- More than 25% of patients reported being severely or very
severely impacted by their migraine attacks
- Study showed higher levels of migraine-related disability were
associated with increased HCRU costs in primary care and specialty
settings among adults with episodic migraine
- Data suggests that assessment of disability in people with
migraine may support prescribing of cost-effective interventions,
particularly among those with severe disability
NEW HAVEN, Conn., April 14, 2022 /PRNewswire/ -- Biohaven
Pharmaceutical Holding Company Ltd. (NYSE: BHVN) announced the
publication of new real-world research showing that as
migraine-related disability increases, healthcare utilization also
increases among Americans with episodic migraine. Targeting high
disability patients with effective treatments may reduce disability
and improve the cost-effectiveness of medical care among primary
care and specialty providers. These findings were published in the
March issue of Headache: The Journal of Head and Face Pain,
the official journal of the American Headache Society.
Participants in this retrospective, cohort study were identified
using claims and electronic health record data from the Decision
Resources Group database. Adults (aged ≥18 years) were eligible if
they had received a diagnosis of migraine with or without aura, as
defined by International Classification of Disease codes (ICD-9 or
ICD-10) and completed a Migraine Disability Assessment Scale
(MIDAS) questionnaire between January
2016 and December 2018.
Researchers explored the associations between migraine-related
disability, as measured by MIDAS score, and the cost of HCRU for
the 6 months after MIDAS assessment in primary care and specialty
settings.
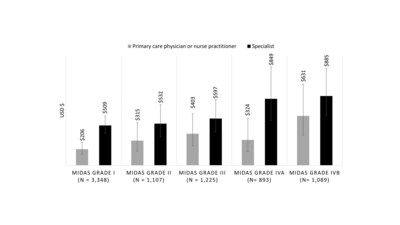
One of the important findings of the study is that among the
7,662 adults who were analyzed, 26% (1,982) reported severe or very
severe disability. This translates to a median of approximately 33
days per quarter (derived from source data) wherein migraine
patients were unable to pursue activities of daily living
(absenteeism) or were severely impaired in these activities
(presenteeism). Adjusted six month cost estimates, summarized in
Figures 1 and 2, showed that higher levels of migraine-related
disability were associated with increased medical and pharmacy
costs.
"This study describes the relationships between MIDAS grades,
healthcare resource utilization, and direct medical costs," said
Richard Lipton, M.D., professor and
vice chair of Neurology at Albert Einstein College of Medicine, who
developed the MIDAS instrument and is senior author on the study.
"Our findings suggest that disability assessments in people with
migraine may be used to target individuals with the most to gain
from effective treatment. The cost offsets of effective treatment
should be assessed in these most disabled and costly patient
groups."
Gil L'Italien Ph.D., Senior Vice President, GHEOR &
Epidemiology, Biohaven, and co-author of the study, observed, "This
analysis, derived from claims and health record data representing
90% of health plans in the United
States further underscores the association between migraine
disability and healthcare resource utilization, and establishes the
MIDAS questionnaire as a useful tool for predicting and potentially
controlling costs associated with migraine management."
About Migraine
Nearly 40 million people in the
U.S. suffer from migraine and the World Health Organization
classifies migraine as one of the 10 most disabling medical
illnesses. Migraine is characterized by debilitating attacks
lasting four to 72 hours with multiple symptoms, including
pulsating headaches of moderate to severe pain intensity that can
be associated with nausea or vomiting, and/or sensitivity to sound
(phonophobia) and sensitivity to light (photophobia). There is a
significant unmet need for new treatments as more than 90 percent
of people with migraine are unable to work or function normally
during an attack.
About Biohaven
Biohaven is a commercial-stage
biopharmaceutical company with a portfolio of innovative,
best-in-class therapies to improve the lives of patients with
debilitating neurological and neuropsychiatric diseases, including
rare disorders. Biohaven's Neuroinnovation™ portfolio includes
FDA-approved NURTEC® ODT (rimegepant) for the acute and
preventive treatment of migraine and a broad pipeline of late-stage
product candidates across three distinct mechanistic platforms:
CGRP receptor antagonism for the acute and preventive treatment of
migraine; glutamate modulation for obsessive-compulsive disorder,
and spinocerebellar ataxia; MPO inhibition for amyotrophic lateral
sclerosis; Kv7 Ion Channel Activators (Kv7), and Myostatin. More
information about Biohaven is available at
www.biohavenpharma.com and Nurtec ODT at www.nurtec.com.
Forward-Looking Statements
This news release includes forward-looking statements within the
meaning of the Private Securities Litigation Reform Act of 1995.
These forward-looking statements involve substantial risks and
uncertainties, including statements regarding the ability to
commercialize NURTEC®️ ODT (rimegepant), delays or
problems in the supply or manufacture of commercial product,
complying with applicable U.S. regulatory requirements, the
expected timing, commencement and outcomes of Biohaven's planned
and ongoing clinical trials, the timing of planned interactions
with the FDA, the timing and outcome of regulatory filings, the
potential commercialization of Biohaven's product candidates, the
potential for Biohaven's product candidates to be first in class or
best in class therapies and the effectiveness and safety of
Biohaven's product candidates. Various important
factors could cause actual results or events to differ materially
from those that may be expressed or implied by our forward-looking
statements. Additional important factors to be considered in
connection with forward-looking statements are described in the
"Risk Factors" section of Biohaven's Annual Report on Form 10-K for
the year ended December 31, 2021,
filed with the Securities and Exchange Commission on February 25, 2022, and Biohaven's subsequent
filings with the Securities and Exchange Commission. The
forward-looking statements are made as of this date and Biohaven
does not undertake any obligation to update any forward-looking
statements, whether as a result of new information, future events
or otherwise, except as required by law.
NURTEC and NURTEC ODT are registered trademarks of Biohaven
Pharmaceutical Ireland DAC. Neuroinnovation is a trademark of
Biohaven Pharmaceutical Holding Company Ltd.
Biohaven Contact
Jennifer Porcelli
Vice President, Investor Relations
jennifer.porcelli@biohavenpharma.com
201-248-0741
Media Contact
Mike
Beyer
Sam Brown Inc.
mikebeyer@sambrown.com
312-961-2502
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-real-world-study-highlights-increased-healthcare-utilization-among-americans-with-episodic-migraine-having-higher-levels-of-migraine-related-disability-301525370.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-real-world-study-highlights-increased-healthcare-utilization-among-americans-with-episodic-migraine-having-higher-levels-of-migraine-related-disability-301525370.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.