As filed with the Securities and Exchange Commission on July 7, 2022
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM F-3
REGISTRATION STATEMENT UNDER THE SECURITIES
ACT OF 1933
SCISPARC LTD.
(Exact name of registrant as specified in its charter)
Not Applicable
(Translation of Registrant’s Name into English)
| State of Israel |
|
Not Applicable |
|
(State or other jurisdiction of
incorporation or organization) |
|
(I.R.S. Employer
Identification No.) |
20 Raul Wallenberg Street, Tower A,
Tel Aviv 6971916 Israel
Tel: (+972) (3) 717-5777
(Address and telephone number of registrant’s
principal executive offices)
Puglisi & Associates
850 Library Ave., Suite 204
Newark, DE 19711
Tel: (302) 738-6680
(Name, address, and telephone number of agent for
service)
Copies to:
|
Oded Har-Even, Esq.
Howard E. Berkenblit, Esq.
Sullivan & Worcester LLP
1633 Broadway
New York, NY 10019
Tel: (212) 660-3000 |
Dr. Shachar Hadar, Adv.
Meitar | Law Offices
16 Abba Hillel Silver Rd.
Ramat Gan 52506, Israel
Tel: (+972) (3) 610-3100 |
Approximate date of commencement of proposed sale
to the public: From time to time after the effective date of this Registration Statement.
If only securities being registered on this Form
are being offered pursuant to dividend or interest reinvestment plans, please check the following box. ☐
If any of the securities being registered on this
Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, check the following box. ☒
If this Form is filed to register additional securities
for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration
statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed
pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of
the earlier effective registration statement for the same offering. ☐
If this Form is a registration statement pursuant
to General Instruction I.C. or a post-effective amendment thereto that shall become effective upon filing with the Commission pursuant
to Rule 462(e) under the Securities Act, check the following box. ☐
If this Form is a post-effective amendment to
a registration statement filed pursuant to General Instruction I.C. filed to register additional securities or additional classes of securities
pursuant to Rule 413(b) under the Securities Act, check the following box. ☐
Indicate by check mark whether the registrant
is an emerging growth company as defined in Rule 405 of the Securities Act of 1933.
Emerging growth company ☒
If an emerging growth company that prepares its
financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant has elected not to use the extended transition
period for complying with any new or revised financial accounting standards† provided pursuant to Section 7(a)(2)(B) of the Securities
Act. ☐
† The term “new or revised financial
accounting standard” refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification
after April 5, 2012.
The Registrant hereby amends this Registration
Statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which
specifically states that this Registration Statement shall thereafter become effective in accordance with Section 8(a) of the Securities
Act of 1933 or until this Registration Statement shall become effective on such date as the Securities and Exchange Commission, acting
pursuant to said Section 8(a), may determine.
The
information in this prospectus is not complete and may be changed. The selling shareholder may not sell these securities until the registration
statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities
and is not soliciting an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
Subject
to Completion, dated July 7, 2022
PROSPECTUS

SCISPARC LTD.
Up to 10,638,300 Ordinary
Shares
This prospectus relates to
the resale, by the selling shareholder identified in this prospectus, of up to 10,638,300 ordinary shares, no par value, or the Ordinary
Shares, consisting of (i) up to 335,000 Ordinary Shares held by the selling shareholder, (ii) up to 3,211,100 Ordinary Shares issuable
upon exercise of pre-funded warrants, or the Pre-Funded Warrants, and (iii) up to 7,092,200 Ordinary Shares issuable upon the exercise
of warrants, or the Ordinary Warrants, and, together with the Pre-Funded Warrants, the Warrants.
The selling shareholder is
identified in the table on page 8. No Ordinary Shares or Warrants are being registered hereunder for sale by us. While we will not receive
any proceeds from the sale of the Ordinary Shares by the selling shareholder, we will receive cash proceeds equal to the total exercise
price of the Warrants that are exercised. The exercise price of the Pre-Funded Warrants is $0.001 per Ordinary Share and the exercise
price of the Ordinary Warrants is $2.63 per Ordinary Share. See “Use of Proceeds.” The selling shareholder may sell all or
a portion of the Ordinary Shares from time to time in market transactions through any market on which our Ordinary Shares are then traded,
in negotiated transactions or otherwise, and at prices and on terms that will be determined by the then prevailing market price or at
negotiated prices directly or through a broker or brokers, who may act as agent or as principal or by a combination of such methods of
sale. See “Plan of Distribution.”
Our Ordinary Shares are listed
on the Nasdaq Capital Market, or Nasdaq, under the symbol “SPRC.”
On July 6, 2022, the last reported sale price of our Ordinary Shares was $2.49 per share. We do not intend to apply for
listing of the Warrants on any securities exchange or other nationally recognized trading system. There is no established market for
the Warrants.
We are an emerging growth
company, as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and are subject to reduced public company reporting
requirements.
AN INVESTMENT IN OUR
SECURITIES INVOLVES RISKS. SEE THE SECTION ENTITLED “RISK FACTORS” BEGINNING ON PAGE 3 AND IN OUR ANNUAL REPORT ON FORM
20-F FOR THE FISCAL YEAR ENDED DECEMBER 31, 2021.
Neither the Securities
and Exchange Commission nor any state or other securities commission has approved or disapproved of these securities or determined if
this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus
is , 2022
TABLE OF CONTENTS
You
should rely only on the information contained in this prospectus, including information incorporated by reference herein, and any free
writing prospectus prepared by or on behalf of us or to which we have referred you. Neither we nor the selling shareholder have authorized
anyone to provide you with different information. If anyone provides you with different or inconsistent information, you should not rely
on it. This prospectus does not constitute an offer to sell, or a solicitation of an offer to purchase, the securities offered by this
prospectus in any jurisdiction to or from any person to whom or from whom it is unlawful to make such offer tor solicitation of an offer
in such jurisdiction. The information in this prospectus is accurate only as of the date of this prospectus, regardless of the time of
delivery of this prospectus or any sale of our securities.
For
investors outside of the United States: We have not done anything that would permit this offering or possession or distribution of this
prospectus in any jurisdiction where action for that purpose is required, other than in the United States. You are required to inform
yourselves about and to observe any restrictions relating to this offering and the distribution of this prospectus. In this prospectus,
“we,” “us,” “our,” the “Company” and “SciSparc” refer to SciSparc Ltd.
Our
reporting currency and functional currency is the U.S. dollar. Unless otherwise expressly stated or the context otherwise requires,
references in this prospectus to “NIS” are to New Israeli Shekels, and references to “dollars” or “$”
mean U.S. dollars.
This prospectus incorporates
by reference statistical, market and industry data and forecasts which we obtained from publicly available information and independent
industry publications and reports that we believe to be reliable sources. These publicly available industry publications and reports generally
state that they obtain their information from sources that they believe to be reliable, but they do not guarantee the accuracy or completeness
of the information. Although we believe that these sources are reliable, we have not independently verified the information contained
in such publications.
We
report our financial information under International Financial Reporting Standards, as issued by the International Accounting Standards
Board. None of the financial statements incorporated by reference in this prospectus were prepared in accordance with generally accepted
accounting principles in the United States.
OUR COMPANY
We
are a specialty clinical-stage pharmaceutical company. Our focus is creating and enhancing a portfolio of technologies and assets based
on cannabinoid therapies. With this focus, the Company is currently engaged in the development of the following pharmaceutical compositions
comprising N-acylethanolamines and cannabinoids, such as Palmitoylethanolamide (PEA) and/or Δ9-tetrahydrocannabinol (THC) and/or
non-psychoactive cannabidiol (CBD) and/or other cannabinoid receptor agonists: SCI-110 (formerly THX-110) for the treatment of Tourette
syndrome for the treatment of obstructive sleep apnea and for the treatment of Alzheimer’s disease and agitation; SCI-160 (formerly
THX-160) for the treatment of pain; and SCI-210 (formerly THX-210) for the treatment of Autism Spectrum Disorder and epilepsy.
ABOUT THIS OFFERING
| Ordinary Shares currently outstanding |
|
6,737,840 Ordinary Shares (assumes the exercise in full of the Pre-Funded Warrants but not the Ordinary Warrants). |
| |
|
|
| Ordinary Shares offered by the selling shareholder |
|
Up to 10,638,300 Ordinary Shares, consisting of (i) up to 335,000 Ordinary Shares held by the selling shareholder, (ii) up to 3,211,100 Ordinary Shares issuable upon exercise of the Pre-Funded Warrants, and (iii) up to 7,092,200 Ordinary Shares issuable upon the exercise of the Ordinary Warrants. |
| |
|
|
| Ordinary Shares to be outstanding assuming exercise of the Warrants |
|
13,830,040 Ordinary Shares. |
| |
|
|
| Use of proceeds: |
|
We will not receive any proceeds from the sale
of the Ordinary Shares by the selling shareholder. All net proceeds from the sale of the Ordinary Shares covered by this prospectus will
go to the selling shareholder. However, we will receive cash proceeds equal to the total exercise price of the Warrants that are exercised.
We intend to use the proceeds from the exercise
of the Warrants for working capital, which includes research and development, to advance our technology and general corporate purposes
and pursuing strategic opportunities including expanding our pipeline. See “Use of Proceeds.”
|
| Risk factors: |
|
You should read the “Risk Factors” section starting on
page 3 of this prospectus, and “Item 3. - Key Information – D. Risk Factors” in our Annual Report on Form 20-F for
the year ended December 31, 2021, or the 2021 Annual Report, incorporated by reference herein, and other information included or
incorporated by reference in this prospectus for a discussion of factors to consider carefully before deciding to invest in our
securities. |
| |
|
|
| Nasdaq symbol: |
|
“SPRC.” |
The number of Ordinary
Shares to be outstanding immediately after this offering as shown above assume the exercise of all of the Warrants currently outstanding
are exercised and is based on 6,737,840 Ordinary Shares (assumes the exercise in full of the Pre-Funded Warrants but not the Ordinary
Warrants) outstanding as of June 28, 2022. This number excludes:
| ● | 183,571 Ordinary Shares issuable
upon the exercise of options outstanding under our 2015 Share Option Plan, at a weighted average exercise price of $15.56 per share; |
| ● | 173,572 Ordinary Shares reserved
for issuance and available for future grant under our 2015 Share Option Plan; |
| ● | 2,397,179 Ordinary Shares issuable
upon the exercise of outstanding warrants (that are not included in the Warrants) to purchase 2,397,179 Ordinary Shares, with exercise
prices ranging from $245 to $5.02 per Ordinary Share; and |
| ● | 18,156 Ordinary Shares issuable
upon the exercise of outstanding pre-funded warrants (that are not included in the Warrants) to purchase 18,156 Ordinary Shares, with
exercise prices of $0.001 per Ordinary Share. |
RISK FACTORS
Investing in our securities
involves risks. Please carefully consider the risk factors described in our periodic reports filed with the Securities and Exchange Commission,
or SEC, including those set forth under the caption “Item 3. Key Information - D. Risk Factors” in our 2021 Annual Report,
which is incorporated by reference in this prospectus. Before making an investment decision, you should carefully consider these risks
as well as other information we include or incorporate by reference in this prospectus. You should be able to bear a complete loss of
your investment.
NOTE REGARDING FORWARD-LOOKING
STATEMENTS
Some of the statements made
under “Risk Factors,” “Use of Proceeds,” and elsewhere in this prospectus, including in our 2021 Annual Report,
incorporated by reference herein, and other information included or incorporated by reference in this prospectus, constitute forward-looking
statements. Forward-looking statements are often characterized by the use of forward-looking terminology such as “may,” “will,”
“expect,” “anticipate,” “estimate,” “continue,” “believe,” “should,”
“intend,” “project” or other similar words, but are not the only way these statements are identified.
These forward-looking statements
may include, but are not limited to, statements relating to our objectives, plans and strategies, statements that contain projections
of results of operations or of financial condition, expected capital needs and expenses, statements relating to the research, development,
completion and use of our products, and all statements (other than statements of historical facts) that address activities, events or
developments that we intend, expect, project, believe or anticipate will or may occur in the future.
Forward-looking statements
are not guarantees of future performance and are subject to risks and uncertainties. We have based these forward-looking statements on
assumptions and assessments made by our management in light of their experience and their perception of historical trends, current conditions,
expected future developments and other factors they believe to be appropriate.
Important factors that could
cause actual results, developments and business decisions to differ materially from those anticipated in these forward-looking statements
include, among other things:
| ● | our ability to raise capital
through the issuance of additional securities; |
| ● | our ability to advance the development
our product candidates, including the anticipated starting and ending dates of our anticipated clinical trials; |
| ● | our assessment of the potential
of our product candidates to treat certain indications; |
| ● | our ability to successfully
receive approvals from the U.S. Food and Drug Administration, or FDA, or other regulatory bodies, including approval to conduct clinical
trials, the scope of those trials and the prospects for regulatory approval of, or other regulatory action with respect to our product
candidates, including the regulatory pathway to be designated to our product candidates; |
| ● | the regulatory environment and
changes in the health policies and regimes in the countries in which we operate, including the impact of any changes in regulation and
legislation that could affect the pharmaceutical industry; |
| ● | our ability to commercialize
our existing product candidates and future sales of our existing product candidates or any other future potential product candidates; |
| ● | our ability to meet our expectations
regarding the commercial supply of our product candidates; |
| ● | the overall global economic
environment; |
| ● | the impact of COVID-19 and resulting
government actions on us; |
| ● | the impact of competition and
new technologies; |
| ● | general market, political and
economic conditions in the countries in which we operate; |
| ● | projected capital expenditures
and liquidity; |
| ● | the impact of competition and
new technologies; |
| ● | changes in our strategy; |
| ● | those factors referred to in
“Item 3. Key Information – D. Risk Factors,” “Item 4. Information on the Company,” and “Item 5. Operating
and Financial Review and Prospects,” of our 2021 Annual Report as well other factors in the 2021 Annual Report. |
These statements are only
current predictions and are subject to known and unknown risks, uncertainties, and other factors that may cause our or our industry’s
actual results, levels of activity, performance or achievements to be materially different from those anticipated by the forward-looking
statements. We discuss many of these risks in this prospectus in greater detail under the heading “Risk Factors” and other
risk factors contained in the documents incorporated by reference herein. You should not rely upon forward-looking statements as predictions
of future events.
Although we believe that the
expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, performance, or achievements.
Except as required by law, we are under no duty to update or revise any of the forward-looking statements, whether as a result of new
information, future events or otherwise, after the date of this prospectus.
USE
OF PROCEEDS
We will not receive any proceeds
from the sale of the Ordinary Shares by the selling shareholder. All net proceeds from the sale of the Ordinary Shares covered by this
prospectus will go to the selling shareholder. However, we will receive cash proceeds equal to the total exercise price of the Warrants
that are exercised.
We intend to use the proceeds
from the exercise of the Warrants for working capital, which includes research and development, to advance our technology and general
corporate purposes and pursuing strategic opportunities including expanding our pipeline.
Pending our use of the net
proceeds from the exercise of the Warrants, we may invest the net proceeds in a variety of capital preservation investments, including
short-term, investment grade, interest bearing instruments and U.S. government securities, as decided by our board of directors from time
to time.
capitalization
The following table sets forth
our cash and cash equivalents and our capitalization as of December 31, 2021:
| |
● |
on a pro forma basis to give effect to the issuance of (i) 335,000 Ordinary Shares, Pre-Funded Warrants to purchase 3,211,100 Ordinary Shares and Ordinary Warrants to purchase 7,092,200 Ordinary Shares pursuant to the Purchase Agreement (as defined below), and (ii) the exercise of Pre-Funded Warrants to purchase 100,000 Ordinary Shares; and |
| ● | on an pro forma as adjusted
basis to give effect to the full exercise of the Warrants. |
You should read this table
in conjunction with the section titled “Item 5. Operating and Financial Review and Prospects” of our 2021 Annual Report and
our financial statements and related notes included in our 2021 Annual Report, incorporated by reference herein.
| | |
As of December 31, 2021 | |
| USD in thousands | |
Actual | | |
Pro Forma | | |
Pro Forma
As
Adjusted | |
| Cash | |
$ | 6,875 | | |
| 15,913 | | |
$ | 34,565 | |
| Total assets | |
$ | 9,916 | | |
| 18,954 | | |
$ | 37,606 | |
| Total liabilities | |
$ | 1,775 | | |
| 8,327 | | |
$ | 1,775 | |
| Shareholders’ equity: | |
| | | |
| | | |
| | |
| Share capital and premium | |
$ | 58,541 | | |
| 61,026 | | |
$ | 86,231 | |
| Ordinary Shares, no par value: 25,714,285 Ordinary Shares authorized; 3,091,740 Ordinary Shares issued and outstanding (actual); 3,526,740 Ordinary Shares outstanding (pro forma); 10,618,940 Ordinary Shares outstanding (pro forma as adjusted) | |
| | | |
| | | |
| | |
| Reserve for share-based payment transactions | |
$ | 4,331 | | |
$ | 4,331 | | |
$ | 4,331 | |
| Warrants | |
$ | 5,190 | | |
$ | 5,190 | | |
$ | 5,190 | |
| Foreign currency translation reserve | |
$ | 497 | | |
$ | 497 | | |
$ | 497 | |
| Transactions with non-controlling interests | |
$ | 559 | | |
$ | 550 | | |
$ | 550 | |
| Accumulated loss | |
$ | (60,977 | ) | |
$ | (60,977 | ) | |
$ | (60,977 | ) |
| Total equity | |
$ | 8,141 | | |
$ | 10,626 | | |
$ | 35,831 | |
The table above assumes the
exercise of all of the Warrants currently outstanding are exercised and is based on 3,091,740 Ordinary Shares outstanding as of December
31, 2021. This number excludes:
| ● | 183,571 Ordinary Shares issuable
upon the exercise of options outstanding under our 2015 Share Option Plan, at a weighted average exercise price of $15.56 per share; |
| ● | 173,572 Ordinary Shares reserved
for issuance and available for future grant under our 2015 Share Option Plan; |
| |
● |
2,397,179 Ordinary Shares issuable upon the exercise of outstanding warrants (that are not included in the Warrants) to purchase 2,397,179 Ordinary Shares, with exercise prices ranging from $245 to $5.02 per Ordinary Share; and 18,156 Ordinary Shares issuable upon the exercise of outstanding pre-funded warrants (that are not included in the Warrants) to purchase 18,156 Ordinary Shares, with exercise prices of $0.001 per Ordinary Share. |
SELLING SHAREHOLDER
On
May 27, 2022, we entered into a definitive securities purchase agreement, or the Purchase Agreement, with a healthcare-focused institutional
investor providing for the issuance, in a private placement, of an aggregate of 3,546,100 units and pre-funded units at a purchase
price of $2.82 per unit (or $0.001 less per pre-funded unit). Each unit and pre-funded unit consists of one Ordinary Share (or Ordinary
Share equivalent) and two Ordinary Warrants each exercisable for one Ordinary Share for $2.57 (for a total of 7,092,200 Ordinary Shares
underlying the Ordinary Warrants). The Ordinary Warrants initially had an expiration date of June 1, 2027.
On June 30, 2022, the Company
and the institutional investor entered into a letter agreement and agreed, among other things, to amend: (i) the Ordinary Warrants to:
(a) extend the termination date for exercising the Ordinary Warrants from June 1, 2027 to June 1, 2029; and (b) amend the exercise price
from $2.57 per share to $2.63 per share; and (ii) the Purchase Agreement to reflect a new purchase price of $2.88 per unit (or $0.001
less per pre-funded unit), resulting in additional gross proceeds to the Company of $212,766.
The selling shareholder acquired
the Ordinary Shares being registered for resale pursuant to this prospectus pursuant to the Purchase Agreement. We have agreed to file
the registration statement of which this prospectus forms a part covering the resale of the Ordinary Shares sold pursuant to the Purchase
Agreement, including upon the exercise of the Warrants. We are registering the Ordinary Shares in order to permit the selling shareholder
to offer the Ordinary Shares for resale from time to time.
Other than the relationships
as purchasers under the Purchase Agreement and described herein, to our knowledge, the selling shareholder is not an employee or supplier
of ours or our affiliates. Within the past three years, other than the relationships described herein, the selling shareholder has not
held a position as an officer a director of ours, nor has it had any material relationship of any kind with us or any of our affiliates.
All information with respect to share ownership has been furnished by the selling shareholder, unless otherwise noted. The Ordinary Shares
being offered are being registered to permit public secondary trading of such Ordinary Shares and the selling shareholder may offer all
or part of the Ordinary Shares it owns for resale from time to time pursuant to this prospectus. The selling shareholder has no family
relationships with our officers, other directors or controlling shareholders.
A selling shareholder who
is an affiliate of a broker-dealer and any participating broker-dealer are deemed to be “underwriters” within the meaning
of the Securities Act of 1933, as amended, or the Securities Act, and any commissions or discounts given to any such selling shareholder
or broker-dealer may be regarded as underwriting commissions or discounts under the Securities Act.
The term “selling shareholder”
also includes any transferees, pledgees, donees, or other successors in interest to the selling shareholder named in the table below.
Unless otherwise indicated, to our knowledge, the selling shareholder named in the table below has sole voting and investment power (subject
to applicable community property laws) with respect to the Ordinary Shares set forth opposite its name. We will file a supplement to this
prospectus (or a post-effective amendment to the registration statement of which this prospectus forms a part, if necessary) to name successors
to the selling shareholder who are able to use this prospectus to resell the Ordinary Shares registered hereby.
The table below lists the
selling shareholder and other information regarding the beneficial ownership of the Ordinary Shares held by the selling shareholder.
The second column lists the
number of Ordinary Shares beneficially owned and the percentage ownership represented by the Ordinary Shares beneficially owned by the
selling shareholder, based on its ownership of Ordinary Shares, as of June 28, 2022.
The third column lists the
total Ordinary Shares being offered by this prospectus by the selling shareholder.
The fourth column assumes
the sale of all of the Ordinary Shares offered by the selling shareholder pursuant to this prospectus and lists the percentage ownership
represented by the Ordinary Shares beneficially owned by the selling shareholder assuming the sale of all the Ordinary Shares offered
by the selling shareholder pursuant to this prospectus. The selling shareholder may sell all, some or none of its shares in this offering.
See “Plan of Distribution.”
| Name of Selling Shareholder | |
Ordinary Shares
Beneficially Owned
Prior to Offering(1) | | |
Maximum
Number of
Ordinary Shares to be Sold
Pursuant to this
Prospectus | | |
Ordinary Shares Owned
Immediately After Sale of Maximum Number of Shares in this Offering | |
| | |
Number | | |
Percentage(2) | | |
| | |
Number | | |
Percentage(2) | |
| Armistice Capital
Master Fund Ltd.(3) | |
| 10,638,300 | (4) | |
| 9.99% | (5) | |
| 10,638,300 | | |
| - | | |
| - | |
| (1) |
Beneficial ownership is determined in accordance with SEC rules and generally includes voting or investment power with respect to securities. Ordinary Shares subject to warrants currently exercisable, or exercisable within 60 days of June 28, 2022, are counted as outstanding for computing the percentage of the selling stockholder holding such options or warrants but are not counted as outstanding for computing the percentage of any other selling stockholder. |
| (2) |
Applicable percentage of ownership is based on 6,737,740 Ordinary Shares outstanding as of June 28, 2022 (assumes the exercise in full of the Pre-Funded Warrants but not the Ordinary Warrants). |
| (3) |
The Ordinary Shares are directly held by Armistice Capital Master Fund Ltd., or the Master Fund, a Cayman Islands exempted company, and may be deemed to be indirectly beneficially owned by Armistice Capital, LLC or Armistice, as the investment manager of the Master Fund; and (ii) Steven Boyd, as the Managing Member of Armistice Capital. Armistice and Steven Boyd disclaim beneficial ownership of the reported securities except to the extent of their respective pecuniary interest therein. The address of the Master Fund is c/o Armistice Capital, LLC, 510 Madison Avenue, 7th Floor, New York, NY 10022. |
| (4) |
Consists of 335,000 Ordinary Shares, 7,092,200 Ordinary Shares issuable upon the exercise of Ordinary Warrants and 3,211,100 Ordinary Shares issuable upon the exercise of Pre-Funded Warrants. The Ordinary Warrants are subject to a 4.99% beneficial ownership limitation and the Pre-Funded Warrants are subject to a 9.99% beneficial ownership limitation, each of which prohibit the Master Fund from converting any portion of the warrants to the extent that, following such conversion, the Master Fund’s ownership of our Ordinary Shares would exceed the applicable ownership limitation. |
| (5) |
The percentage gives effect to the 9.99% beneficial ownership limitation set forth in the selling shareholder’s Ordinary Warrant. |
PLAN OF DISTRIBUTION
The selling shareholder of
the securities and any of its pledgees, assignees and successors-in-interest may, from time to time, sell any or all of the securities
covered hereby on the principal trading market or any other stock exchange, market or trading facility on which the securities are traded
or in private transactions. These sales may be at fixed or negotiated prices. A selling shareholder may use any one or more of the following
methods when selling securities:
| ● | ordinary brokerage transactions
and transactions in which the broker-dealer solicits purchasers; |
| ● | block trades in which the broker-dealer
will attempt to sell the securities as agent but may position and resell a portion of the block as principal to facilitate the transaction; |
| ● | purchases by a broker-dealer
as principal and resale by the broker-dealer for its account; |
| ● | an exchange distribution in
accordance with the rules of the applicable exchange; |
| ● | privately negotiated transactions; |
| ● | settlement of short sales; |
| ● | in transactions through broker-dealers
that agree with the selling shareholder to sell a specified number of such securities at a stipulated price per security; |
| ● | through the writing or settlement
of options or other hedging transactions, whether through an options exchange or otherwise; |
| ● | a combination of any such methods
of sale; or |
| ● | any other method permitted pursuant
to applicable law. |
The selling shareholder may
also sell securities under Rule 144 or any other exemption from registration under the Securities Act, if available, rather than under
this prospectus.
Broker-dealers engaged by
the selling shareholder may arrange for other brokers-dealers to participate in sales. Broker-dealers may receive commissions or discounts
from the selling shareholder (or, if any broker-dealer acts as agent for the purchaser of securities, from the purchaser) in amounts to
be negotiated, but, except as set forth in a supplement to this prospectus, in the case of an agency transaction not in excess of a customary
brokerage commission in compliance with Rule 2440 of the Financial Industry Regulatory Authority, or FINRA, and in the case of a principal
transaction a markup or markdown in compliance with FINRA IM-2440.
In connection with the sale
of the securities or interests therein, the selling shareholder may enter into hedging transactions with broker-dealers or other financial
institutions, which may in turn engage in short sales of the securities in the course of hedging the positions they assume. The selling
shareholder may also sell securities short and deliver these securities to close out his short positions, or loan or pledge the securities
to broker-dealers that in turn may sell these securities. The selling shareholder may also enter into option or other transactions with
broker-dealers or other financial institutions or create one or more derivative securities which require the delivery to such broker-dealer
or other financial institution of securities offered by this prospectus, which securities such broker-dealer or other financial institution
may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction).
The selling shareholder and
any broker-dealers or agents that are involved in selling the securities may be deemed to be “underwriters” within the meaning
of the Securities Act in connection with such sales. In such event, any commissions received by such broker-dealers or agents and any
profit on the resale of the securities purchased by them may be deemed to be underwriting commissions or discounts under the Securities
Act. Each selling shareholder has informed the Company that it does not have any written or oral agreement or understanding, directly
or indirectly, with any person to distribute the securities.
We are required to pay certain
fees and expenses incurred by us incident to the registration of the securities. We have agreed to indemnify the selling shareholder against
certain losses, claims, damages and liabilities, including liabilities under the Securities Act.
We agreed to keep this prospectus
effective until the earlier of (i) the date on which the securities may be resold by the Selling Stockholders without registration and
without regard to any volume or manner-of-sale limitations by reason of Rule 144, without the requirement for the Company to be in compliance
with the current public information under Rule 144 under the Securities Act or any other rule of similar effect or (ii) all of the securities
have been sold pursuant to this prospectus or Rule 144 under the Securities Act or any other rule of similar effect. The resale securities
will be sold only through registered or licensed brokers or dealers if required under applicable state securities laws. In addition, in
certain states, the resale securities covered hereby may not be sold unless they have been registered or qualified for sale in the applicable
state or an exemption from the registration or qualification requirement is available and is complied with.
Under applicable rules and
regulations under the Securities Exchange Act of 1934, as amended, or the Exchange Act, any person engaged in the distribution of the
resale securities may not simultaneously engage in market making activities with respect to the Ordinary Shares for the applicable restricted
period, as defined in Regulation M, prior to the commencement of the distribution. In addition, the selling shareholder will be subject
to applicable provisions of the Exchange Act and the rules and regulations thereunder, including Regulation M, which may limit the timing
of purchases and sales of the Ordinary Shares by the selling shareholder or any other person. We will make copies of this prospectus available
to the selling shareholder and have informed it of the need to deliver a copy of this prospectus to each purchaser at or prior to the
time of the sale (including by compliance with Rule 172 under the Securities Act).
Offer Restrictions Outside the United States
Other than in the United States,
no action has been taken by us that would permit a public offering of the securities offered by this prospectus in any jurisdiction where
action for that purpose is required. The securities offered by this prospectus may not be offered or sold, directly or indirectly, nor
may this prospectus or any other offering material or advertisements in connection with the offer and sale of any such securities be distributed
or published in any jurisdiction, except under circumstances that will result in compliance with the applicable rules and regulations
of that jurisdiction. Persons into whose possession this prospectus comes are advised to inform themselves about and to observe any restrictions
relating to the offering and the distribution of this prospectus. This prospectus does not constitute an offer to sell or a solicitation
of an offer to buy any securities offered by this prospectus in any jurisdiction in which such an offer or a solicitation is unlawful.
LEGAL MATTERS
Certain legal matters concerning
this offering were passed upon for us by Sullivan & Worcester LLP, New York, New York. Certain legal matters with respect to the legality
of the issuance of the securities offered by this prospectus were passed upon for us by Meitar | Law Offices, Tel Aviv, Israel.
EXPERTS
The consolidated financial
statements of SciSparc Ltd. appearing in our Annual Report on Form 20-F for the year ended December 31, 2021 have been audited by Kost
Forer Gabbay & Kasierer, Certified Public Accountants (Isr.), a member firm of Ernst & Young Global, an independent registered
public accounting firm, as set forth in their report thereon, included therein. Such consolidated financial statements are incorporated
herein by reference in reliance upon such report given on the authority of said firm as experts in accounting and auditing.
EXPENSES
The following are the estimated
expenses of this offering payable by us with respect to the Ordinary Shares issuable upon exercise of the Warrants. With the exception
of the SEC registration fee, all amounts are estimates and may change:
| SEC registration fee | |
$ | 2,199.16 | |
| Legal fees and expenses | |
$ | 100,000 | |
| Accounting fees and expenses | |
$ | 7,500 | |
| Miscellaneous | |
$ | 5,000 | |
| Total | |
$ | 114,700 | |
ENFORCEABILITY OF CIVIL LIABILITIES
We are incorporated under
the laws of the State of Israel. Service of process upon us and upon our directors and officers and the Israeli experts named in the registration
statement of which this prospectus forms a part, a substantial majority of whom reside outside of the United States, may be difficult
to obtain within the United States. Furthermore, because substantially all of our assets and a substantial of our directors and officers
are located outside of the United States, any judgment obtained in the United States against us or any of our directors and officers may
not be collectible within the United States.
We have been informed by our
legal counsel in Israel, Meitar | Law Offices, that it may be difficult to assert U.S. securities law claims in original actions instituted
in Israel. Israeli courts may refuse to hear a claim based on an alleged violation of U.S. securities laws reasoning Israel is not the
most appropriate forum to bring such a claim. In Israeli courts, the content of applicable U.S. law must be proved as a fact which can
be a time-consuming and costly process and certain matters of procedure will also be governed by Israeli law.
Subject to specified time
limitations and legal procedures, Israeli courts may enforce a U.S. judgment in a civil matter which, subject to certain exceptions, is
non-appealable, including judgments based upon the civil liability provisions of the Securities Act and the Exchange Act and including
a monetary or compensatory judgment in a non-civil matter, provided that among other things:
| ● | the judgment was rendered by
a court which was, according to the laws of the state of the court, competent to render the judgment; |
| ● | the obligation imposed by the
judgment is enforceable according to the rules relating to the enforceability of judgments in Israel and the substance of the judgment
is not contrary to public policy; and |
| ● | the judgment is executory in
the state in which it was given. |
Even if these conditions are met, an Israeli court
will not declare a foreign civil judgment enforceable if:
| ● | the judgment was given in a
state whose laws do not provide for the enforcement of judgments of Israeli courts (subject to exceptional cases); |
| ● | the enforcement of the judgment
is likely to prejudice the sovereignty or security of the State of Israel; |
| ● | the judgment was obtained by
fraud; |
| ● | the opportunity given to the
defendant to bring its arguments and evidence before the court was not reasonable in the opinion of the Israeli court; |
| ● | the judgment was rendered by
a court not competent to render it according to the laws of private international law as they apply in Israel; |
| ● | the judgment is contradictory
to another judgment that was given in the same matter between the same parties and that is still valid; or |
| ● | at the time the action was brought
in the foreign court, a lawsuit in the same matter and between the same parties was pending before a court or tribunal in Israel. |
If
a foreign judgment is enforced by an Israeli court, it generally will be payable in Israeli currency, which can then be converted into
non-Israeli currency and transferred out of Israel. The usual practice in an action before an Israeli court to recover an amount in a
non-Israeli currency is for the Israeli court to issue a judgment for the equivalent amount in Israeli currency at the rate of exchange
in force on the date of the judgment, but the judgment debtor may make payment in foreign currency. Pending collection, the amount of
the judgment of an Israeli court stated in Israeli currency ordinarily will be linked to the Israeli consumer price index plus interest
at the annual statutory rate set by Israeli regulations prevailing at the time. Judgment creditors must bear the risk of unfavorable exchange
rates.
WHERE YOU CAN FIND ADDITIONAL INFORMATION
This prospectus is part of
a registration statement on Form F-3 that we filed with the SEC relating to the securities offered by this prospectus, which includes
additional information. You should refer to the registration statement and its exhibits for additional information. Whenever we make reference
in this prospectus to any of our contracts, agreements or other documents, the references are not necessarily complete and you should
refer to the exhibits attached to the registration statement for copies of the actual contract, agreements or other document.
We are subject to the informational
requirements of the Exchange Act applicable to foreign private issuers. As a “foreign private issuer,” we are exempt from
the rules under the Exchange Act prescribing certain disclosure and procedural requirements for proxy solicitations, and our officers,
directors and principal shareholders are exempt from the reporting and “short-swing” profit recovery provisions contained
in Section 16 of the Exchange Act, with respect to their purchases and sales of shares. In addition, we are not required to file annual,
quarterly and current reports and financial statements with the SEC as frequently or as promptly as U.S. companies whose securities are
registered under the Exchange Act. However, we will file with the SEC, within 120 days after the end of each fiscal year, or such applicable
time as required by the SEC, an annual report on Form 20-F containing financial statements audited by an independent registered public
accounting firm, and may furnish to the SEC, on Form 6-K, unaudited interim financial information.
You can review our SEC
filings and the registration statements by accessing the SEC’s internet site at http://www.sec.gov. We maintain a corporate website
at http://www.scisparc.com. Information contained on, or that can be accessed through, our website does not constitute a part of this
prospectus. We have included our website address in this prospectus solely as an inactive textual reference.
INCORPORATION OF CERTAIN INFORMATION BY REFERENCE
The SEC allows us to “incorporate
by reference” the information we file with it, which means that we can disclose important information to you by referring you to
those documents. The information incorporated by reference is considered to be part of this prospectus and information we file later with
the SEC will automatically update and supersede this information. The documents we are incorporating by reference as of their respective
dates of filing are:
| ● | our Annual Report on Form 20-F
for the year ended December 31, 2021, filed on April 28, 2022; |
| ● | our Reports of Foreign Private Issuer on Form 6-K filed on March
17, 2022 (first and second paragraphs and the section titled “Forward-Looking Statements” of Exhibit 99.1 only), May
11, 2022; May 24,
2022, May 26, 2022
(first, third, fourth and fifth paragraphs and the section titled “Forward-Looking Statements” of Exhibit 99.1 only), May
27, 2022, June 1,
2022, June 2, 2022
(first and second paragraphs and the section titled “Forward-Looking Statements” of Exhibit 99.1 only); June
9, 2022 (first, second and third paragraphs and the section titled “Forward-Looking Statements” of Exhibit 99.1
only) and June 24, 2022
(first, second, third and sixth paragraphs and the section titled “Forward-Looking Statements” of Exhibit 99.1 only); June
29, 2022 (first, second, fifth and sixth paragraphs and the section titled “Forward-Looking Statements” of Exhibit
99.1 only); July 1, 2022; July
7, 2022; and July 7, 2022; and |
| ● | the description of our securities
contained in our Form 8-A filed on December 20, 2021 (File No. 001-38041), and including any further amendment or report filed for the
purpose of updating such description. |
All subsequent annual reports
filed by us pursuant to the Exchange Act on Form 20-F prior to the termination of the offering shall be deemed to be incorporated by reference
to this prospectus and to be a part hereof from the date of filing of such documents. We may also incorporate part or all of any Form
6-K subsequently submitted by us to the SEC prior to the termination of the offering by identifying in such Forms 6-K that they, or certain
parts of their contents, are being incorporated by reference herein, and any Forms 6-K so identified shall be deemed to be incorporated
by reference in this prospectus and to be a part hereof from the date of submission of such documents. Any statement contained in a document
incorporated or deemed to be incorporated by reference herein shall be deemed to be modified or superseded for purposes of this prospectus
to the extent that a statement contained herein or in any other subsequently filed document which also is incorporated or deemed to be
incorporated by reference herein modifies or supersedes such statement. Any such statement so modified or superseded shall not be deemed,
except as so modified or superseded, to constitute a part of this prospectus.
We will provide you without
charge, upon your written or oral request, a copy of any of the documents incorporated by reference in this prospectus, other than exhibits
to such documents which are not specifically incorporated by reference into such documents. Please direct your written or telephone requests
to us at: SciSparc Ltd., 20 Raul Wallenberg Street, Tower A, Tel Aviv, 6971916 Israel. Attention: Oz Adler, Chief Executive Officer and
Chief Financial Officer, telephone number: (+972) (3) 717-5777.

SciSparc Ltd.
Up to 10,638,300 Ordinary
Shares
PROSPECTUS
, 2022
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 8. Indemnification of Directors and Officers
Under the Companies Law, a
company may not exculpate an office holder from liability for a breach of the duty of loyalty. An Israeli company may exculpate an office
holder in advance from liability to the company, in whole or in part, for damages caused to the company as a result of a breach of duty
of care but only if a provision authorizing such exculpation is included in its articles of association. Our Articles of Association contain
such a provision. An Israeli company may not exculpate a director from liability arising out of a prohibited dividend or distribution
to shareholders.
An Israeli company may indemnify
an office holder in respect of the following liabilities and expenses incurred for acts performed as an office holder, either in advance
of an event or following an event provided a provision authorizing such indemnification is contained in its articles of association:
| ● | a financial liability imposed
on him or her in favor of another person pursuant to a judgment, including a settlement or arbitrator’s award approved by a court.
However, if an undertaking to indemnify an office holder with respect to such liability is provided in advance, then such an undertaking
must be limited to events which, in the opinion of the board of directors, can be foreseen based on the company’s activities when
the undertaking to indemnify is given, and to an amount or according to criteria determined by the board of directors as reasonable under
the circumstances, and such undertaking shall detail the abovementioned events and amount or criteria; |
| ● | reasonable litigation expenses,
including legal fees, incurred by the office holder (a) as a result of an investigation or proceeding instituted against him or her by
an authority authorized to conduct such investigation or proceeding, provided that (i) no indictment was filed against such office holder
as a result of such investigation or proceeding; and (ii) no financial liability, such as a criminal penalty, was imposed upon him or
her as a substitute for the criminal proceeding as a result of such investigation or proceeding or, if such financial liability was imposed,
it was imposed with respect to an offense that does not require proof of criminal intent; and (b) in connection with a monetary sanction; |
| ● | reasonable litigation expenses,
including legal fees, incurred by the office holder or imposed by a court (i) in proceedings instituted against him or her by the company,
on its behalf or by a third party, or (ii) in connection with criminal proceedings in which the office holder was acquitted, or (iii)
as a result of a conviction for a crime that does not require proof of criminal intent; and |
| ● | expenses, including reasonable
litigation expenses and legal fees, incurred by an office holder in relation to an administrative proceeding instituted against such
office holder, or certain compensation payments made to an injured party imposed on an office holder by an administrative proceeding,
pursuant to certain provisions of the Israeli Securities Law. |
An Israeli company may insure
an office holder against the following liabilities incurred for acts performed as an office holder if and to the extent provided in the
company’s articles of association:
| ● | a breach of the duty of loyalty
to the company, to the extent that the office holder acted in good faith and had a reasonable basis to believe that the act would not
prejudice the company; |
| ● | a breach of the duty of care
to the company or to a third party, including a breach arising out of the negligent conduct of the office holder; |
| ● | a financial liability imposed
on the office holder in favor of a third party; |
| ● | a financial liability imposed
on the office holder in favor of a third party harmed by a breach in an administrative proceeding; and |
| ● | expenses, including reasonable
litigation expenses and legal fees, incurred by the office holder as a result of an administrative proceeding instituted against him
or her, pursuant to certain provisions of the Israeli Securities Law. |
An Israeli company may not
indemnify or insure an office holder against any of the following:
| ● | a breach of the duty of loyalty,
except to the extent that the office holder acted in good faith and had a reasonable basis to believe that the act would not prejudice
the company; |
| ● | a breach of duty of care committed
intentionally or recklessly, excluding a breach arising out of the negligent conduct of the office holder; |
| ● | an act or omission committed
with intent to derive illegal personal benefit; or |
| ● | a fine, monetary sanction or
forfeit levied against the office holder. |
Under the Israeli Companies
Law, exculpation, indemnification and insurance of office holders must be approved by the compensation committee, the board of directors
(and, with respect to directors and the chief executive officer, by the shareholders). However, under regulations promulgated under the
Companies Law, the insurance of office holders shall not require shareholder approval and may be approved by only the compensation committee,
if the engagement terms are determined in accordance with the company’s compensation policy and that policy was approved by the
shareholders by the same special majority required to approve a compensation policy, provided that the insurance policy is on market terms
and the insurance policy is not likely to materially impact the company’s profitability, assets or obligations.
Our Articles of Association
allow us to exculpate, indemnify and insure our office holders for any liability imposed on them as a consequence of an act (including
any omission) which was performed by virtue of being an office holder. Our office holders are currently covered by a directors and officers’
liability insurance policy.
We have entered into agreements
with each of our directors and executive officers exculpating them in advance from liability to us for damages caused to us as a result
of a breach of duty of care, and undertaking to indemnify them. This exculpation and indemnification is limited both in terms of amount
and coverage and it covers certain amounts regarding administrative proceedings insurable or indemnifiable under the Companies Law and
our Articles of Association.
In the opinion of the SEC,
however, indemnification of directors and office holders for liabilities arising under the Securities Act, is against public policy and
therefore unenforceable.
There is no pending litigation
or proceeding against any of our office holders as to which indemnification is being sought, nor are we aware of any pending or threatened
litigation that may result in claims for indemnification by any office holder.
Item 9. Exhibits
| Exhibit
Number |
|
Description
of Document |
| |
|
|
| 3.1 |
|
Amended and Restated Articles of Association of SciSparc Ltd. (unofficial English translation from Hebrew original) (filed as Exhibit 1.1 to Form 20-F (File No. 001-38041) as filed on April 28, 2022 and incorporated herein by reference). |
| |
|
|
| 4.1 |
|
Form
of Ordinary Share Purchase Warrant, (filed as Exhibit 99.5 to Form 6-K (File No. 001-38041) filed on May 27, 2022, and incorporated
herein by reference). |
| |
|
|
| 4.2 |
|
Form
of Pre-Funded Ordinary Share Purchase Warrant, (filed as Exhibit 99.6 to Form 6-K (File No. 001-38041) filed on May 27, 2022, and
incorporated herein by reference). |
| |
|
|
| 4.3 |
|
Amended and Restated Ordinary Share Purchase Warrant, dated June 30, 2022 (filed as Exhibit 99.2 to Form 6-K (File No. 001-38041) filed on July 1, 2022, and incorporated herein by reference). |
| |
|
|
| 5.1 |
|
Opinion
of Meitar | Law Offices, Israeli counsel to the Registrant. |
| |
|
|
| 10.1 |
|
Letter Agreement by and between SciSparc Ltd. and the investor named therein, dated June 30, 2022 (filed as Exhibit 99.1 to Form 6-K (File No. 001-38041) filed on July 1, 2022, and incorporated herein by reference). |
| |
|
|
| 23.1 |
|
Consent
of Kost Forer Gabbay & Kasierer, Certified Public Accountants (Isr.), a member firm of Ernst & Young Global, with respect
to the financial statements of SciSparc Ltd. |
| |
|
|
| 23.2 |
|
Consent
of Meitar | Law Offices (included in Exhibit 5.1). |
| |
|
|
| 24.1 |
|
Power
of Attorney. |
| |
|
|
| 107 |
|
Filing
Fee Table. |
Item 10. Undertakings
| (a) | The undersigned Registrant hereby
undertakes: |
1. To file, during any period
in which offers or sales are being made, a post-effective amendment to this registration statement:
(i) To include any prospectus required
by section 10(a)(3) of the Securities Act of 1933;
(ii) To reflect in the prospectus any
facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which,
individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding
the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed
that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the
form of prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no
more than 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the
effective registration statement.
(iii) To include any material information
with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information
in the registration statement;
provided, however, that paragraphs (a)(1)(i),
(a)(1)(ii) and a(l)(iii) do not apply if the registration statement is on Form S-3 or Form F-3 and the information required to be included
in a post-effective amendment by those paragraphs is contained in reports filed with or furnished to the SEC by the registrant pursuant
to Section 13 or Section 15(d) of the Securities Exchange Act of 1934 that are incorporated by reference in the registration statement,
or is contained in a form of prospectus filed pursuant to Rule 424(b) that is part of the registration statement.
(2) That, for the purpose
of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration
statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial
bona fide offering thereof.
(3) To remove from registration
by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
(4) To file a post-effective
amendment to the registration statement to include any financial statements required by Item 8.A. of Form 20-F at the start of any delayed
offering or throughout a continuous offering. Financial statements and information otherwise required by Section 10(a)(3) of the Act need
not be furnished, provided, that the registrant includes in the prospectus, by means of a post-effective amendment, financial statements
required pursuant to this paragraph (a)(4) and other information necessary to ensure that all other information in the prospectus is at
least as current as the date of those financial statements. Notwithstanding the foregoing, with respect to registration statements on
Form F-3, a post-effective amendment need not be filed to include financial statements and information required by Section 10(a)(3) of
the Act or Rule 3-19 of this chapter if such financial statements and information are contained in periodic reports filed with or furnished
to the Commission by the registrant pursuant to Section 13 or Section 15(d) of the Securities Exchange Act of 1934 that are incorporated
by reference in the Form F-3.
(5) That, for the purpose
of determining liability under the Securities Act to any purchaser:
| (i) | If the Registrant is relying
on Rule 430B: |
| A. | Each prospectus filed by the
Registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as of the date the filed prospectus was
deemed part of and included in the registration statement; and |
| B. | Each prospectus required to
be filed pursuant to Rule 424(b)(2), (b)(5), or (b)(7) as part of a registration statement in reliance on Rule 430B relating to an offering
made pursuant to Rule 415(a)(1)(i), (vii), or (x) for the purpose of providing the information required by section 10(a) of the Securities
Act of 1933 shall be deemed to be part of and included in the registration statement as of the earlier of the date such form of prospectus
is first used after effectiveness or the date of the first contract of sale of securities in the offering described in the prospectus.
As provided in Rule 430B, for liability purposes of the issuer and any person that is at that date an underwriter, such date shall be
deemed to be a new effective date of the registration statement relating to the securities in the registration statement to which that
prospectus relates, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. Provided,
however, that no statement made in a registration statement or prospectus that is part of the registration statement or made
in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration
statement will, as to a purchaser with a time of contract of sale prior to such effective date, supersede or modify any statement that
was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately
prior to such effective date. |
| |
(i) |
If the registrant is subject to Rule 430C, each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use. |
(6) That, for the purpose
of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities
the undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration
statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to
such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will
be considered to offer or sell such securities to such purchaser:
| |
(i) |
Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424; |
| |
(ii) |
Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant; |
| |
(iii) |
The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and |
| |
(iv) |
Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser. |
(b) Insofar as indemnification for liabilities
arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the Registrant pursuant to
the foregoing provisions, or otherwise, the Registrant has been advised that in the opinion of the Securities and Exchange Commission
such indemnification is against public policy as expressed in the Securities Act of 1933 and is, therefore, unenforceable. In the event
that a claim for indemnification against such liabilities (other than the payment by the Registrant of expenses incurred or paid by a
director, officer or controlling person of the Registrant in the successful defense of any action, suit or proceeding) is asserted by
such director, officer or controlling person in connection with the securities being registered, the Registrant will, unless in the opinion
of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether
such indemnification by it is against public policy as expressed in the Securities Act of 1933, as amended, and will be governed by the
final adjudication of such issue.
SIGNATURES
Pursuant to the requirement
of the Securities Act of 1933, the Registrant certifies that it has reasonable grounds to believe that it meets all of the requirements
for filing on Form F-3 and has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized,
the City of Tel Aviv, State of Israel on July 7, 2022.
| |
SCISPARC LTD. |
| |
|
|
| |
By: |
/s/ Oz Adler |
| |
|
Oz Adler |
| |
|
Chief Executive Officer |
POWER OF ATTORNEY
The undersigned officers and
directors of SciSparc Ltd. hereby constitute and appoint Oz Adler and Amitay Weiss with full power of substitution, our true and lawful
attorney-in-fact and agent to take any actions to enable the Company to comply with the Securities Act, and any rules, regulations and
requirements of the SEC, in connection with this registration statement on Form F-3, including the power and authority to sign for us
in our names in the capacities indicated below any and all further amendments to this registration statement and any other registration
statement filed pursuant to the provisions of Rule 462 under the Securities Act.
Pursuant to the requirements
of the Securities Act of 1933, this registration statement has been signed by each of the following persons in the capacities and on the
dates indicated:
| Signature |
|
Title |
|
Date |
| |
|
|
|
|
| /s/ Oz Adler |
|
Chief Executive Officer and Chief Financial Officer |
|
July 7, 2022 |
| Oz Adler |
|
(Principal Executive Officer and Principal Financial Officer) |
|
|
| |
|
|
|
|
| /s/ Amitay Weiss |
|
Chairman of the Board of Directors |
|
July 7, 2022 |
| Amitay Weiss |
|
|
|
|
| |
|
|
|
|
| /s/ Amnon Ben Shay |
|
Director |
|
July 7, 2022 |
| Amnon Ben Shay |
|
|
|
|
| |
|
|
|
|
| /s/ Alon Dayan |
|
Director |
|
July 7, 2022 |
| Alon Dayan |
|
|
|
|
| |
|
|
|
|
| /s/ Itschak Shrem |
|
Director |
|
July 7, 2022 |
| Itschak Shrem |
|
|
|
|
| |
|
|
|
|
| /s/ Liat Sidi |
|
Director |
|
July 7, 2022 |
| Liat Sidi |
|
|
|
|
| |
|
|
|
|
| /s/ Lior Vider |
|
Director |
|
July 7, 2022 |
| Lior Vider |
|
|
|
|
SIGNATURE OF AUTHORIZED REPRESENTATIVE IN THE
UNITED STATES
Pursuant to the Securities
Act of 1933, as amended, the undersigned, Puglisi & Associates duly authorized representative in the United States of SciSparc Ltd.,
has signed this registration statement on July 7, 2022.
| |
Puglisi & Associates. |
| |
|
| |
/s/ Donald J. Puglisi |
| |
Donald J. Puglisi
Managing Director |
II-7
SciSparc (NASDAQ:SPRC)
Historical Stock Chart
From Mar 2024 to Apr 2024
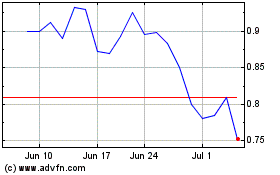
SciSparc (NASDAQ:SPRC)
Historical Stock Chart
From Apr 2023 to Apr 2024