to
visualize regions with clinically significant bacterial loads and
more species of concern. With point-of-care information on
bacterial load and its locations through use of a
MolecuLight device, clinicians can act immediately to tailor their
cleaning, debridement, antimicrobial strategies and treatments
accordingly."
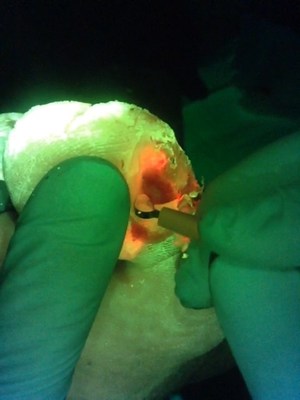
This video (courtesy of Rose
Raizman) illustrates the importance of visualizing the
location of elevated bacterial load in a wound. In this scenario,
the clinician is using MolecuLight i:X to inform
their decision-making and target their wound hygiene to the areas
of red fluorescence. Regions of red, indicating that the wound
contains clinically significant (>104 CFU/g) levels
of bacterial burden, are clearly visible on the patient's diabetic
foot ulcer (see image).
In addition, the FDA has also recognized MolecuLight's ability
to visualize regions containing troublesome bacterial species at
the point-of-care. The MolecuLight device can be used to enable
fluorescence-guided tissue biopsies to these regions to detect a
higher number of pathogens of interest (defined by the CDC as
increasing risk to develop antibiotic resistance) compared to
standard-of-care-guided biopsies. The CDC has identified antibiotic
resistance as "one of the greatest global public health
challenges of our time"1. Strategies to
combat antibiotic resistance include containing emerging threats
through early detection and aggressive response and improving
appropriate antibiotic use through antimicrobial stewardship
programs. The expanded use of diagnostic tools, like MolecuLight,
to improve accuracy and speed of pathogen detection has been called
out to help improve appropriate antibiotic selection and reduce
unnecessary antibiotic use1.
MolecuLight was the first to receive FDA de novo
clearance for its MolecuLight i:X imaging
platform and has subsequently received three additional FDA 510(k)
clearances for the device.
|
Reference:
|
|
1CDC. Antibiotic
Resistance Threats in the United States, 2019. Atlanta, GA: US
Department of Health and Human Services, CDC;2019
|
About MolecuLight
Inc.
MolecuLight Inc. is a privately-owned medical imaging
company that has developed and is commercializing its proprietary
fluorescent imaging platform technology in multiple clinical
markets. MolecuLight's suite of commercially released devices,
including the MolecuLight i:X® and
DX™ fluorescence imaging systems and their accessories,
provide point-of-care handheld imaging devices for the global wound
care market for the real-time detection and localization of
elevated bacterial load in wounds and for digital wound
measurement. MolecuLight procedures performed in the United States can benefit from an
available reimbursement pathway including two CPT® codes
for physician work to perform "fluorescence imaging for
bacterial presence, location, and load" and facility payment
for Hospital Outpatient Department
(HOPD) and Ambulatory Surgical Center
(ASC) settings through an Ambulatory Payment
Classification (APC) assignment. The company is also
commercializing its unique fluorescence imaging platform technology
for other markets with globally relevant unmet needs including food
safety, consumer cosmetics and other key industrial markets.
www.moleculight.com
Image: Download at:
https://moleculight.box.com/s/ax8758gz0f8amouhcjjvs4xzkfa9vofo
Video: https://youtu.be/HKOCGBlIQj4
Rob Sandler, Chief Marketing
Officer, MolecuLight Inc., T. +1.647.362.4684,
rsandler@moleculight.com
Photo -
https://mma.prnewswire.com/media/1849819/MolecuLight_FDA_510_k__Clearance_Expands_Labelling_of_MolecuLigh.jpg