NEW HAVEN, Conn., Feb. 22, 2021 /PRNewswire/ -- Biohaven
Pharmaceutical Holding Company Ltd. (NYSE: BHVN), a
commercial-stage biopharmaceutical company with a portfolio of
innovative, late-stage product candidates, today announced that a
hyperimmune globulin mimic (HGM) developed with Biohaven's
proprietary MATE platform has demonstrated functional binding and
neutralization of the SARS-CoV-2 virus, including the strains known
as the "English" and "South African" variants (also known as
B.1.1.7 and B.1.351, respectively). The preliminary experiments,
conducted by Biohaven Labs and an academic collaborator
demonstrated that BHV-1200 substantially reduced viral entry into
cells. Accelerated development of the COVID-19 MATE program has
been supported by the Bill and Melinda Gates Foundation.
Vlad Coric M.D., Chief Executive
Officer of Biohaven commented, "The battle against COVID-19 –like
the virus itself–is dynamic and ever changing. Prophylactic and
therapeutic agents with broad specificity against current and
future strains will be essential in this ongoing fight to beat the
virus. Results from our recent preclinical testing are an exciting
advancement for this platform technology, and shows that our lead
novel MATE conjugate, BHV-1200, exhibits broad and potent antiviral
activity against not only wild type SARS-CoV-2 spike protein but
also against mutations of growing clinical relevance, such as those
associated with reduced susceptibility to therapeutic monoclonal
antibodies and recent SARS-CoV-2 strains." Dr. Coric added, "We
believe that BHV-1200 could lead to enhanced efficacy and other
benefits over convalescent plasma and alternative antibody-based
approaches. Biohaven is excited to advance BHV-1200 into a
full clinical development program. We are deeply grateful to
the vision and critical funding support provided by The Bill &
Melinda Gates Foundation that has accelerated this novel technology
towards the clinic."
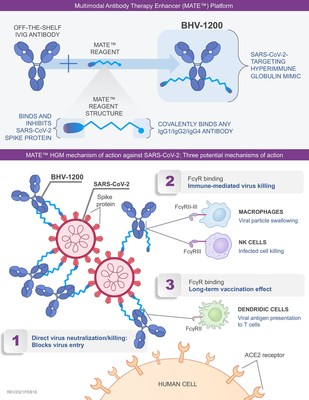
Biohaven's proprietary MATE conjugation technology uses a new
class of synthetic peptide binders to target the spike protein of
SARS-CoV-2 that are then selectively conjugated to commercially
available intravenous immunoglobulin (see Figure 1). The Biohaven
synthetic binders for SARS-CoV-2 were designed to establish a much
wider area and number of contacts with the spike protein than other
agents like monoclonal antibodies. Importantly, the binding and
potent neutralizing activity observed with BHV-1200 was consistent
across multiple strains of the SARS-CoV-2 virus, including the
"English" and "South African" variants. These variants contain
multiple mutations in the spike protein that have been reported to
reduce the binding and neutralizing activity of currently available
antibody-based COVID-19 therapies and sera from SARS-CoV-2 vaccine
recipients. In addition, the in vitro data indicate that BHV-1200
may activate important immune system components including
antibody-dependent cellular phagocytosis (ADCP) and antibody
dependent cellular cytotoxicity (ADCC). Biohaven's proprietary
MATE-conjugation technology could also be used against other
infectious diseases by changing the targeting moiety of its
antibody binders.
Dr. Charles Conway, Chief
Scientific Officer at Biohaven stated, "Our recent in vitro data
provide good evidence that BHV-1200 can neutralize the new strains
of SARS-CoV-2 that have recently appeared across the globe. Our
MATE-conjugation technology could enable commonly used and
commercially available IVIG plasma products, which is not specific
for the SARS-CoV-2 virus, to be redirected to target the viral
spike protein. We look forward to continue to evaluate the
therapeutic utility of this treatment both in the lab and in the
clinic."
About Biohaven
Biohaven is a commercial-stage biopharmaceutical company with a
portfolio of innovative, best-in-class therapies to improve the
lives of patients with debilitating neurological and
neuropsychiatric diseases, including rare disorders. Biohaven's
neuroinnovation portfolio includes FDA-approved NURTEC® ODT
(rimegepant) for the acute treatment of migraine and a broad
pipeline of late-stage product candidates across three distinct
mechanistic platforms: CGRP receptor antagonism for the acute and
preventive treatment of migraine; glutamate modulation for
obsessive-compulsive disorder, Alzheimer's disease, and
spinocerebellar ataxia; and myeloperoxidase (MPO) inhibition for
multiple system atrophy and amyotrophic lateral sclerosis. Biohaven
Labs is a research and discovery arm of the company developing
next-generation, bispecific compounds. More information about
Biohaven is available at www.biohavenpharma.com.
Forward-Looking Statements
This news release includes
forward-looking statements within the meaning of the Private
Securities Litigation Reform Act of 1995. These forward-looking
statements involve substantial risks and uncertainties, including
statements that are based on the current expectations and
assumptions of the Company's management and the ability of BHV-1200
to produce enhanced efficacy following the full clinical
development program. All statements, other than statements of
historical facts, included in this press release regarding the
Company's business and product candidate plans and objectives are
forward-looking statements. Forward-looking statements include
those related to the Company's strategies and objectives and its
ability to achieve those objectives. The use of certain words,
including "believe", "continue", "may", "on track", "expects" and
"will" and similar expressions, are intended to identify
forward-looking statements. Various important factors could cause
actual results or events to differ materially from those that may
be expressed or implied by our forward-looking statements.
Additional important factors to be considered in connection with
forward-looking statements are described in the "Risk Factors"
section of Biohaven's Annual Report on Form 10-K filed with the
Securities and Exchange Commission on February 26,
2020 and Biohaven's Quarterly Report on Form 10-Q for the
quarter ended September 30, 2020 filed with the
Securities and Exchange Commission on November 9, 2020. The
forward-looking statements are made as of this date and Biohaven
does not undertake any obligation to update any forward-looking
statements, whether as a result of new information, future events
or otherwise, except as required by law.
NURTEC and NURTEC ODT are registered trademarks of Biohaven
Pharmaceutical Ireland DAC.
MATE is a trademark of Kleo Pharmaceuticals, Inc. (a subsidiary
of Biohaven and D/B/A Biohaven Labs)
Biohaven Contact
Dr. Vlad
Coric
Chief Executive Officer
Vlad.Coric@biohavenpharma.com
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohavens-bhv-1200-a-multimodal-antibody-therapy-enhancer-mate-demonstrates-effective-neutralization-of-multiple-strains-of-covid-19-301232221.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohavens-bhv-1200-a-multimodal-antibody-therapy-enhancer-mate-demonstrates-effective-neutralization-of-multiple-strains-of-covid-19-301232221.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.