Abbott Labs Enters Into Consent Decree with FDA For Michigan Baby Formula Plant
May 16 2022 - 5:10PM
Dow Jones News
By Denny Jacob
Abbott Laboratories said Monday it has agreed to enter into a
consent decree with the Food and Drug Administration to resume
production and maintain its Sturgis, Mich.-based infant formula
plant.
Once the FDA confirms the initial requirements for start-up have
been met, Abbott could restart the site within two weeks, the
health care products company said. From the time it restarts the
site, the company said it will take six to eight weeks before
products are available on shelves.
The company said it would begin production of EleCare, Alimentum
and metabolic formulas first and then begin production of Similac
and other formulas.
"Our No. 2 priority is getting infants and families the
high-quality formulas they need, and this is a major step toward
re-opening our Sturgis facility so we can ease the nationwide
formula shortage," Chief Executive Robert Ford said in prepared
remarks.
Write to Denny Jacob at denny.jacob@wsj.com
(END) Dow Jones Newswires
May 16, 2022 16:55 ET (20:55 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
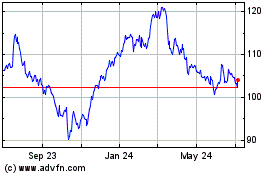
Abbott Laboratories (NYSE:ABT)
Historical Stock Chart
From Mar 2024 to Apr 2024
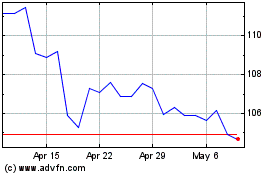
Abbott Laboratories (NYSE:ABT)
Historical Stock Chart
From Apr 2023 to Apr 2024