Eli Lilly Asks FDA to Authorize Covid-19 Antibody Drug
October 07 2020 - 9:19AM
Dow Jones News
By Peter Loftus
Eli Lilly & Co. said it has requested U.S. authorization of
the emergency use of an experimental antibody-based treatment for
people with recently diagnosed, mild-to-moderate Covid-19,
following positive results from clinical testing.
The company said it is seeking the authorization for its drug,
code named LY-CoV555, which was derived from a blood sample of one
of the earliest U.S. survivors of Covid-19. The company said last
month the drug reduced the rate of hospitalization compared with a
placebo in a study.
Lilly said Wednesday it is manufacturing doses and could have
100,000 doses this month and as many as one million by the end of
the year.
Lilly also said a new study showed that combining LY-CoV555 with
another antibody reduced viral load, symptoms and hospitalizations
in Covid-19 patients. The company expects to seek emergency use
authorization for the combination in November.
Lilly's antibody is meant to mimic the antibodies of the human
immune system that can neutralize the new coronavirus. The approach
is similar to the experimental antibody-based treatment from
Regeneron Pharmaceuticals that was given to President Trump.
Write to Peter Loftus at peter.loftus@wsj.com
(END) Dow Jones Newswires
October 07, 2020 09:04 ET (13:04 GMT)
Copyright (c) 2020 Dow Jones & Company, Inc.
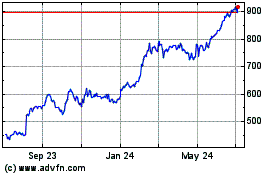
Eli Lilly (NYSE:LLY)
Historical Stock Chart
From Mar 2024 to Apr 2024
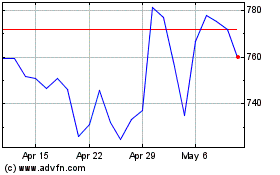
Eli Lilly (NYSE:LLY)
Historical Stock Chart
From Apr 2023 to Apr 2024