New ‘Negative Pressure Scavenger Kit’ Helps Hospitals Manage Infectious Particles When Treating Patients with COVID-19, S...
August 12 2020 - 8:45AM
Business Wire
- FELIX-1 negative pressure scavenger kit creates localized
negative pressure area around the patient’s face
- All profits from kit sales will be donated to support frontline
clinicians and their families through non-profit COVID-19 relief
funds
Vapotherm, Inc. (NYSE: VAPO), a global medical technology
company focused on the development and commercialization of its
proprietary high velocity therapy, which is used to treat patients
of all ages suffering from respiratory distress, today announced
the release of a low-cost, easy to use kit to help clinicians
create a zone of relative negative pressure at the patient's face
which may mitigate the risk of transmission of potentially
infectious particles while treating COVID-19 patients. All profits
from the kits will be distributed to non-profit organizations
supporting front-line clinicians fighting the COVID-19
pandemic.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20200812005383/en/
Patient treated with Vapotherm high
velocity therapy while using the FELIX-1 negative pressure
scavenger kit (Photo: Business Wire)
The FELIX-1 negative pressure scavenger kit is a preassembled
tool compatible with high velocity therapy, an advanced form of
high flow nasal cannula increasingly used by clinicians in
supporting the respiratory needs of COVID-19 patients. The FELIX-1
design is replicable with off-the-shelf components, and is named
for Felix Khusid, a respiratory therapy leader working in New York
City who shared this previously implemented design with Vapotherm
during the first wave of COVID-19.
“It’s important that clinicians are protected to the greatest
extent possible while using the critical tools they need to support
patients and their communities through this pandemic, ” said Joe
Army, CEO of Vapotherm, “When Felix presented us with his approach
to potentially improve caregiver safety, we made it our mission to
help get this tool into hospitals as quickly and broadly as
possible.”
In computational fluid dynamics simulations performed by
Vapotherm, the FELIX-1 negative pressure scavenger kit was capable
of capturing up to 97% of particle mass when connected to an
in-room vacuum system and used in conjunction with Vapotherm high
velocity therapy.
Vapotherm will donate all profits from the sale of these kits to
non-profit funds to support caregivers serving on the front lines
of the COVID-19 pandemic, such as those established by the American
Association of Respiratory Care, Emergency Nurses Association, and
the American College of Emergency Physicians
Vapotherm, Inc. (NYSE: VAPO) is a publicly traded
developer and manufacturer of advanced respiratory technology based
in Exeter, New Hampshire, USA. The company develops innovative,
comfortable, non-invasive technologies for respiratory support of
patients with chronic or acute breathing disorders. Over 2.3
million patients have been treated with Vapotherm high velocity
therapy. For more information, visit www.vapotherm.com.
Vapotherm high velocity therapy is mask-free noninvasive
ventilatory support and is a front-line tool for relieving
respiratory distress—including hypercapnia, hypoxemia, and dyspnea.
It allows for the fast, safe treatment of undifferentiated
respiratory distress with one user-friendly tool. The Precision
Flow system’s mask-free interface delivers optimally conditioned
breathing gases, making it comfortable for patients and reducing
the risks and care complexities associated with mask therapies.
While being treated, patients can talk, eat, drink and take oral
medication.
Website Information Vapotherm routinely posts important
information for investors on the Investor Relations section of its
website, http://investors.vapotherm.com/. Vapotherm intends to use
this website as a means of disclosing material, non-public
information and for complying with Vapotherm’s disclosure
obligations under Regulation FD. Accordingly, investors should
monitor the Investor Relations section of Vapotherm’s website, in
addition to following Vapotherm’s press releases, Securities and
Exchange Commission filings, public conference calls, presentations
and webcasts. The information contained on, or that may be accessed
through, Vapotherm’s website is not incorporated by reference into,
and is not a part of, this document.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200812005383/en/
PR and Media Contact: Greg Ramade, VP of Marketing,
+1-(603)-658-0628, pr@vtherm.com
Vapotherm (NYSE:VAPO)
Historical Stock Chart
From Mar 2024 to Apr 2024
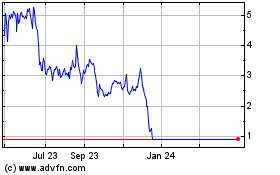
Vapotherm (NYSE:VAPO)
Historical Stock Chart
From Apr 2023 to Apr 2024