Emergent BioSolutions to Acquire from Chimerix its Exclusive Worldwide Rights to TEMBEXA® (brincidofovir), the First FDA-Approved Smallpox Oral Antiviral for All Ages
May 16 2022 - 6:32AM

Emergent BioSolutions Inc. (NYSE: EBS)
announced today that it has entered into a definitive agreement
with Chimerix, Inc. (NASDAQ: CMRX), to acquire Chimerix’s exclusive
worldwide rights to TEMBEXA® (brincidofovir), the first antiviral
approved by the U.S. Food and Drug Administration (FDA) for all age
groups for the treatment of smallpox. TEMBEXA was approved in June
2021 and is indicated for the treatment of human smallpox disease
in adult and pediatric patients, including neonates.
“The addition of TEMBEXA to Emergent’s portfolio of medical
countermeasures builds upon our core capabilities and leverages our
long and successful history partnering with the U.S. government to
address dangerous public health threats,” said Robert G. Kramer,
president and CEO of Emergent. “It exemplifies our thoughtful
M&A strategy as part of our 2024 growth plan and positions us
better to deliver value for our shareholders.”
“This transaction expands and further diversifies our medical
countermeasures business with the addition of a small molecule
therapeutic that aligns with the government’s smallpox preparedness
strategy,” said Paul Williams, SVP government/MCM business at
Emergent. “It is expected to be accretive upon first product
delivery under the anticipated BARDA contract within three to six
months from closing.”
Transaction DetailsUnder the terms of the
agreement, Emergent will pay Chimerix a $225 million one-time
upfront payment in cash upon closing and up to a total of $100
million in milestone payments contingent on the potential exercise
by the U.S. government of procurement options following the base
period. The closing payment and the milestone payments may be
adjusted based on the actual procurement value. The terms also
include sales-based royalties contingent on future potential
worldwide procurement during the exclusivity period of TEMBEXA on a
market-to-market basis. Chimerix remains eligible to receive a
portion of the regulatory milestone payments associated with the
license to SymBio Pharmaceuticals Ltd. for indications other than
orthopox infections.
Emergent anticipates that the transaction will be funded using
currently available funds. Closing ConditionsThis
transaction is subject to customary closing conditions, including
expiration or early termination of the applicable waiting period
under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as
amended (HSR Act).
The transaction is further conditioned on the execution of an
anticipated procurement contract between Chimerix and the
Biomedical Advanced Research and Development Authority (BARDA) as
well as receipt of any required consent from BARDA to a
pre-novation agreement to be entered into with Emergent, upon which
time Emergent would be poised to deliver the first shipment of
TEMBEXA to the U.S. Strategic National Stockpile (SNS) upon
completion of customary pre-shipment obligations. In December 2021,
BARDA issued a sole source request for proposal (RFP) to procure up
to 1.7 million treatment courses of TEMBEXA. Chimerix expects a
BARDA procurement contract award as early as second quarter of
2022.
Subject to the satisfaction or waiver of the closing conditions,
the companies expect the transaction to close as early as the end
of the second quarter of 2022. ABOUT
TEMBEXATEMBEXA is an oral antiviral formulated as
100 mg tablets and 10 mg/mL oral suspension dosed once
weekly for two weeks. TEMBEXA is indicated for the treatment of
human smallpox disease in adult and pediatric patients, including
neonates. TEMBEXA is not indicated for the treatment of diseases
other than human smallpox disease.
In June 2021, the FDA approved TEMBEXA tablets and oral
suspension for the treatment of smallpox. TEMBEXA is approved for
adult and pediatric patients and is the first and only smallpox
therapy approved for neonates. The oral suspension formulation is
particularly important for patients who have difficulty swallowing
due to age or medical status. Please read full prescribing
information here.
About SmallpoxSmallpox is a highly contagious
disease caused by the variola virus. Historically, smallpox was one
of the deadliest diseases in history with a case fatality rate of
approximately 30%. Despite successful eradication of smallpox in
the 1970s, there is considerable concern that variola virus could
reappear, either through accidental release or as a weapon of
bioterrorism. According to the U.S. Centers for Disease
Control and Prevention (CDC), variola virus is ranked in the
highest risk category for bioterrorism agents (Category A) due to
its ease of transmission, high mortality rate, and potential to
cause public panic and social disruption. Based on a recent report
– The Department of Health and Human Services Fiscal Year 2023
Public Health and Social Services Emergency Fund Justification of
Estimates for Appropriations Committee – smallpox remains a threat
of high concern to both the domestic and international community.
BARDA's goal is to ensure adequate vaccine supply for all
Americans, including special populations, and to make available at
least two different therapeutic agents as recommended by the
National Academy of Medicine of the National Academies of Sciences,
Engineering, and Medicine.
About Emergent BioSolutionsAt Emergent, our
mission is to protect and enhance life. For over 20 years, we’ve
been at work defending people from things we hope will never
happen—so we are prepared just in case they ever do. We provide
solutions for complex and urgent public health threats through a
portfolio of vaccines and therapeutics that we develop and
manufacture for governments and consumers. We also offer a range of
integrated contract development and manufacturing services for
pharmaceutical and biotechnology customers. To learn more about how
we plan to protect or enhance 1 billion lives by 2030, visit
our website and follow us on LinkedIn, Twitter,
and Instagram.Safe Harbor StatementThis press
release includes forward-looking statements within the meaning of
the Private Securities Litigation Reform Act of 1995. Any
statements, other than statements of historical fact, including
statements regarding the timing of product deliveries; the
potential benefits of the acquisition to Emergent and the timing of
the acquisition becoming accretive; Emergent’s 2024 growth plan;
becoming better positioned to deliver value for Emergent
shareholders; the timing and ability of Chimerix to secure the
anticipated BARDA procurement contract; the parties’ ability to
consummate the transactions contemplated under the agreement,
satisfaction of conditions in connection with the acquisition, the
parties’ ability to meet expectations regarding the timing and
completion of the transaction and any other statements containing
the words “believes,” “expects,” “anticipates,” “intends,” “plans,”
“estimates” and similar expressions, are forward-looking
statements. These forward-looking statements are based on
Emergent’s current intentions, beliefs and expectations regarding
future events. Emergent cannot guarantee that any forward-looking
statement will be accurate. The reader should realize that if
underlying assumptions prove inaccurate or unknown risks or
uncertainties materialize, actual results could differ materially
from expectations. The reader is, therefore, cautioned not to place
undue reliance on any forward-looking statement. Any
forward-looking statement speaks only as of the date of this press
release, and, except as required by law, Emergent does not
undertake to update any forward-looking statement to reflect new
information, events or circumstances.
There are a number of important factors that could cause actual
results to differ materially from those indicated by such
forward-looking statements, including, but not limited to,
uncertainties as to the satisfaction of the closing conditions with
respect to the transaction; the potential inability of Chimerix to
secure the anticipated BARDA procurement contract; the timing and
volume of deliveries and exercise of options under the anticipated
BARDA procurement contract; and, following award of the anticipated
BARDA procurement contract, the ability of the parties to novate it
to Emergent. The reader should consider this cautionary statement,
as well as the risk factors identified in Emergent’s periodic
reports filed with the SEC, when evaluating the forward-looking
statements contained herein.
Investor ContactRobert BurrowsVice
President, Investor Relationsburrowsr@ebsi.com(240) 413-1917
Media ContactMatt HartwigSenior
Director, Media Relationsmediarelations@ebsi.com
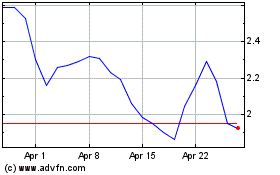
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From Mar 2024 to Apr 2024
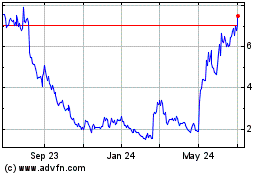
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From Apr 2023 to Apr 2024