- Nurtec ODT open-label extension study is the first of
its kind evaluating a CGRP-targeted medication in patients who are
using it as a preventive and an as-needed acute treatment for
migraine
- Over the course of this open-label
extension study, > 80% of patients experienced ≥50% reduction
and approximately 50% experienced a 100% reduction
-
Complete data from the Phase 3 zavegepant nasal spray study
highlight its ultra-rapid pain relief in as little as 15 minutes
that lasts through 48 hours after a single dose
-
New studies showed positive efficacy, safety and
pharmacokinetics data for Nurtec ODT as an acute treatment of
migraine in adults from China and
Korea
NEW
HAVEN, Conn., June 13,
2022 /PRNewswire/ -- Biohaven Pharmaceutical Holding
Company Ltd. (NYSE: BHVN) today announced new data from three
late-breakers and three oral presentations supporting the safety
and efficacy of Nurtec® ODT (rimegepant) and zavegepant
nasal spray for the treatment of migraine. The oral presentations
included a first of its kind study in migraine reporting on the
safety and benefits of Nurtec ODT when used as a preventive
treatment and as-needed for acute treatment of migraine. The data
were presented at the 64th Annual Scientific Meeting of the
American Headache Society (AHS) held from June 9-12 in Denver,
CO.
Elyse Stock, MD, Chief Medical
Officer of Biohaven commented, "Biohaven is proud of the growing
body of clinical efficacy and safety data for Nurtec ODT and
zavegepant, the building blocks of our CGRP franchise. We are
committed to advancing research for migraine and bringing effective
treatment options to the millions of patients living with it. We
hope these data will give confidence to healthcare providers that
Nurtec ODT can offer an effective, all-in-one treatment for
patients to both prevent and treat migraine attacks. With the US
FDA's recent acceptance of the NDA filing for zavegepant nasal
spray, we are excited about its potential as a new therapy for the
acute treatment of migraine."
These new data reinforce Nurtec ODT's efficacy and safety for
patients using it to both prevent and treat migraine attacks.
Biohaven also presented full Phase 3 efficacy and safety data for
zavegepant nasal spray. The data show zavegepant nasal spray was
effective for the acute treatment of migraine, achieving its
coprimary endpoints and providing an ultra-rapid onset of pain
relief at the earliest measured time point of 15 minutes and
sustained benefits through 48 hours after a single intranasal dose,
with favorable safety and tolerability.
Richard Lipton, MD, lead author
and presenter of the 52-week, open-label Nurtec ODT extension
study, and Professor and Vice Chair of Neurology at the Albert
Einstein College of Medicine and Director of the Montefiore
Headache Center commented, "I'm honored to share results from a
first of its kind study – one that looked at Nurtec ODT's safety,
tolerability, and outcomes when used as an acute and preventive
treatment of migraine over 52 weeks after the 12-week double-blind
prevention study. Given these positive outcomes coupled with a
consistently safe and well-tolerated profile, the data continue to
suggest that Nurtec ODT represents an effective, flexible treatment
option for patients."
The oral presentations spotlighting the new data were presented
on Saturday, June 11. Details from
all late-breaker and oral presentations are shared below.
Late-Breakers
- Patterns
of Medication Utilization and Migraine Frequency in Adults Using
Rimegepant for Both Preventive and Acute Treatment for Migraine:
Results from a 52-Week, Open-Label Extension Study
- A 52-week, open-label extension study of rimegepant 75 mg dosed
every other day for preventive treatment plus as-needed for acute
treatment of migraine demonstrated that the monthly frequency of
moderate or severe migraine days and use of other acute migraine
medications were uncommon and consistent between scheduled versus
nonscheduled rimegepant dosing days, suggesting that the benefits
of rimegepant are sustained. On nonscheduled dosing days,
rimegepant was used as an acute treatment approximately 1 day per
month, and additional acute medications were rarely utilized.
-
Efficacy, Safety, and Tolerability of Rimegepant 75 mg Orally
Disintegrating Tablet for the Acute Treatment of Migraine: Results
from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial
in Adults From China and Korea (Study BHV3000-310)
- In a large Phase 3 trial evaluating the safety and efficacy of
rimegepant for the acute treatment of migraine in 1,431 patients in
the People's Republic of China and
the Republic of Korea, rimegepant was determined to be superior to
placebo for both coprimary endpoints: pain freedom at 2 hours
post-dose (19.8% vs 10.7%, P<.0001) and freedom from most
bothersome symptom (MBS) (50.5% vs 35.8%, P<.0001). A single
dose of rimegepant 75 mg, without rescue medication, was superior
to placebo demonstrating sustained pain relief from 2 hours
post-dose through 48 hours post-dose.
- A Phase
1, Randomized, Placebo-controlled, Single- and Multiple- dose,
Double-blind Study to Evaluate the Pharmacokinetics and Safety of
Rimegepant Orally Disintegrating Tablets 75 mg in Healthy Chinese
Adults
- This Phase 1 study evaluated safety and pharmacokinetics of
rimegepant in 16 patients of Chinese ethnicity. Overall,
administration of single and multiple rimegepant 75 mg doses was
safe and well tolerated in healthy Chinese adults. The PPK model
supports the rimegepant 75 mg dose in the Chinese population for
the acute treatment of migraine, repeated up to once daily, with
the conclusion that there are no significant ethnicity differences
of rimegepant PK between Chinese and non-Chinese participants.
Oral and Poster Presentations
- Safety
and Tolerability of Rimegepant Every Other Day for Preventive
Treatment of Migraine Plus As-Needed for Acute Treatment of
Migraine: Results from a 52-Week, Open-Label Extension Study
- In a long-term, open-label extension phase of the Phase 2/3
study of rimegepant 75 mg as a preventive treatment of migraine in
adults, 603 patients were administered rimegepant every other day
plus as-needed on nonscheduled dosing days for up to one year. The
data showed that rimegepant 75 mg is safe and well tolerated with
no liver safety concerns. The most common on-treatment adverse
events (AEs) were upper respiratory tract infection (7.1%),
nasopharyngitis (6.3%), and back pain (4.3%). The rate of
discontinuation due to AEs was 2.8%. Though serious AEs occurred in
2.2% of subjects, none were considered to be related to
rimegepant.
- A separate poster presentation of this study presented analyses
of reductions in migraine frequency over time. Over the course of
the study, 80.9% of patients experienced ≥50% reduction in moderate
or severe monthly migraine days, 65.8% experienced a ≥75%
reduction, and 49.3% experienced a 100% reduction.
- Efficacy
and Safety of Zavegepant Nasal Spray for the Acute Treatment of
Migraine: Results of a Phase 3 Double-Blind, Randomized, Placebo
Controlled Trial
- Complete results from this Phase 3 clinical trial evaluating
the efficacy and safety of zavegepant nasal spray in 1,405
randomized patients reported that zavegepant is effective for the
acute treatment of migraine with favorable safety and tolerability.
Zavegepant was superior to placebo for the coprimary endpoints:
freedom from pain 2 hours post-dose (23.6% vs 14.9%, P<.0001)
and freedom from most bothersome symptom 2 hours post-dose (39.6%
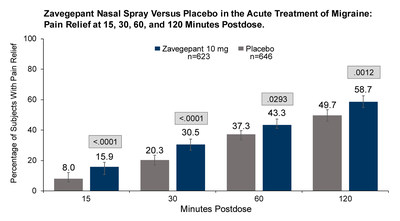
vs 31.1%, P=.0012). Secondary endpoints included pain relief at 15
minutes (15.9% vs 8.0%, P<.0001) and 2 hours (58.7% vs 49.7%,
P=.0012); return to normal function at 30 minutes (10.5% vs 6.1%,
P=.0059) and 2 hours (35.8% vs 25.6%, P=.0001); and sustained pain
relief 2 to 48 hours (36.1% vs 29.6%, P=.013) post-dose.
-
Increased Placebo Response Over Time in Oral Migraine Preventive
Trials: A Systematic Literature Review and Meta-analysis
- This study evaluated trends in placebo response over time in
preventive migraine treatment clinical trials from January 1990 to August
2021. The researchers observed a significant increase in
oral placebo response across time, indicating that routes of
administration (oral and injectable) may exhibit different levels
of contextual effect. Additionally, increasing temporal trends in
placebo response may bias future meta-analyses intended to
indirectly compare the effectiveness of preventive migraine
medications where the time span of included trials is large. This
can also impact study design and approval of future migraine
treatments due to decreasing effect sizes.
Dr. Lipton is a paid consultant and stockholder of Biohaven.
About NURTEC ODT
NURTEC ODT (rimegepant) is the first and only calcitonin
gene-related peptide (CGRP) receptor antagonist available in a
quick-dissolve ODT formulation that is approved by
the U.S. Food and Drug Administration (FDA) for the acute
treatment of migraine with or without aura and the preventive
treatment of episodic migraine in adults. The activity of the
neuropeptide CGRP is thought to play a causal role in migraine
pathophysiology. NURTEC ODT is a CGRP receptor antagonist that
works by reversibly blocking CGRP receptors, thereby inhibiting the
biologic activity of the CGRP neuropeptide. The recommended dose of
NURTEC ODT is 75 mg, taken as needed, up to once daily to treat or
every other day to help prevent migraine attacks. For more
information about NURTEC ODT, visit www.nurtec.com. The most
common adverse reaction was nausea and abdominal pain/indigestion.
Avoid concomitant administration of NURTEC ODT with strong
inhibitors of CYP3A4, strong or moderate inducers of CYP3A. Avoid
another dose of NURTEC ODT within 48 hours when it is administered
with moderate inhibitors of CYP3A4 or potent inhibitors of
P-gp.
Indication
NURTEC ODT orally disintegrating tablets is a prescription medicine
that is used to treat migraine in adults. It is for the acute
treatment of migraine attacks with or without aura and the
preventive treatment of episodic migraine. It is not known if
NURTEC ODT is safe and effective in children.
Important Safety Information
Do not take NURTEC ODT if you are allergic to NURTEC
ODT (rimegepant) or any of its ingredients.
Before you take NURTEC ODT, tell your healthcare provider (HCP)
about all your medical conditions, including if you:
- have liver problems,
- have kidney problems,
- are pregnant or plan to become pregnant,
- are breastfeeding or plan to breastfeed.
Tell your HCP about all the medicines you take, including
prescription and over-the-counter medicines, vitamins, and herbal
supplements.
NURTEC ODT may cause serious side effects including allergic
reactions, trouble breathing and rash. This can happen days after
you take NURTEC ODT. Call your HCP or get emergency help right away
if you have swelling of the face, mouth, tongue, or throat or
trouble breathing. This occurred in less than 1% of patients
treated with NURTEC ODT.
The most common side effects of NURTEC ODT were
nausea (2.7%) and stomach pain/indigestion (2.4%). These are not
the only possible side effects of NURTEC ODT. Tell your HCP if you
have any side effects.
You are encouraged to report side effects of prescription
drugs to the FDA.
Visit www.fda.gov/medwatch or call
1–800–FDA–1088 or report side effects to Biohaven at
1–833–4NURTEC.
See full Prescribing
Information and Patient
Information.
About Zavegepant
Zavegepant is a third generation, high affinity, selective and
structurally unique, small molecule CGRP receptor antagonist from
Biohaven's NOJECTION™ Migraine Platform and the only CGRP receptor
antagonist in clinical development with both intranasal and oral
formulations. The efficacy and safety profile of intranasal
zavegepant for the acute treatment of migraine, as compared to
placebo, was shown in a randomized controlled Phase 2/3
dose-ranging trial with a total of over 1000 patients who received
zavegepant. In this study, zavegepant showed statistical
superiority to placebo on the coprimary endpoints of 2-hour freedom
from pain and freedom from a patients' most bothersome symptom
(either nausea, photophobia or phonophobia). This was the second
zavegepant pivotal clinical trial to meet these coprimary
endpoints. The U.S. Food and Drug Administration has accepted a new
drug application for zavegepant with a Prescription Drug User Fee
Act date in the first quarter of 2023.
About Biohaven
Biohaven is a global commercial-stage
biopharmaceutical company with a portfolio of innovative,
best-in-class therapies to improve the lives of patients with
debilitating neurological and neuropsychiatric diseases, including
rare disorders. Biohaven's Neuroinnovation™ portfolio includes
FDA-approved NURTEC® ODT (rimegepant) for the acute and
preventive treatment of migraine (EMA-approved as
VYDURA® for the acute treatment of migraine with or
without aura, and prophylaxis of episodic migraine in adults who
have at least four migraine attacks per month) and a broad pipeline
of late-stage product candidates across five distinct mechanistic
platforms: CGRP receptor antagonism for the acute and preventive
treatment of migraine and other CGRP-mediated diseases; glutamate
modulation for obsessive-compulsive disorder and spinocerebellar
ataxia; myeloperoxidase (MPO) inhibition for amyotrophic lateral
sclerosis; Kv7 ion channel activators for focal epilepsy and
neuronal hyperexcitability, and myostatin inhibition for
neuromuscular diseases. More information about Biohaven is
available at www.biohavenpharma.com.
NURTEC, NURTEC ODT and VYDURA are registered trademarks of
Biohaven Pharmaceutical Ireland DAC. Neuroinnovation and NOJECTION
are trademarks of Biohaven Pharmaceutical Holding Company Ltd.
Forward-looking Statement
This news release includes forward-looking statements within the
meaning of the Private Securities Litigation Reform Act of 1995.
The use of certain words, including "believe", "continue", "may",
"will", "anticipate", "expect" and similar expressions, are
intended to identify forward-looking statements. These
forward-looking statements involve substantial risks and
uncertainties, including statements that are based on the current
expectations and assumptions of Biohaven's management about NURTEC
ODT as an acute treatment for patients with migraine and preventive
treatment for migraine. Factors that could affect these
forward-looking statements include those related to: Biohaven's
ability to effectively commercialize NURTEC ODT, delays or problems
in the supply or manufacture of NURTEC ODT, complying with
applicable U.S. regulatory requirements, the expected
timing, commencement and outcomes of Biohaven's planned and ongoing
clinical trials; the timing of planned interactions and filings
with the FDA; the timing and outcome of expected regulatory
filings; the potential commercialization of Biohaven's product
candidates; the potential for Biohaven's product candidates to be
first in class or best in class therapies; and the effectiveness
and safety of Biohaven's product candidates. Various important
factors could cause actual results or events to differ materially
from those that may be expressed or implied by forward-looking
statements. Additional important factors to be considered in
connection with forward-looking statements are described in the
"Risk Factors" section of the Company's Annual Report on Form 10-K
filed with the Securities and Exchange Commission on February 25, 2022, and in Biohaven's subsequent
filings with the Securities and Exchange Commission. The
forward-looking statements are made as of the date of this new
release, and Biohaven does not undertake any obligation to update
any forward-looking statements, whether as a result of new
information, future events or otherwise, except as required by
law.
Biohaven Contact
Jennifer Porcelli
201-248-0741
Vice President, Investor Relations
jennifer.porcelli@biohavenpharma.com
Media Contact
Mike
Beyer
Sam Brown Inc.
mikebeyer@sambrown.com
312-961-2502
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-presents-new-migraine-data-at-64th-annual-scientific-meeting-of-the-american-headache-society-including-first-of-its-kind-study-for-nurtec-odt-rimegepant-and-complete-phase-3-data-for-zavegepant-nasal-spray-301566294.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-presents-new-migraine-data-at-64th-annual-scientific-meeting-of-the-american-headache-society-including-first-of-its-kind-study-for-nurtec-odt-rimegepant-and-complete-phase-3-data-for-zavegepant-nasal-spray-301566294.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.