Filed Pursuant to Rule 424(b)(5)
Registration No. 333-231922
PROSPECTUS Supplement
(To Prospectus dated June 26, 2019)

8,407,411
Shares of Common Stock
We are offering 8,407,411 shares of our common stock pursuant
to this prospectus supplement and accompanying prospectus to certain institutional investors.
In a concurrent private placement, we are selling to our affiliate,
Syntone Ventures LLC, or Syntone, 823,045 shares of our common stock at a purchase price of $1.215 per share, for an aggregate
purchase price of approximately $1.0 million. The shares being issued in the concurrent private placement to Syntone are being
offered pursuant to the exemption provided in Section 4(a)(2) under the Securities Act of 1933, as amended, or the Securities
Act, and Rule 506 promulgated thereunder and are not being registered under the Securities Act at this time or offered pursuant
to this prospectus supplement and the accompanying prospectus.
Our common stock is traded on The Nasdaq Capital Market under
the symbol “OTLK.” On June 19, 2020, the closing sale price of our common stock on The Nasdaq Capital Market was
$1.35 per share.
As of the date of this prospectus supplement, the aggregate
market value of our outstanding shares of common stock held by non-affiliates was $56,782,890, based on 113,935,910 outstanding
shares of common stock, of which 42,061,400 shares were held by non-affiliates, and a price of $1.35 per share, which was the last
reported sale price of our common stock on The Nasdaq Capital Market on June 19, 2020. As of the date of this prospectus supplement,
we have sold $7,720,000 of securities pursuant to General Instruction I.B.6. of Form S-3 during the prior 12-month calendar
period that ends on, and includes, the date of this prospectus (but excluding this offering). In no event will we sell securities
pursuant to such registration statement in a public primary offering with value exceeding more than one-third of our public float
in any 12-month calendar period so long as our public float remains below $75.0 million and General Instruction I.B.6 of Form S-3
continues to apply to us.
This
investment involves a high degree of risk. See “Risk Factors” on page S-4
of this prospectus supplement and any similar section contained in the accompanying prospectus and in the documents that
are incorporated by reference herein and therein.
We
have engaged H.C. Wainwright & Co., LLC, or the placement agent, as our exclusive placement agent in connection with
this offering. The placement agent has no obligation to buy any of the securities from us or to arrange for the purchase or sale
of any specific number or dollar amount of securities. We have agreed to pay the placement agent the placement agent fees set
forth in the table below. See “Plan of Distribution” beginning on page S-11
of this prospectus supplement for more information regarding these arrangements.
|
|
|
Per Share
|
|
|
Total
|
|
|
Offering price
|
|
$
|
1.215
|
|
|
$
|
10,215,004
|
|
|
Placement agent’s fees(1)
|
|
$
|
0.08505
|
|
|
$
|
715,050
|
|
|
Proceeds, before expenses, to us(2)
|
|
$
|
1.12995
|
|
|
$
|
9,499,954
|
|
(1) In
addition, we have agreed to pay the placement agent a management fee of 1.0% of the aggregate gross proceeds raised in this offering,
to pay the placement agent for certain of its expenses, and to issue to the placement agent (or its designees) warrants to purchase
shares of common stock equal to 7.0% of the aggregate number of shares of common stock issued in this offering. See “Plan
of Distribution” beginning on page S-11 of this prospectus supplement
for more information on placement agent compensation.
(2) The amount of the offering proceeds to us presented
in this table does not take into account the sale of our shares to Syntone in the concurrent private placement, nor the exercise
of any of the placement agent warrants.
Neither the Securities and Exchange Commission nor any state
securities commission has approved or disapproved of these securities or determined if this prospectus supplement or the accompanying
prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
Delivery of the shares of common stock is expected to be made
on or about June 24, 2020, subject to the satisfaction of certain closing conditions.
H.C. Wainwright &
Co.
The date of this prospectus supplement
is June 22, 2020.
TABLE OF CONTENTS
Prospectus Supplement
Prospectus
ABOUT THIS PROSPECTUS Supplement
This
document is in two parts. The first part is the prospectus supplement, including the documents incorporated by reference, which
describes the specific terms of this offering. The second part, the accompanying prospectus, including the documents incorporated
by reference, provides more general information. Generally, when we refer to this prospectus, we are referring to both parts of
this document combined. Before you invest, you should carefully read this prospectus supplement, the accompanying prospectus,
all information incorporated by reference herein and therein, as well as the additional information described under “Where
You Can Find More Information” on page S-13 and “Incorporation of Certain
Information by Reference” on page S-14 of this prospectus supplement. These documents
contain information you should consider when making your investment decision. This prospectus supplement may add, update or change
information contained in the accompanying prospectus. To the extent that any statement that we make in this prospectus supplement
is inconsistent with statements made in the accompanying prospectus or any documents incorporated by reference, the statements
made in this prospectus supplement will be deemed to modify or
supersede those made in the accompanying prospectus and such documents incorporated by reference.
You should rely only on the information contained or incorporated
by reference in this prospectus supplement, the accompanying prospectus and in any free writing prospectuses we authorize for use
in connection with this offering. Neither we nor the placement agent have authorized any other person to provide you with any information
that is different. If anyone provides you with different or inconsistent information, you should not rely on it. We are offering
to sell, and seeking offers to buy, the securities offered hereby only in jurisdictions where offers and sales are permitted. The
distribution of this prospectus supplement and the offering of the securities offered hereby in certain jurisdictions may be restricted
by law. Persons outside the United States who come into possession of this prospectus supplement must inform themselves about,
and observe any restrictions relating to, the offering of the securities offered hereby and the distribution of this prospectus
supplement outside the United States. This prospectus supplement does not constitute, and may not be used in connection with, an
offer to sell, or a solicitation of an offer to buy, any securities offered by this prospectus supplement by any person in any
jurisdiction in which it is unlawful for such person to make such an offer or solicitation.
The information appearing in this prospectus supplement, the
accompanying prospectus or any related free writing prospectus that we authorize for use in connection with this offering is accurate
only as of the date on the front of the document and any information we have incorporated by reference is accurate only as of the
date of such document incorporated by reference, regardless of the time of delivery of this prospectus supplement or any related
free writing prospectus, or any sale of a security. Our business, financial condition, results of operations and prospects may
have changed since those dates.
This prospectus supplement, the accompanying prospectus
and the documents incorporated by reference herein and therein contain summaries of certain provisions contained in some of
the documents described herein and therein, but reference is made to the actual documents for complete information. All of
the summaries are qualified in their entirety by the actual documents. Copies of some of the documents referred to have been
filed, will be filed or will be incorporated by reference as exhibits to the registration statement of which this prospectus
supplement and the accompanying prospectus are a part, and you may obtain copies of those documents as described below under
the heading “Where You Can Find More Information.”
Except as otherwise indicated or unless the context otherwise
requires, references to “company,” “we,” “us,” “our” or “Outlook Therapeutics,”
refer to Outlook Therapeutics, Inc. and its consolidated subsidiaries.
Our name “Outlook Therapeutics,” the Outlook Therapeutics
logo, LYTENAVA and other trademarks or service marks of Outlook Therapeutics, Inc. appearing in this prospectus supplement,
the accompanying prospectus and any related free writing prospectus and the information incorporated by reference herein or therein
are the property of Outlook Therapeutics, Inc. Other trademarks, service marks or trade names appearing in this prospectus
supplement, the accompanying prospectus and any related free writing prospectus and the information incorporated by reference herein
or therein are the property of their respective owners. We do not intend our use or display of other companies’ trade names,
trademarks or service marks to imply a relationship with, or endorsement or sponsorship of us by, these other companies.
PROSPECTUS Supplement
SUMMARY
This
summary highlights selected information about us, this offering and the information appearing elsewhere in this prospectus supplement,
the accompanying prospectus and the documents incorporated by reference herein and therein and does not contain all of the information
that you need to consider in making your investment decision. You should carefully read the entire prospectus supplement, the
accompanying prospectus and any related free writing prospectus that we authorize for use in connection with this offering, including
the risks of investing in our securities discussed under the heading “Risk Factors” beginning on page S-4 of
this prospectus supplement and under similar headings in the documents incorporated by reference. You should also carefully read
the information incorporated by reference into this prospectus supplement and the accompanying prospectus, including our financial
statements and related notes, and the exhibits to the registration statement of which this prospectus supplement and the
accompanying prospectus are a part, before making your investment decision.
Overview
We are a late clinical-stage biopharmaceutical
company working to develop the first ophthalmic formulation of bevacizumab approved by the U.S. Food and Drug Administration, or
the FDA, for use in retinal indications. Our goal is to launch as the first and only approved bevacizumab in the United States,
Europe, Japan and other markets for the treatment of wet age-related macular degeneration, or wet AMD, diabetic macular edema,
or DME, and branch retinal vein occlusion, or BRVO.
ONS-5010 (LYTENAVA (bevacizumab-vikg))
is an investigational ophthalmic formulation of bevacizumab under development to be administered as an intravitreal injection for
the treatment of wet AMD and other retinal diseases. Bevacizumab is a full-length, humanized anti-VEGF (Vascular Endothelial Growth
Factor) recombinant monoclonal antibody that inhibits VEGF and associated angiogenic activity. The study design for our Phase 3
clinical program to evaluate ONS-5010 as an ophthalmic formulation of bevacizumab was reviewed at an end of Phase 2 meeting with
the FDA in April 2018, and we filed our investigational new drug application with the FDA in the first quarter of calendar
2019.
Currently, the cancer drug Avastin
(bevacizumab) is used off-label for the treatment of wet AMD and other retinal diseases such as DME and BRVO even though
Avastin has not been approved by regulatory authorities for use in these diseases. If the ONS-5010 clinical program is
successful, it will support our plans to submit for regulatory approval in multiple markets in 2021 including the United
States, Europe and Japan, as well as other markets. Because there are no approved bevacizumab products for the treatment of
retinal diseases in such major markets, we are developing ONS-5010 as a standard Biologics License Application and not using
the biosimilar drug development pathway that would be required if Avastin were an approved drug for the targeted diseases. If
approved, we believe ONS-5010 has potential to mitigate risks associated with off-label use of bevacizumab. Off-label use of
bevacizumab is currently estimated to account for at least 50% of all wet AMD prescriptions in the United States.
Company Information
We initially incorporated in January 2010 in New Jersey,
in October 2015, we reincorporated in Delaware by merging with and into a Delaware corporation and in November 2018,
we changed our name to Outlook Therapeutics, Inc. Our headquarters are located at 4260 U.S. Route 1, Monmouth Junction, New
Jersey, 08552, and our telephone number at that location is (609) 619-3990. Our website address is www.outlooktherapeutics.com.
The information contained on, or that can be accessed through, our website is not part of, and is not incorporated by reference
into this prospectus supplement or the accompanying prospectus.
The Offering
|
|
|
|
Common stock offered by us
|
|
8,407,411 shares
|
|
|
|
|
|
Offering price per share
|
|
$1.215
|
|
|
|
|
|
Shares of common stock to be outstanding immediately after this offering
|
|
123,166,366 shares (assuming no exercise of the placement agent warrants but including the 823,045 shares of common stock to be issued to Syntone in the concurrent private placement)
|
|
|
|
|
|
Concurrent private placement
|
|
In a concurrent private placement, we are also selling to our affiliate, Syntone, 823,045 shares of our common stock at a purchase price of $1.215 per share, for an aggregate purchase price of approximately $1.0 million. The shares being issued in the concurrent private placement to Syntone are being offered pursuant to the exemption provided in Section 4(a)(2) under the Securities Act and Rule 506 promulgated thereunder and are not being registered under the Securities Act at this time or offered pursuant to this prospectus supplement and the accompanying prospectus
|
|
|
|
|
|
Use of proceeds
|
|
We estimate that our net proceeds from this offering will be
approximately $8.9 million after deducting placement agent fees and other estimated offering expenses payable by us. We intend
to use the net proceeds from this offering for working capital and general corporate
purposes, including in support of our ONS-5010 development program. See the section titled “Use of Proceeds” on
page S-7 in this prospectus supplement.
|
|
|
|
|
|
Risk factors
|
|
Investment in our securities involves
a high degree of risk. You should read the section titled “Risk Factors” on page S-4 in this prospectus supplement,
the accompanying prospectus and in the documents incorporated
by reference herein and therein for a discussion of factors to consider before investing in our securities.
|
|
|
|
|
|
Nasdaq Capital Market symbol
|
|
“OTLK”
|
The number of shares of common stock outstanding is based on
113,935,910 shares of common stock outstanding as of March 31, 2020, on a pro forma basis including 16,000,000 shares of common
stock issued to Syntone on June 2, 2020 in a private placement and 8,184,718 shares of common stock issued to the holders
of our convertible senior secured notes upon conversion of an aggregate principal amount and accrued interest of $5.0 million from
April 1, 2020 through June 22, 2020, and excludes:
|
|
·
|
2,470 shares of common stock issuable upon the exercise of outstanding
performance-based stock unit awards, or PSUs, whose terms provide for settlement in shares of common stock or cash at our discretion,
with a weighted-average exercise price of $49.97 per share;
|
|
|
·
|
2,213,470 shares of common stock issuable upon exercise of outstanding
stock options granted under the our 2015 Equity Incentive Plan, or the 2015 Plan, with a weighted-average exercise price of $2.33
per share;
|
|
|
·
|
1,639,404 shares of common stock reserved for future issuance under
the 2015 Plan, as well as any future automatic increases in the number of shares of common stock reserved for future issuance under
the 2015 Plan;
|
|
|
·
|
68,145 shares of common stock reserved for future issuance under our
2016 Employee Stock Purchase Plan, or the ESPP, as well as any automatic increases in the number of shares of common stock reserved
for future issuance under the ESPP;
|
|
|
·
|
shares of common stock issuable upon conversion of outstanding senior
secured convertible notes having an aggregate principal amount of $3.0 million as of the date of this prospectus supplement, which
are convertible, at the option of the holder, into shares of our common stock at a conversion price equal to 90% of the two lowest
closing bid prices in the 20 trading days immediately preceding such conversion, subject to a floor price of $0.232 per share;
and
|
|
|
·
|
6,463,329 shares of common stock issuable upon exercise of outstanding
warrants with a weighted-average exercise price of $2.52 per share.
|
Unless otherwise indicated, all information in this prospectus
supplement assumes no settlement of the outstanding PSUs or exercise of options or warrants described above and no exercise of
the placement agent warrants to be issued as compensation to the placement agent for this offering.
RISK FACTORS
Investing
in our securities involves a high degree of risk. Before deciding whether to invest in our securities, you should consider carefully
the risks and uncertainties described below, together with the risks and uncertainties discussed under the heading “Risk
Factors” contained in our Annual Report on Form 10-K for the year ended September 30, 2019 as filed with the SEC,
and as incorporated by reference herein, and as the same may be amended, supplemented or superseded by the risks and uncertainties
described under similar headings in the other documents that are filed after the date hereof and incorporated by reference herein.
The risks described in these documents are not the only ones we face, but those that we consider to be
material. There may be other unknown or unpredictable economic, business, competitive, regulatory or other factors that could
have material adverse effects on our future results. Past financial performance may not be a reliable indicator of future performance,
and historical trends should not be used to anticipate results or trends in future periods. If any of these risks actually occurs,
our business, financial condition, results of operations or cash flow could be seriously harmed. This could cause the trading
price of our common stock to decline, resulting in a loss of all or part of your investment. Please also read carefully the section
herein titled “Special Note Regarding Forward-Looking Statements” on page S-6.
Our stock price is volatile and your investment may suffer
a decline in value.
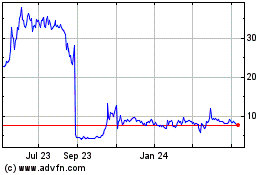
The closing market price for our common stock has varied between
a high of $7.68 on April 4, 2019, and a low of $0.51 on March 18, 2020, in the twelve-month period ended March 31,
2020. As a result of fluctuations in the price of our common stock, you may be unable to sell your shares at or above the price
you paid for them. The market price of our common stock is likely to continue to be volatile and subject to significant price and
volume fluctuations in response to market, industry and other factors, including the risk factors described under the section captioned
“Risk Factors” contained in our Annual Report on Form 10-K for the year ended September 30, 2019, which is
incorporated by reference in this prospectus supplement and the accompanying prospectus in its entirety. The market price of our
common stock may also be dependent upon the valuations and recommendations of the analysts who cover our business. If the results
of our business do not meet these analysts’ forecasts, the expectations of investors or the financial guidance we provide
to investors in any period, the market price of our common stock could decline.
In addition, the stock markets in general, and the markets for
biotechnology stocks in particular, have experienced significant volatility that has often been unrelated to the operating performance
of particular companies. These broad market fluctuations may adversely affect the trading price of our common stock and, consequently,
adversely affect the price at which you could sell the shares that you purchase in this offering. In the past, following periods
of volatility in the market or significant price declines, securities class-action litigation has often been instituted against
companies. Such litigation, if instituted against us, could result in substantial costs and diversion of management’s attention
and resources, which could materially and adversely affect our business, financial condition, results of operations and growth
prospects.
If you purchase shares in this offering, you will suffer
immediate dilution of your investment. You will experience further dilution if we issue additional equity securities in future
financing transactions.
Because the public offering price per share of our common stock
is higher than the net tangible book value per share of our common stock, you will suffer immediate and substantial dilution in
the net tangible book value of the common stock you purchase in this offering. Investors purchasing shares of common stock in this
offering will incur immediate dilution of approximately $1.075 per share. In addition, we have stock options and warrants outstanding,
as well as outstanding senior secured notes that are convertible into shares of our common stock. To the extent that such outstanding
securities are exercised or converted into shares of our common stock, investors purchasing our securities in this offering may
experience further dilution.
In
addition, we may choose to raise additional capital due to market conditions or strategic considerations even if we believe
we have sufficient funds for our current or future operating plans. To the extent that additional capital is raised through the
sale of equity or convertible debt securities, the issuance of these securities could result in further dilution to our stockholders
or result in downward pressure on the price of our common stock. See the section titled “Dilution” on page S-8
for a more detailed discussion of the dilution you will incur if you purchase common stock in this offering.
Our management will have broad discretion in the use of
the net proceeds from this offering and may invest or spend the proceeds in ways with which you do not agree and in ways that may
not yield a return.
Our
management will have broad discretion in the application of the net proceeds from this offering,
including for any of the purposes described in the section titled “Use of Proceeds” on page S-7
and you will not have the opportunity as part of your investment decision to assess whether the net proceeds are being
used appropriately. Because of the number and variability of factors that will determine our use of the net proceeds from
this offering, their ultimate use may vary from their currently intended use. The failure by our management to apply these funds
effectively could harm our business. Pending their use, we may invest the net proceeds from this offering in investment-grade,
interest-bearing securities. These investments may not yield a favorable return to our securityholders.
Our independent registered public accounting firm has
indicated that our recurring losses from operations raise substantial doubt about our ability to continue as a going concern.
As described in their audit report, our auditors have
included an explanatory paragraph that states that we have incurred recurring losses and negative cash flows from operations
and has a stockholders’ deficit at September 30, 2019 of $16.1 million, $6.7 million of senior secured notes that
may become due on December 22, 2019, $3.6 million of unsecured indebtedness due on demand and $1.0 million of unsecured
indebtedness also due on demand but subject to a forbearance agreement through March 2020, that raise substantial doubt
about the our ability to continue as a going concern. While $1.0 million of unsecured indebtedness was converted into shares
of our common stock prior to December 31, 2019, and we exchanged the $6.7 million of aggregate principal amount of the
senior secured notes due December 2019, along with accrued and unpaid interest for new notes due December 2020
having an aggregate principal amount of $7.6 million (including an exchange fee), which notes have been converting to common
stock since April 1, 2020 at the option of the holders thereof in accordance with their terms following stockholder
approval, there is nevertheless still substantial doubt about our ability to continue as a going concern.
Our consolidated financial statements do not include any adjustments
that might result from the outcome of this uncertainty. If we cannot continue as a viable entity, our securityholders may lose
some or all of their investment in our company. As of December 31, 2019, we had a stockholders’ deficit of $30.9 million,
$7.6 million of senior secured notes due December 2020, and $3.6 million of unsecured indebtedness due on demand.
BioLexis Pte. Ltd., or BioLexis, has beneficial ownership
of a significant percentage of our common stock, has the right to designate a majority of our board of directors, and is able to
exert significant control over matters subject to stockholder approval, preventing new investors from influencing significant corporate
decisions.
BioLexis beneficially owns 54,656,003 shares of our common stock
as of March 31, 2020. Accordingly, prior to this offering BioLexis beneficially owned approximately 47.5% of our common stock
and controls approximately 46.9% of our outstanding voting power. Under an investor rights agreement, as amended, with BioLexis,
BioLexis also currently has the power to designate a majority of our board of directors, and two of our seven board members are
designated by BioLexis. BioLexis’ interests may not coincide with the interests of other securityholders. BioLexis has the
ability to influence our company through both its ownership position and control of our board of directors, which may prevent or
discourage unsolicited acquisition proposals or offers for our capital stock that you may believe are in your best interest as
one of our securityholders.
A substantial number of shares of common stock may be
sold in the market following this offering, which may depress the market price for our common stock.
Sales of a substantial number of shares of our common stock
in the public market following this offering could cause the market price of our common stock to decline. A substantial majority
of the outstanding shares of our common stock are, and the shares of our common stock offered hereby will be, freely tradable without
restriction or further registration under the Securities Act.
SPECIAL NOTE REGARDING FORWARD-LOOKING
STATEMENTS
This prospectus supplement, the accompanying prospectus and
the documents incorporated by reference herein and therein, contain forward-looking statements within the meaning of Section 27A
of the Securities Act and Section 21E of the Securities Exchange Act of 1934, as amended, or the Exchange Act, about us and
our industry that involve substantial risks and uncertainties. All statements, other than statements of historical facts contained
in this prospectus supplement, the accompanying prospectus and the documents incorporated by reference herein and therein, including
statements regarding our future financial condition, business strategy and plans, and objectives of management for future operations,
are forward-looking statements. In some cases you can identify these statements by forward-looking words such as “believe,”
“may,” “will,” “estimate,” “continue,” “anticipate,” “intend,”
“could,” “would,” “project,” “plan,” “expect” or the negative or plural
of these words or similar expressions. These forward-looking statements include, but are not limited to, statements concerning
the following:
|
|
·
|
the timing and the success of the design of the clinical trials and
planned clinical trials of our lead product candidate, ONS-5010;
|
|
|
·
|
whether the results of our clinical trials will be sufficient to support
domestic or global regulatory approvals;
|
|
|
·
|
our ability to obtain and maintain regulatory approval for ONS-5010
in the United States and other markets if we successfully complete clinical trials;
|
|
|
·
|
our expectations regarding the potential market size and the size
of the patient populations for our product candidates, if approved, for commercial use;
|
|
|
·
|
our ability to fund our working capital requirements;
|
|
|
·
|
the rate and degree of market acceptance of our current and future
product candidates;
|
|
|
·
|
the implementation of our business model and strategic plans for our
business and product candidates;
|
|
|
·
|
developments or disputes concerning our intellectual property or other
proprietary rights;
|
|
|
·
|
our ability to maintain and establish collaborations or obtain additional
funding;
|
|
|
·
|
our expectations regarding government and third-party payor coverage
and reimbursement;
|
|
|
·
|
our ability to compete in the markets we serve;
|
|
|
·
|
the factors that may impact our financial results;
|
|
|
·
|
the anticipated use of proceeds from this offering; and
|
|
|
·
|
our estimates regarding the sufficiency of our cash resources and
our need for additional funding.
|
These statements reflect our current views with respect to future
events and are based on assumptions and are subject to risks and uncertainties, including risks associated with the ongoing COVID-19
global pandemic. Given these uncertainties, you should not place undue reliance on these forward-looking statements. We discuss
in greater detail many of these risks under the heading “Risk Factors” contained in our most recent annual report on
Form 10-K, which is incorporated by reference into this prospectus supplement in its entirety, as well as any amendments thereto
reflected in subsequent filings with the SEC. Also, these forward-looking statements represent our estimates and assumptions only
as of the date of the document containing the applicable statement. Unless required by law, we undertake no obligation to update
or revise any forward-looking statements to reflect new information or future events or developments. Thus, you should not assume
that our silence over time means that actual events are bearing out as expressed or implied in such forward-looking statements.
You should read this prospectus supplement and the accompanying prospectus, together with the documents we have filed with the
SEC that are incorporated by reference herein and therein and any free writing prospectus that we may authorize for use in connection
with this offering completely and with the understanding that our actual future results may be materially different from what we
expect. We qualify all of the forward-looking statements in the foregoing documents by these cautionary statements.
This prospectus supplement, the accompanying prospectus
and the documents incorporated by reference herein and therein contain market data and industry statistics and forecasts that
are based on independent industry publications and other publicly available information. Although we believe that these
sources are reliable, we do not guarantee the accuracy or completeness of this information and we have not independently
verified this information. Although we are not aware of any misstatements regarding the market and industry data presented in
this prospectus supplement, the accompanying prospectus and the documents incorporated by reference herein and therein, these
estimates involve risks and uncertainties and are subject to change based on various factors, including those discussed under
the heading “Risk Factors” contained in this prospectus supplement, the accompanying prospectus and any related
free writing prospectus, and under similar headings in the other documents that are incorporated by reference into this
prospectus supplement and the accompanying prospectus. Accordingly, investors should not place undue reliance on this
information.
USE OF PROCEEDS
We estimate that the net proceeds from this offering, after
deducting placement agent fees and estimated offering expenses payable by us, will be approximately $8.9 million.
We intend to use the net proceeds from this offering for working
capital and general corporate purposes, including in support of our ONS-5010 development program.
The amounts and timing of our use of the net proceeds from this
offering will depend on a number of factors, such as the timing and progress of our research and development efforts for ONS-5010,
the timing and progress of any partnering and commercialization efforts, technological advances and the competitive environment
for ONS-5010. Accordingly, our management will have broad discretion in the timing and application of these proceeds. Pending application
of the net proceeds as described above, we intend to temporarily invest the proceeds in short-term, interest-bearing instruments.
Dilution
If you invest in our common stock in this offering, your ownership
interest will be immediately diluted to the extent of the difference between the offering price per share of our common stock and
the pro forma as adjusted net tangible book value per share of our common stock immediately after this offering.
As of March 31, 2020, we had a historical net tangible
book deficit of $20.5 million, or $(0.23) per share of common stock. Our historical net tangible book deficit per share represents
the amount of our total tangible assets less total liabilities, divided by the total number of shares of common stock outstanding
at March 31, 2020.
As of March 31, 2020, we had a pro forma net tangible book
value of $7.3 million, or $0.06 per share of common stock. Our pro forma net tangible book value per share represents the amount
of our total tangible assets less total liabilities, divided by the total number of shares of common stock outstanding at March 31,
2020, after giving effect to (i) the issuance of 16,000,000 shares of common stock to Syntone on June 2, 2020 in a private
placement for $16,000,000 of gross cash proceeds; (ii) 8,184,718 shares of common stock issued to the holders of our convertible
senior secured notes upon conversion of an aggregate principal amount and accrued interest of $5.0 million from April 1, 2020
through June 22, 2020; and (iii) $3.3 million of proceeds from the sale of our unused New Jersey net operating losses
and research and development tax credits in May 2020.
Adjusted pro forma net tangible book value dilution per share
to new investors represents the difference between the amount per share paid by purchasers of shares of common stock in this offering
and the pro forma as adjusted net tangible book value per share of common stock immediately after completion of this offering and
the concurrent private placement to Syntone. After giving effect to the sale of 8,407,411 shares of common stock at a price per
share of $1.215 in this offering and the sale of 823,045 shares of common stock to Syntone at a price per share of $1.215 in the
concurrent private placement, and after deducting placement agent fees and estimated offering expenses payable by us, our pro forma
as adjusted net tangible book value as of March 31, 2020 would have been approximately $17.2 million, or approximately $0.14
per share. This amount represents an immediate increase in pro forma net tangible book value of $0.08 per share to our existing
stockholders and an immediate dilution in the pro forma as adjusted net tangible book value of approximately $1.075 per share to
new investors purchasing shares of common stock in this offering.
The following table illustrates this dilution on a per share
basis:
|
Offering price per share
|
|
|
|
|
|
$
|
1.215
|
|
|
Net tangible book deficit per share as of March 31, 2020
|
|
$
|
(0.23
|
)
|
|
|
|
|
|
Increase in net tangible book value per share attributable to pro forma adjustments
|
|
$
|
0.29
|
|
|
|
|
|
|
Pro forma net tangible book value per share as of March 31, 2020
|
|
$
|
0.06
|
|
|
|
|
|
|
Increase in pro forma net
tangible book value per share attributable to new investors participating in this offering
|
|
$
|
0.08
|
|
|
|
|
|
|
Pro forma as adjusted net tangible book value per share after giving effect to this offering and the concurrent private placement
|
|
|
|
|
|
$
|
0.14
|
|
|
Dilution per share to new investors participating in this offering
|
|
|
|
|
|
$
|
1.075
|
|
In addition, we may choose to raise additional capital due to
market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans.
To the extent that we raise additional capital through the sale of equity or convertible debt securities, the issuance of these
securities could result in further dilution to our stockholders.
The discussion of dilution, and the table quantifying it, assume
no exercise or settlement of any outstanding equity awards, exercise of warrants, including the placement agent warrants issued
as compensation to the placement agent for this offering, or other potentially dilutive securities. The exercise of potentially
dilutive securities having an exercise price less than the offering price would increase the dilutive effect to new investors.
The above discussion and table are based on 113,935,910 shares
of common stock outstanding as of March 31, 2020, on a pro forma basis including 16,000,000 shares of common stock issued
to Syntone on June 2, 2020 in a private placement and 8,184,718 shares of common stock issued to the holders of our convertible
senior secured notes upon conversion of an aggregate principal amount and accrued interest of $5.0 million from April 1, 2020
through June 22, 2020, and excludes:
|
|
•
|
2,470 shares of common stock issuable upon the exercise of outstanding PSUs whose terms provide for settlement in shares of
common stock or cash at our discretion, with a weighted-average exercise price of $49.97;
|
|
|
•
|
2,213,470 shares of common stock issuable upon exercise of outstanding stock options granted under the 2015 Plan, with a weighted-average
exercise price of $2.33 per share;
|
|
|
•
|
1,639,404 shares of common stock reserved for future issuance under the 2015 Plan, as well as any future automatic increases
in the number of shares of common stock reserved for future issuance under the 2015 Plan;
|
|
|
•
|
68,145 shares of common stock reserved for future issuance under the ESPP, as well as any automatic increases in the number
of shares of common stock reserved for future issuance under the ESPP;
|
|
|
•
|
shares of common stock issuable upon conversion of outstanding senior secured convertible notes having an aggregate principal
amount of $3.0 million as of the date of this prospectus supplement, which are convertible, at the option of the holder, into shares
of our common stock at a conversion price equal to 90% of the two lowest closing bid prices in the 20 trading days immediately
preceding such conversion, subject to a floor price of $0.232 per share; and
|
|
|
•
|
6,463,329 shares of common stock issuable upon exercise of outstanding warrants with a weighted-average exercise price of $2.52
per share.
|
Private
Placement Transaction
In a concurrent private placement, we are also selling to our
affiliate, Syntone, 823,045 shares of our common stock at a purchase price per share of $1.215, for an aggregate purchase price
of approximately $1.0 million.
The shares of our common stock are being offered to Syntone
pursuant to the exemption from registration provided in Section 4(a)(2) under the Securities Act and Rule 506 promulgated
thereunder and are not being offered pursuant to this prospectus supplement and the accompanying prospectus. Accordingly, Syntone
may only sell the shares of our common stock issued to it the concurrent private placement transaction pursuant to an effective
registration statement under the Securities Act covering the resale of those shares, an exemption under Rule 144 under the
Securities Act or another applicable exemption under the Securities Act.
PLAN OF DISTRIBUTION
Pursuant to an engagement letter, dated as of June 2, 2020,
we have engaged H.C. Wainwright & Co., LLC, or Wainwright or the placement agent, to act as our exclusive placement agent
in connection with this offering. Under the terms of the engagement letter, Wainwright is not purchasing the securities offered
by us in this offering and is not required to sell any specific number or dollar amount of securities, but will assist us in this
offering on a reasonable best efforts basis. The terms of this offering were subject to market conditions and negotiations between
us, Wainwright and prospective investors. Wainwright will have no authority to bind us by virtue of the engagement letter. Wainwright
may engage sub-agents or selected dealers to assist with this offering. We may not sell the entire amount of the shares of our
common stock offered pursuant to this prospectus supplement.
Wainwright proposes to arrange for the sale of the shares we
are offering pursuant to this prospectus supplement and accompanying prospectus to certain institutional and accredited investors
through a securities purchase agreement directly between each investor and us. We will only sell to such investors who have entered
into the securities purchase agreement with us.
The securities purchase agreement will be included as an exhibit
to a Current Report on Form 8-K that we will be filing with the SEC and that will be incorporated by reference into the registration
statement of which this prospectus supplement forms a part.
Delivery of the shares of our common stock offered hereby is
expected to occur on or about June 24, 2020, subject to satisfaction of certain closing conditions.
We have agreed to pay Wainwright a cash fee equal to 7.0% of
the aggregate gross proceeds raised in the offering. We have also agreed to pay Wainwright an additional 1.0% of the aggregate
gross proceeds from this offering as a management fee, $35,000 for non-accountable expenses, up to $100,000 for fees and expenses
of legal counsel and other out-of-pocket expenses and up to $12,900 for clearing expenses. We estimate the total offering expenses
of this offering that will be payable by us, excluding the placement agent’s fees and expenses, will be approximately $366,000.
In addition, Wainwright will also receive warrants to purchase that number of shares of our common stock equal to 7.0% of the aggregate
number of shares of our common stock sold in this offering, or 588,519 shares of common stock, at an exercise price of $1.51875
per share and a term of five years from the effective date of this offering. Pursuant to FINRA Rule 5110(g), the compensation
warrants and any shares issued upon exercise of the compensation warrants shall not be sold, transferred, assigned, pledged or
hypothecated, or be the subject of any hedging, short sale, derivative, put or call transaction that would result in the effective
economic disposition of the securities by any person for a period of 180 days immediately following the date of effectiveness or
commencement of sales of this offering, except the transfer of any security:
|
|
·
|
by operation of law or by reason of reorganization of our company;
|
|
|
·
|
to any FINRA member firm participating in the offering and the officers
or partners thereof, if all securities so transferred remain subject to the lock-up restriction set forth above for the remainder
of the time period;
|
|
|
·
|
if the aggregate amount of securities of our company held by the holder
of the compensation warrants or related persons do not exceed 1% of the securities being offered;
|
|
|
·
|
that is beneficially owned on a pro-rata
basis by all equity owners of an investment fund, provided that no participating member manages or otherwise directs investments
by the fund, and participating members in the aggregate do not own more than 10% of the equity in the fund; or
|
|
|
·
|
the exercise or conversion of any security,
if all securities remain subject to the lock-up restriction set forth above for the remainder of the time period.
|
We have granted Wainwright a 10-month right of first refusal
to act as our exclusive underwriter or placement agent for certain future capital raising transactions undertaken by us, provided
that this offering is consummated. Wainwright shall also be entitled to the foregoing cash and warrant compensation with respect
to certain investors contacted by Wainwright or introduced to us by Wainwright during the term of the engagement letter that invest
in any subsequent capital-raising transaction during the 10-month period following the termination or expiration of the engagement
letter.
We have agreed to indemnify Wainwright and specified other persons
against certain liabilities relating to or arising out of Wainwright’s activities under the engagement letter, including
for liabilities under the Securities Act, and to contribute to payments that Wainwright may be required to make in respect of such
liabilities.
Wainwright may be deemed to be an underwriter within the meaning
of Section 2(a)(11) of the Securities Act, and any commissions received by it and any profit realized on the resale of the
securities sold by it while acting as principal might be deemed to be underwriting discounts or commissions under the Securities
Act. As an underwriter, Wainwright would be required to comply with the requirements of the Securities Act and the Exchange Act,
including, without limitation, Rule 415(a)(4) under the Securities Act and Rule 10b-5 and Regulation M under the
Exchange Act. These rules and regulations may limit the timing of purchases and sales of shares of our common stock by Wainwright
acting as principal. Under these rules and regulations, Wainwright:
|
|
·
|
may not engage in any stabilization activity in connection with our
securities; and
|
|
|
·
|
may not bid for or purchase any of our securities or attempt to induce
any person to purchase any of our securities, other than as permitted under the Exchange Act, until it has completed its participation
in the distribution.
|
From time to time, Wainwright has and may provide in the future
various advisory, investment and commercial banking and other services to us in the ordinary course of business, for which they
have received and may continue to receive customary fees and commissions. Wainwright acted as exclusive financial advisor for our
December 2019 warrant restructuring transaction and as exclusive placement agent for our February 2020 registered direct
offering and concurrent private placement, for which it received compensation. Except as disclosed in this prospectus supplement,
we have no present arrangements with Wainwright for any further services.
The transfer agent for our common stock is American Stock Transfer &
Trust Company, LLC.
Our shares of common stock are listed on The Nasdaq Capital
Market under the symbol “OTLK.”
LEGAL MATTERS
The validity of the securities offered by this prospectus supplement
and the accompanying prospectus will be passed upon for us by Cooley LLP. Ellenoff Grossman & Schole LLP, New York, New
York is acting as counsel to the placement agent in connection with this offering.
EXPERTS
The consolidated financial statements of Outlook Therapeutics, Inc.
as of September 30, 2019 and 2018, and for the years then ended, have been incorporated by reference in this prospectus supplement
in reliance upon the report of KPMG LLP, independent registered public accounting firm, incorporated by reference herein, and upon
the authority of said firm as experts in accounting and auditing. The audit report covering the consolidated financial statements
for the year ended September 30, 2019 contains an explanatory paragraph that states that the Company has incurred recurring
losses and negative cash flows from operations and has stockholders’ deficit at September 30, 2019 of $16.1 million,
$6.7 million of senior secured notes that may become due on December 22, 2019, $3.6 million of unsecured indebtedness due
on demand and $1.0 million of unsecured indebtedness also due on demand but subject to a forbearance agreement through March 2020,
that raise substantial doubt about the Company’s ability to continue as a going concern. The consolidated financial statements
do not include any adjustments that might result from the outcome of that uncertainty.
WHERE YOU CAN FIND MORE INFORMATION
This prospectus supplement and the accompanying prospectus are
part of a registration statement on Form S-3 we filed with the SEC and do not contain all of the information set forth in
the registration statement and the exhibits to the registration statement. For further information with respect to us and the securities
we are offering under this prospectus supplement and the accompanying prospectus, we refer you to the registration statement and
the exhibits and schedules filed as a part of the registration statement. You should rely only on information contained in this
prospectus supplement, the accompanying prospectus and incorporated by reference herein and therein. We have not authorized any
person to provide you with different information. We are not making an offer of these securities in any state where the offer is
not permitted. You should not assume that the information in this prospectus supplement is accurate as of any date other than the
date on the front page of this prospectus supplement, regardless of the time of delivery of this prospectus supplement or
any sale of the securities offered by this prospectus supplement.
We file annual, quarterly and current reports, proxy statements
and other information with the SEC. Our SEC filings are available to the public at the SEC’s website at http://www.sec.gov.
INCORPORATION OF CERTAIN INFORMATION
BY REFERENCE
The SEC allows us to incorporate by reference the information
we file with it, which means that we can disclose important information to you by referring you to another document that we have
filed separately with the SEC. You should read the information incorporated by reference because it is an important part of this
prospectus supplement and the accompanying prospectus. Information in this prospectus supplement supersedes information incorporated
by reference that we filed with the SEC prior to the date of this prospectus supplement, while information that we file later with
the SEC will automatically update and supersede the information in this prospectus supplement and the accompanying prospectus.
We incorporate by reference into this prospectus supplement and the accompanying prospectus the information and documents listed
below that we have filed with the SEC (Commission File No. 001-37759):
|
|
·
|
our
Annual Report on Form 10-K for the fiscal year ended September 30, 2019, filed
with the SEC on December 19, 2019;
|
|
|
·
|
our
Quarterly Reports on Form 10-Q for the quarters ended December 31, 2019, filed
with the SEC on February 14, 2020 and March 31, 2020, filed with the SEC on
May 15, 2020;
|
|
|
·
|
our
Current Reports on Form 8-K, filed with the SEC on December 6, 2019, December 19, 2019, December 23, 2019, January 23, 2020, January 31, 2020, February 24, 2020, March 24, 2020, April 2, 2020, April 17, 2020, April 21, 2020,
May 11, 2020, May 28, 2020 and June 17, 2020, to the extent the information
in such reports is filed and not furnished;
|
|
|
·
|
the
description of our common stock set forth in our registration statement on Form 8-A,
filed with the SEC on April 29, 2016, as amended on May 11, 2016, including
any further amendments thereto or reports filed for the purposes of updating this description.
|
We also incorporate by reference any future filings (other
than Current Reports furnished under Item 2.02 or Item 7.01 of Form 8-K and exhibits filed on such form that
are related to such items unless such Form 8-K expressly provides to the contrary) made with the SEC pursuant to
Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act, including those made after the date of this prospectus
supplement, until the termination of the offering of the common stock made by this prospectus supplement and the accompanying
prospectus. Information in such future filings updates and supplements the information provided in this prospectus supplement
and the accompanying prospectus. Any statements in any such future filings will automatically be deemed to modify and
supersede any information in any document we previously filed with the SEC that is incorporated or deemed to be incorporated
herein by reference to the extent that statements in the later filed document modify or replace such earlier statements.
We will furnish without charge to you, upon written or oral
request, a copy of any or all of the documents incorporated by reference into this prospectus supplement and the accompanying prospectus,
including exhibits that are specifically incorporated by reference into such documents. You should direct any requests for documents
to Outlook Therapeutics, Inc., Attention: Corporate Secretary, 4260 U.S. Route 1, Monmouth Junction, New Jersey 08852. Our
phone number is (609) 619-3990. You may also view the documents that we file with the SEC and incorporate by reference in this
prospectus supplement and the accompanying prospectus on our corporate website at www.outlooktherapeutics.com. The information
on our website is not incorporated by reference and is not a part of this prospectus supplement.
PROSPECTUS
$100,000,000

Common
Stock
Preferred
Stock
Debt Securities
Warrants
From time to time, we may offer and sell up to $100,000,000
of any combination of the securities described in this prospectus, either individually or in combination. We may also offer common
stock or preferred stock upon conversion of debt securities, common stock upon conversion of preferred stock, or common stock,
preferred stock or debt securities upon the exercise of warrants.
This prospectus provides a general description of the securities
we may offer. We will provide the specific terms of these offerings and securities in one or more supplements to this prospectus.
We may also authorize one or more free writing prospectuses to be provided to you in connection with these offerings. The prospectus
supplement and any related free writing prospectus may also add, update or change information contained in this prospectus. You
should carefully read this prospectus, the applicable prospectus supplement and any related free writing prospectus, as well as
any documents incorporated by reference, before buying any of the securities being offered.
This prospectus may not be used to consummate a sale of securities
unless accompanied by a prospectus supplement.
We may sell these securities directly to investors, through
agents designated from time to time or to or through underwriters or dealers, on a continuous or delayed basis. For additional
information on the methods of sale, you should refer to the section titled “Plan of Distribution” in this prospectus
and in the applicable prospectus supplement. If any agents or underwriters are involved in the sale of any securities with respect
to which this prospectus is being delivered, the names of such agents or underwriters and any applicable fees, discounts or commissions
and over-allotment options will be set forth in a prospectus supplement. The price to the public of such securities and the net
proceeds we expect to receive from such sale will also be set forth in a prospectus supplement.
Our common stock is listed on The Nasdaq Capital Market under
the trading symbol “OTLK.” On May 31, 2019, the last reported sale price of our common stock on The Nasdaq Capital
Market was $2.40 per share. The applicable prospectus supplement will contain information, where applicable, as to other listings,
if any, on The Nasdaq Capital Market or other securities exchange of the securities covered by the applicable prospectus supplement.
Investing
in our securities involves a high degree of risk. You should review carefully the risks and uncertainties described under the
heading “Risk Factors” on page 4 of this prospectus and any similar section contained in the applicable prospectus
supplement and in any related free writing prospectuses we have authorized for use in connection with a specific offering, and
under similar headings in the documents that are incorporated by reference into this prospectus.
Neither the Securities and Exchange Commission nor any state
securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete.
Any representation to the contrary is a criminal offense.
The date of this
prospectus is June 26, 2019.
TABLE OF CONTENTS
ABOUT THIS PROSPECTUS
This prospectus is part of a registration statement on Form
S-3 that we filed with the Securities and Exchange Commission, or SEC, using a “shelf” registration process. Under
this shelf registration statement, we may sell from time to time in one or more offerings up to a total dollar amount of $100,000,000
of common stock and preferred stock, various series of debt securities and/or warrants to purchase any of such securities, either
individually or in combination with other securities as described in this prospectus.
This prospectus provides you with a general description of the
securities we may offer. Each time we sell any type or series of securities under this prospectus, we will provide a prospectus
supplement that will contain more specific information about the terms of that offering. We may also authorize one or more free
writing prospectuses to be provided to you that may contain material information relating to these offerings. We may also add,
update or change in a prospectus supplement or free writing prospectus any of the information contained in this prospectus or in
the documents we have incorporated by reference into this prospectus. This prospectus, together with the applicable prospectus
supplement, any related free writing prospectus and the documents incorporated by reference into this prospectus and the applicable
prospectus supplement, will include all material information relating to the applicable offering. You should carefully read both
this prospectus and the applicable prospectus supplement, any related free writing prospectus, together with the information and
documents incorporated by reference herein as described under the heading “Incorporation of Certain Information by Reference.”
This prospectus may not be used to consummate a sale of securities
unless accompanied by a prospectus supplement.
You should rely only on the information contained in, or incorporated
by reference into, this prospectus and any applicable prospectus supplement, along with the information contained in any free writing
prospectuses we have authorized for use in connection with a specific offering. We have not authorized anyone to provide you with
different or additional information. You must not rely upon any information or representation not contained or incorporated by
reference in this prospectus, any applicable prospectus supplement or any related free writing prospectus. This prospectus, any
applicable supplement to this prospectus or any related free writing prospectus do not constitute an offer to sell or the solicitation
of an offer to buy any securities other than the registered securities to which they relate, nor do this prospectus, any applicable
supplement to this prospectus or any related free writing prospectus constitute an offer to sell or the solicitation of an offer
to buy securities in any jurisdiction to any person to whom it is unlawful to make such offer or solicitation in such jurisdiction.
The information appearing in this prospectus, any applicable
prospectus supplement or any related free writing prospectus is accurate only as of the date on the front of the document and any
information we have incorporated by reference is accurate only as of the date of the document incorporated by reference, regardless
of the time of delivery of this prospectus, any applicable prospectus supplement or any related free writing prospectus, or any
sale of a security. Our business, financial condition, results of operations and prospects may have changed since those dates.
This prospectus contains and incorporates by reference market
data and industry statistics and forecasts that are based on independent industry publications and other publicly available information.
Although we believe that these sources are reliable, we do not guarantee the accuracy or completeness of this information and we
have not independently verified this information. Although we are not aware of any misstatements regarding the market and industry
data presented in this prospectus and the documents incorporated herein by reference, these estimates involve risks and uncertainties
and are subject to change based on various factors, including those discussed under the heading “Risk Factors” contained
in the applicable prospectus supplement and any related free writing prospectus, and under similar headings in the other documents
that are incorporated by reference into this prospectus. Accordingly, investors should not place undue reliance on this information.
This prospectus contains summaries of certain provisions contained
in some of the documents described herein, but reference is made to the actual documents for complete information. All of the summaries
are qualified in their entirety by the actual documents. Copies of some of the documents referred to herein have been filed, will
be filed or will be incorporated by reference as exhibits to the registration statement of which this prospectus is a part, and
you may obtain copies of those documents as described below under the heading “Where You Can Find More Information.”
Except as otherwise indicated or unless the context otherwise
requires, references to “company,” “we,” “us,” “our” or “Outlook Therapeutics,”
refer to Outlook Therapeutics, Inc. and its consolidated subsidiaries.
Our name “Outlook Therapeutics,” the Outlook Therapeutics
logo and other trademarks or service marks of Outlook Therapeutics, Inc. appearing in this prospectus and in any prospectus supplement
or free writing prospectus and the information incorporated by reference herein or therein are the property of Outlook Therapeutics,
Inc. Other trademarks, service marks or trade names appearing in this prospectus in any prospectus supplement or free writing prospectus
and the information incorporated by reference herein or therein are the property of their respective owners. We do not intend our
use or display of other companies’ trade names, trademarks or service marks to imply a relationship with, or endorsement
or sponsorship of us by, these other companies.
PROSPECTUS SUMMARY
This summary highlights selected information contained
elsewhere in this prospectus or incorporated by reference herein and does not contain all of the information that you need to consider
in making your investment decision. You should carefully read the entire prospectus, the applicable prospectus supplement and any
related free writing prospectus, including the risks of investing in our securities discussed under the heading “Risk Factors”
contained in the applicable prospectus supplement and any related free writing prospectus, and under similar headings in the other
documents that are incorporated by reference into this prospectus. You should also carefully read the information incorporated
by reference into this prospectus, including our financial statements and related notes, and the exhibits to the registration statement
of which this prospectus is a part, before making your investment decision.
Overview
We are a late clinical-stage biopharmaceutical
company focused on developing and commercializing ONS-5010, a complex, technically challenging and commercially attractive monoclonal
antibody, or mAb, for various ophthalmic indications. Our goal is to launch ONS-5010 as the first, and only, approved bevacizumab
in the United States, Europe, Japan and other markets for the treatment of wet age related macular degeneration, or wet AMD, diabetic
macular edema, or DME, and branch retinal vein occlusion, or BRVO.
ONS-5010 is an innovative mAb therapeutic
product candidate that was reviewed at a successful end of Phase 2 meeting with the FDA conducted in 2018. Our investigational
new drug, or IND, application was filed with, and accepted by, the U.S. Food and Drug Administration, or FDA, in the first quarter
of calendar 2019. We are currently enrolling patients in a Phase 3 clinical trial in Australia (ONS-5010-001) designed to serve
as the first of two adequate and well controlled studies evaluating ONS-5010 against ranibizumab (Lucentis) for wet AMD. Enrollment
in ONS-5010-001 is nearly complete with 51 of the planned 60 patients enrolled as of May 15, 2019. The second of the two Phase
3 studies (ONS-5010-002) has been initiated and is expected to begin enrolling wet AMD patients in the United States, Australia
and New Zealand by the end of June 2019. The ONS-5010-002 study is now expected to enroll a total of at least 220 patients. The
endpoint for both studies is a mean increase in baseline visual acuity of at least five letters at 11 months for ONS-5010 dosed
on a monthly basis compared to Lucentis dosed using the approved alternative dosing regimen of three monthly doses followed by
quarterly dosing.
Currently, the cancer drug Avastin (bevacizumab)
is used off-label for the treatment of wet AMD and other retina diseases such as DME and BRVO even though Avastin has not been
approved by regulatory authorities for use in these diseases. If the ONS-5010 clinical program is successful, it will support our
plans to submit for regulatory approval in multiple markets in 2020 including the United States, Europe and Japan. Because there
are no approved bevacizumab products for the treatment of retinal diseases in such major markets, we are developing ONS-5010 as
an innovative therapy and not using the biosimilar drug development pathway that would be normally be required if Avastin were
an approved drug for the targeted diseases. If approved, we believe ONS-5010 has potential to mitigate risks associated with off-label
use of Avastin or other drugs. Off label use of Avastin is currently estimated to account for at least 50% of all wet AMD prescriptions
in the United States.
Risks Associated with our Business
Our
business is subject to numerous risks, as described under the heading “Risk Factors” contained in the applicable prospectus
supplement and in any free writing prospectuses we have authorized for use in connection with a specific offering, and under similar
headings in the documents that are incorporated by reference into this prospectus.
Additional Information
For additional information related to our business and operations,
please refer to the reports incorporated herein by reference, including our Annual Report on Form 10-K for the year ended September 30,
2018 as filed with the SEC on December 18, 2018, our Quarterly Report on Form 10-Q for the quarter ended March 31, 2019
as filed with the SEC on May 15, 2019 and our other Quarterly Reports and our Current Reports on Form 8-K as filed with the
SEC, as described in the section titled “Incorporation of Certain Information by Reference.”
Implications of Being an Emerging Growth
Company
We are an “emerging
growth company,” as defined in the Jumpstart Our Business Startups Act, or the JOBS Act, enacted in April 2012, and
we may remain an emerging company for up to five years from the closing of our IPO in May 2016. For so long as we remain
an emerging growth company, we are permitted and intend to rely on certain exemptions from various public company reporting requirements,
including not being required to have our internal control over financial reporting audited by our independent registered public
accounting firm pursuant to Section 404(b) of the Sarbanes-Oxley Act of 2002, reduced disclosure obligations regarding executive
compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory
vote on executive compensation and any golden parachute payments not previously approved.
Company Information
We initially incorporated in January 2010
in New Jersey as Oncobiologics, Inc., and in October 2015, we reincorporated in Delaware by merging with and into a Delaware corporation.
In November 2018, we changed our name to Outlook Therapeutics, Inc. Our headquarters are located at 7 Clarke Drive, Cranbury, New
Jersey, 08512, and our telephone number at that location is (609) 619-3990. Our website address is www.outlooktherapeutics.com.
The information contained on, or that can be accessed through, our website is not part of, and is not incorporated by reference
into this prospectus.
THE SECURITIES WE MAY OFFER
We may offer shares of our common stock and preferred stock,
various series of debt securities and/or warrants to purchase any of such securities, either individually or in combination, up
to a total dollar amount of $100,000,000 from time to time under this prospectus, together with the applicable prospectus supplement
and any related free writing prospectus, at prices and on terms to be determined by market conditions at the time of any offering.
We may also offer common stock, preferred stock and/or debt securities upon the exercise of warrants. This prospectus provides
you with a general description of the securities we may offer. Each time we offer a type or series of securities under this prospectus,
we will provide a prospectus supplement that will describe the specific amounts, prices and other important terms of the securities,
including, to the extent applicable:
|
|
·
|
designation or classification;
|
|
|
·
|
aggregate principal amount or aggregate offering price;
|
|
|
·
|
original issue discount;
|
|
|
·
|
rates and times of payment of interest or dividends;
|
|
|
·
|
redemption, conversion, exercise, exchange or sinking fund terms;
|
|
|
·
|
voting or other rights;
|
|
|
·
|
conversion or exchange prices or rates and, if applicable, any provisions for changes to or adjustments in the conversion or
exchange prices or rates and in the securities or other property receivable upon conversion or exchange;
|
|
|
·
|
voting or other rights; and
|
|
|
·
|
a discussion of material or special U.S. federal income tax considerations.
|
The applicable prospectus supplement and any related free writing
prospectus that we may authorize to be provided to you may also add, update or change any of the information contained in this
prospectus or in the documents we have incorporated by reference. However, no prospectus supplement or free writing prospectus
will offer a security that is not registered and described in this prospectus at the time of the effectiveness of the registration
statement of which this prospectus is a part.
This prospectus may not be used to consummate a sale of
securities unless it is accompanied by a prospectus supplement.
We may sell the securities directly to investors or to or through
agents, underwriters, or dealers. We, and our agents, underwriters or dealers reserve the right to accept or reject all or part
of any proposed purchase of securities. If we do offer securities to or through agents, underwriters or dealers, we will include
in the applicable prospectus supplement:
|
|
·
|
the names of those agents, underwriters or dealers;
|
|
|
·
|
applicable fees, discounts and commissions to be paid to them;
|
|
|
·
|
details regarding over-allotment or other options, if any; and
|
|
|
·
|
the net proceeds to us.
|
Common Stock
We may issue shares of our common stock from time to time. Each
holder of our common stock is entitled to one vote for each share on all matters submitted to a vote of the stockholders, including
the election of directors. Subject to preferences that may be applicable to any then-outstanding preferred stock, holders of common
stock are entitled to receive ratably those dividends, if any, as may be declared from time to time by our board of directors out
of legally available funds. In the event of our liquidation, dissolution or winding up, holders of common stock are entitled to
share ratably in the net assets legally available for distribution to stockholders after the payment of all of our debts and other
liabilities and the satisfaction of any liquidation preference granted to the holders of any then-outstanding shares of preferred
stock. Holders of common stock have no preemptive, conversion or subscription rights and there are no redemption or sinking fund
provisions applicable to the common stock. The rights, preferences and privileges of the holders of common stock are subject to,
and may be adversely affected by, the rights of the holders of shares of any series of preferred stock that we may designate in
the future. In this prospectus, we have summarized certain general features of the common stock under “Description of Capital
Stock — Common Stock.” We urge you, however, to read the applicable prospectus supplement (and any related free writing
prospectus that we may authorize to be provided to you) related to any common stock being offered.
Preferred Stock
We may issue shares of our preferred stock from time to time,
in one or more series. Our board of directors will determine the designations, voting powers, preferences and rights of the preferred
stock, as well as the qualifications, limitations or restrictions thereof, including dividend rights, conversion rights, preemptive
rights, terms of redemption or repurchase, liquidation preferences, sinking fund terms and the number of shares constituting any
series or the designation of any series. Convertible preferred stock will be convertible into our common stock or exchangeable
for other securities. Conversion may be mandatory or at your option and would be at prescribed conversion rates.
If we sell any series of preferred stock under this prospectus,
we will fix the designations, voting powers, preferences and rights of such series of preferred stock, as well as the qualifications,
limitations or restrictions thereof, in the certificate of designation relating to that series. We will file as an exhibit to the
registration statement of which this prospectus is a part, or will incorporate by reference from reports that we file with the
SEC, the form of any certificate of designation that contains the terms of the series of preferred stock we are offering. In this
prospectus, we have summarized certain general features of the preferred stock under “Description of Capital Stock –
Preferred Stock.” We urge you, however, to read the applicable prospectus supplement (and any related free writing prospectus
that we may authorize to be provided to you) related to the series of preferred stock being offered, as well as the complete certificate
of designation that contains the terms of the applicable series of preferred stock.
Debt Securities
We may issue debt securities from time to time, in one or more
series, as either senior or subordinated debt or as senior or subordinated convertible debt. The senior debt securities will rank
equally with any other unsecured and unsubordinated debt. The subordinated debt securities will be subordinate and junior in right
of payment, to the extent and in the manner described in the instrument governing the debt, to all of our senior indebtedness.
Convertible debt securities will be convertible into or exchangeable for our common stock or other securities. Conversion may be
mandatory or at your option and would be at prescribed conversion rates.
Any debt securities issued under this prospectus will be issued
under one or more documents called indentures, which are contracts between us and a national banking association or other eligible
party, as trustee. In this prospectus, we have summarized certain general features of the debt securities under “Description
of Debt Securities.” We urge you, however, to read the applicable prospectus supplement (and any free writing prospectus
that we may authorize to be provided to you) related to the series of debt securities being offered, as well as the complete indentures
that contain the terms of the debt securities. We have filed the form of indenture as an exhibit to the registration statement
of which this prospectus is a part, and supplemental indentures and forms of debt securities containing the terms of the debt securities
being offered will be filed as exhibits to the registration statement of which this prospectus is a part or will be incorporated
by reference from reports that we file with the SEC.
Warrants
We may issue warrants for the purchase of common stock, preferred
stock and/or debt securities in one or more series. We may issue warrants independently or in combination with common stock, preferred
stock and/or debt securities. In this prospectus, we have summarized certain general features of the warrants under “Description
of Warrants.” We urge you, however, to read the applicable prospectus supplement (and any free writing prospectus that we
may authorize to be provided to you) related to the particular series of warrants being offered, as well as the complete warrant
agreements and warrant certificates that contain the terms of the warrants. We have filed forms of the warrant agreements and forms
of warrant certificates containing the terms of the warrants that may be offered as exhibits to the registration statement of which
this prospectus is a part. We will file as exhibits to the registration statement of which this prospectus is a part, or will incorporate
by reference from reports that we file with the SEC, the form of warrant and/or the warrant agreement and warrant certificate,
as applicable, that contain the terms of the particular series of warrants we are offering, and any supplemental agreements, before
the issuance of such warrants.
Any warrants issued under this prospectus may be evidenced by
warrant certificates. Warrants may be issued under an applicable warrant agreement that we enter into with a warrant agent. We
will indicate the name and address of the warrant agent, if applicable, in the prospectus supplement relating to the particular
series of warrants being offered.
RISK FACTORS
Investing in our securities involves a
high degree of risk. Before deciding whether to invest in our securities, you should consider carefully the risks and uncertainties
described under the heading “Risk Factors” contained in the applicable prospectus supplement and any related free writing
prospectus, and discussed under the heading “Risk Factors” contained in our most recent Annual Report on Form 10-K,
as well as any amendments thereto reflected in subsequent filings with the SEC, which are incorporated by reference into this prospectus
in their entirety, together with other information in this prospectus, the documents incorporated by reference and any free writing
prospectus that we may authorize for use in connection with this offering. The risks described in these documents are not the only
ones we face, but those that we consider to be material. There may be other unknown or unpredictable economic, business, competitive,
regulatory or other factors that could have material adverse effects on our future results. Past financial performance may not
be a reliable indicator of future performance, and historical trends should not be used to anticipate results or trends in future
periods. If any of these risks actually occurs, our business, financial condition, results of operations or cash flow could be
seriously harmed. This could cause the trading price of our common stock to decline, resulting in a loss of all or part of your
investment. Please also read carefully the section below titled “Special Note Regarding Forward-Looking Statements.”
SPECIAL NOTE REGARDING FORWARD-LOOKING
STATEMENTS
This prospectus, including the documents that
we incorporate by reference herein, contains, and any applicable prospectus supplement or free writing prospectus including the
documents we incorporate by reference therein may contains forward-looking statements within the meaning of Section 27A of the
Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended,
or the Exchange Act, about us and our industry that involve substantial risks and uncertainties. All statements, other than statements
of historical facts contained in this prospectus, including statements regarding our future financial condition, business strategy
and plans, and objectives of management for future operations, are forward-looking statements. In some cases you can identify these
statements by forward-looking words such as “believe,” “may,” “will,” “estimate,”
“continue,” “anticipate,” “intend,” “could,” “would,” “project,”
“plan,” “expect” or the negative or plural of these words or similar expressions. These forward-looking
statements include, but are not limited to, statements concerning the following:
|
|
·
|
the timing and the success of the design of the clinical trials and planned clinical trials of our lead product candidate,
ONS-5010;
|
|
|
·
|
whether the results of our clinical trials will be sufficient to support domestic or global regulatory approvals;
|
|
|
·
|
our ability to obtain and maintain regulatory approval for ONS-5010 in the United States and other markets if we successfully
complete clinical trials;
|
|
|
·
|
our expectations regarding the potential market size and the size of the patient populations for our product candidates, if
approved, for commercial use;
|
|
|
·
|
our ability to fund our working capital requirements;
|
|
|
·
|
the rate and degree of market acceptance of our current and future product candidates;
|
|
|
·
|
the implementation of our business model and strategic plans for our business and product candidates;
|
|
|
·
|
developments or disputes concerning our intellectual property or other proprietary rights;
|
|
|
·
|
our ability to maintain and establish collaborations or obtain additional funding;
|
|
|
·
|
our expectations regarding government and third-party payor coverage and reimbursement;
|
|
|
·
|
our ability to compete in the markets we serve; and
|
|
|
·
|
the factors that may impact our financial results.
|
These statements reflect our current views
with respect to future events and are based on assumptions and are subject to risks and uncertainties. Given these uncertainties,
you should not place undue reliance on these forward-looking statements. We discuss in greater detail many of these risks under
the heading “Risk Factors” contained in the applicable prospectus supplement, in any free writing prospectuses we may
authorize for use in connection with a specific offering, and in our most recent annual report on Form 10-K and in our most recent
quarterly report on Form 10-Q, which are incorporated by reference into this prospectus in their entirety, as well as any amendments
thereto reflected in subsequent filings with the SEC. Also, these forward-looking statements represent our estimates and assumptions
only as of the date of the document containing the applicable statement. Unless required by law, we undertake no obligation to
update or revise any forward-looking statements to reflect new information or future events or developments. Thus, you should not
assume that our silence over time means that actual events are bearing out as expressed or implied in such forward-looking statements.
You should read this prospectus, any applicable prospectus supplement, together with the documents we have filed with the SEC that
are incorporated by reference and any free writing prospectus that we may authorize for use in connection with this offering completely
and with the understanding that our actual future results may be materially different from what we expect. We qualify all of the
forward-looking statements in the foregoing documents by these cautionary statements.
USE OF PROCEEDS
Except as described in any applicable prospectus supplement
or in any free writing prospectuses we have authorized for use in connection with a specific offering, we currently intend to use
the net proceeds from the sale of the securities offered by us hereunder, if any, for working capital, capital expenditures and
general corporate purposes, which may include, among other things, funding research and development, clinical trials, vendor payable,
potential regulatory submissions, hiring additional personnel and capital expenditures. We may also use a portion of the net proceeds
to in-license, acquire, or invest in additional businesses, technologies, products, or assets, though we currently have no specific
agreements, commitments, or understandings with respect to any in-licensing or acquisitions.
The amounts and timing of our use of the net proceeds from this
offering will depend on a number of factors, such as the timing and progress of our research and development efforts, the timing
and progress of any partnering and commercialization efforts, technological advances and the competitive environment for our products.
As of the date of this prospectus, we cannot specify with certainty all of the particular uses for the net proceeds to us from
the sale of the securities offered by us hereunder. Accordingly, our management will have broad discretion in the timing and application
of these proceeds. Pending application of the net proceeds as described above, we intend to temporarily invest the proceeds in
short-term, interest-bearing instruments.
DESCRIPTION OF CAPITAL STOCK
As
of the date of this prospectus, our authorized capital stock consists of 200,000,000 shares of common stock, par value $0.01 per
share, and 10,000,000 shares of preferred stock, par value $0.01 per share.
The
following summary description of our capital stock is based on the provisions of our amended and restated certificate of incorporation,
our amended and restated bylaws and the applicable provisions of the Delaware General Corporation Law. This information may not
be complete in all respects and is qualified entirely by reference to the provisions of our amended and restated certificate of
incorporation, our amended and restated bylaws and the Delaware General Corporation Law. For information on how to obtain copies
of our amended and restated certificate of incorporation and our amended and restated bylaws, see “Where You Can Find More
Information.”
Common Stock
As
of May 31, 2019, we had 26,050,811 shares of common stock outstanding.
Voting Rights
Each
holder of common stock is entitled to one vote for each share on all matters submitted to a vote of the stockholders. The affirmative
vote of holders of 66⅔% of the voting power of all of the then-outstanding shares of capital stock, voting as a single class,
will be required to amend certain provisions of our amended and restated certificate of incorporation, including provisions relating
to amending our amended and restated bylaws, the classified board, the size of our board, removal of directors, director liability,
vacancies on our board, special meetings, stockholder notices, actions by written consent and exclusive jurisdiction.
Dividends
Subject
to preferences that may apply to any outstanding preferred stock, holders of our common stock are entitled to receive ratably any
dividends that our board of directors may declare out of funds legally available for that purpose on a non-cumulative basis. Our
outstanding senior secured notes first issued beginning December 2016 restrict our ability to pay dividends.
Liquidation
In
the event of our liquidation, dissolution or winding up, holders of our common stock are entitled to share ratably in all assets
remaining after payment of liabilities and the liquidation preference of any outstanding preferred stock.
Rights and Preferences
Holders
of our common stock have no preemptive, conversion, subscription or other rights, and there are no redemption or sinking fund provisions
applicable to our common stock. The rights, preferences and privileges of the holders of our common stock are subject to and may
be adversely affected by the rights of the holders of shares of any series of our preferred stock that we may designate in the
future.
Preferred Stock
Our
board of directors has the authority, without further action by our stockholders, to issue up to 10,000,000 shares of preferred
stock in one or more series and to fix the number, rights, preferences, privileges and restrictions thereof. These rights, preferences
and privileges could include dividend rights, conversion rights, voting rights, terms of redemption, liquidation preferences and
sinking fund terms, and the number of shares constituting any series or the designation of such series, any or all of which may
be greater than the rights of common stock. The issuance of our preferred stock could adversely affect the voting power of holders
of common stock and the likelihood that such holders will receive dividend payments and payments upon liquidation. In addition,
the issuance of preferred stock could have the effect of delaying, deferring or preventing a change in control or other corporate
action. In June 2018, our Board designated 200,000 shares as “Series A-1 Convertible preferred stock.” As
of March 31, 2019, there were 63,250 shares of our Series A-1 Convertible preferred stock issued and outstanding.
Series A-1
Convertible Preferred Stock
As
of March 31, 2019, 200,000 shares of Series A-1 Convertible preferred stock have been authorized for issuance, 63,250 of which
were issued and outstanding at such date. The shares of Series A-1 Convertible preferred stock have a stated value of
$100.00 per share and the 63,250 outstanding shares are convertible into approximately 1,195,295 shares of our common stock as
of March 31, 2019.
Dividends
The
Series A-1 Convertible preferred stock accrue dividends at a rate of 10% per annum, compounded quarterly, payable quarterly
at our option in cash or in kind in additional shares of Series A-1 Convertible preferred stock. The Series A-1 Convertible
preferred stock are also entitled to dividends on an as-if-converted basis in the same form as any dividends actually paid on shares
of our common stock or other securities. The initial conversion rate is subject to appropriate adjustment in the event of a stock
split, stock dividend, combination, reclassification or other recapitalization affecting our common stock.
Voting
The
holders of the Series A-1 Convertible preferred stock have the right to vote on matters submitted to a vote of our stockholders
on an as-converted basis. In addition, without the prior written consent of a majority of the outstanding shares of Series A-1
Convertible preferred stock, we may not take certain actions.
The
terms of the Series A-1 Convertible preferred stock distinguish between certain liquidation events (such as a voluntary or
involuntary liquidation, dissolution or winding up of our company) and “deemed” liquidation events (such as a sale
of all or substantially all of our assets, various merger and reorganization transactions, being delisted from the Nasdaq and the
occurrence of an event of default under the terms of the senior secured notes), in each case as defined in the Certificate of Designation.
In the event of a liquidation (as defined in the Certificate of Designation) the liquidation preference payable equals the sum
of (A) 550% of the stated value per share plus (B) (x) 550% of any accrued but unpaid preferred dividends
(as defined in the Certificate of Designation) plus (y) any unpaid participating dividends (as defined in the Certificate
of Designation). In the case of a deemed liquidation event (as defined in the Certificate of Designation), the multiplier is increased
to 600%.
Conversion
The
Series A-1 Convertible preferred stock is convertible at any time at the option of the holder based on the then applicable
conversion rate. If conversion is in connection with a liquidation (as defined in the Certificate of Designation), the holder is
entitled to receive 550% of the number of shares of common stock issuable based upon the then applicable conversion rate. In the
event of a deemed liquidation event (as defined in the Certificate of Designation), the multiplier is increased to 600%.
Redemption
Additionally,
the holder may require the Company to redeem the Series A-1 Convertible preferred stock in the event of deemed liquidation
event for the sum of (A) 600% of the stated value per share plus (B) an amount equal to (x) 600% of any accrued,
but unpaid, preferred dividends plus (y) any unpaid participating dividends, although such redemption may not be made without
the consent of the senior secured noteholders if such notes are outstanding at the time of any such redemption.
Common Stock Equivalents
As
of March 31, 2019, we had issued and outstanding 16,131 performance stock unit awards, 7,156 restricted stock unit awards and
541,746 stock option awards under our equity incentive plans. At such date, we also had outstanding warrants to acquire an
aggregate of 5,660,949 shares of our common stock. Subsequent to March 31, 2019 we issued warrants to acquire an aggregate of
20,680,000 shares of our common stock. In addition, as March 31, 2019, we had 63,250 shares of our Series A-1
Convertible preferred stock issued and outstanding. We also had outstanding at March 31, 2019 an aggregate $8.5 million
principal amount of senior secured notes, which are convertible (along with accrued but unpaid interest thereon) into shares
of our common stock at an initial conversion price of $8.9539 per share and an aggregate $1.0 million of
unsecured notes, which are exchangeable (along with accrued and unpaid interest thereon) into shares of our common stock at
an initial exchange rate of $13.44 per share. As of March 31, 2019, the outstanding senior secured notes were
convertible into an aggregate of 957,482 shares of our common stock, and the outstanding unsecured notes were convertible into an aggregate of 147,347 shares of our common
stock.
Stockholder Registration
Rights
Certain
holders of our securities, including certain holders of 5% of our capital stock, certain of our directors, and the holder of the
Series A-1 Convertible preferred stock are entitled to certain rights with respect to registration of such securities under
the Securities Act. These securities are referred to as registrable securities. The holders of these registrable securities possess
registration rights pursuant to the terms of registration rights agreements.
In
general, the registration of shares of our common stock pursuant to the exercise of registration rights enables the holders to
trade such shares without restriction under the Securities Act when the applicable registration statement is declared effective.
We generally have agreed to pay the registration expenses for such registration statements, other than underwriting discounts,
selling commissions and stock transfer taxes, of the shares registered.
Generally,
in an underwritten offering, the managing underwriter, if any, has the right, subject to specified conditions, to limit the number
of shares the holders may include. We must use commercially reasonable efforts to keep the registration statement effective until
the earlier of the date on which all registrable securities covered by such registration statement have been sold, or at such time
that the holders of the registrable securities can sell their shares under Rule 144 of the Securities Act during any three-month
period.
Anti-Takeover Provisions
of Delaware Law and Our Charter Documents
Section 203
of the Delaware General Corporation Law
We
are subject to Section 203 of the Delaware General Corporation Law, which prohibits a Delaware corporation from engaging in
any business combination with any interested stockholder for a period of three years after the date that such stockholder
became an interested stockholder, with the following exceptions:
|
|
·
|
before such date, the board of directors of the corporation approved
either the business combination or the transaction that resulted in the stockholder becoming an interested stockholder;
|
|
|
·
|
upon completion of the transaction that resulted in the stockholder
becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding
at the time the transaction began, excluding for purposes of determining the voting stock outstanding (but not the outstanding
voting stock owned by the interested stockholder) those shares owned (1) by persons who are directors and also officers
and (2) employee stock plans in which employee participants do
not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange
offer; and
|
|
|
·
|
on or after such date, the business combination is approved by the
board of directors and authorized at an annual or special meeting of the stockholders, and not by written consent, by the affirmative
vote of at least 66⅔% of the outstanding voting stock that is not owned by the interested stockholder.
|
In
general, Section 203 defines a “business combination” to include the following:
|
|
·
|
any merger or consolidation involving the corporation and the interested
stockholder;
|
|
|
·
|
any sale, transfer, pledge or other disposition of 10% or more of
the assets of the corporation involving the interested stockholder;
|
|
|
·
|
subject to certain exceptions, any transaction that results in the
issuance or transfer by the corporation of any stock of the corporation to the interested stockholder;
|
|
|
·
|
any transaction involving the corporation that has the effect of
increasing the proportionate share of the stock or any class or series of the corporation beneficially owned by the interested
stockholder;
|
|
|
·
|
the receipt by the interested stockholder of the benefit of any
loans, advances, guarantees, pledges or other financial benefits by or through the corporation; and
|
|
|
·
|
in general, Section 203 defines an “interested stockholder”
as an entity or person who, together with the person’s affiliates and associates, beneficially owns, or within three years
prior to the time of determination of interested stockholder status did own, 15% or more of the outstanding voting stock of the
corporation.
|
The statute could prohibit or delay mergers or other takeover or change in control attempts and, accordingly,
may discourage attempts to acquire us even though such a transaction may offer our stockholders the opportunity to sell their stock
at a price above the prevailing market price.
Amended and Restated
Certificate of Incorporation and Amended and Restated Bylaws, as Amended
Among
other things, our amended and restated certificate of incorporation and amended and restated bylaws, as amended:
|
|
·
|
permit our board of directors to issue up to 10,000,000 shares of
preferred stock, with any rights, preferences and privileges as they may designate, including the right to approve an acquisition
or other change in control;
|
|
|
·
|
provide that the authorized number of directors may be changed only
by resolution of our board of directors;
|
|
|
·
|
provide that our board of directors is classified into three classes
of directors;
|
|
|
·
|
provide that, subject to the rights of any series of preferred stock
to elect directors, directors may only be removed for cause, which removal may be effected, subject to any limitation imposed by
law, by the holders of at least a majority
of the voting power of all of our then-outstanding shares of the capital
stock entitled to vote generally at an election of directors;
|
|
|
·
|
provide that all vacancies, including newly created directorships,
may, except as otherwise required by law, be filled by the affirmative vote of a majority of directors then in office, even if
less than a quorum;
|
|
|
·
|
require that any action to be taken by our stockholders must be
effected at a duly called annual or special meeting of stockholders and not be taken by written consent or electronic transmission;
|
|
|
·
|
provide that stockholders seeking to present proposals before a
meeting of stockholders or to nominate candidates for election as directors at a meeting of stockholders must provide advance notice
in writing, and also specify requirements as to the form and content of a stockholder’s notice;
|
|
|
·
|
provide that special meetings of our stockholders may be called
only by the chairman of our board of directors, our chief executive officer or president or by our board of directors pursuant
to a resolution adopted by a majority of the total number of authorized directors; and
|
|
|
·
|
not provide for cumulative voting rights, therefore allowing the
holders of a majority of the shares of common stock entitled to vote in any election of directors to elect all of the directors
standing for election, if they should so choose.
|
The
amendment of any of these provisions requires approval by the holders of at least 66⅔% of the voting power of all of our
then-outstanding common stock entitled to vote generally in the election of directors, voting together as a single class.
The
combination of these provisions may make it more difficult for our existing stockholders to replace our board of directors as well
as for another party to obtain control of us by replacing our board of directors. Because our board of directors has the power
to retain and discharge our officers, these provisions could also make it more difficult for existing stockholders or another party
to effect a change in management. In addition, the authorization of undesignated preferred stock makes it possible for our board
of directors to issue preferred stock with voting or other rights or preferences that could impede the success of any attempt to
change our control.
These
provisions are intended to enhance the likelihood of continued stability in the composition of our board of directors and its policies
and to discourage coercive takeover practices and inadequate takeover bids. These provisions are also designed to reduce our vulnerability
to hostile takeovers and to discourage certain tactics that may be used in proxy fights. However, such provisions could have the
effect of discouraging others from making tender offers for our shares and may have the effect of delaying changes in our control
or management. As a consequence, these provisions may also inhibit fluctuations in the market price of our stock that could result
from actual or rumored takeover attempts. We believe that the benefits of these provisions, including increased protection of our
potential ability to negotiate with the proponent of an unfriendly or unsolicited proposal to acquire or restructure our company,
outweigh the disadvantages of discouraging takeover proposals, because negotiation of takeover proposals could result in an improvement
of their terms.
Choice of Forum
Our
amended and restated certificate of incorporation, as amended, and our amended and restated bylaws, as amended, provides that the
Court of Chancery of the State of Delaware is the exclusive forum for any derivative action or proceeding brought on our behalf;
any action asserting a breach of fiduciary duty; any action asserting a claim against us arising pursuant to the Delaware General
Corporation Law, our certificate of incorporation or our bylaws; or any action asserting a claim against us that is governed by
the internal affairs doctrine. The enforceability of similar choice of forum provisions in other companies’ certificates
of incorporation has been challenged in legal proceedings, and it is possible that, in connection with one or more actions or proceedings
described above, a court could find the choice of forum provisions contained in our amended and restated certificate of incorporation
to be inapplicable or unenforceable.
Listing
Our
common stock and Series A warrants are listed on The Nasdaq Capital Market under the trading symbols “OTLK” and “OTLKW,”
respectively. The applicable prospectus supplement will contain information, where applicable, as to any other listing, if any,
on The Nasdaq Capital Market or any securities market or other exchange of the preferred stock or other securities covered by such
prospectus supplement.
Transfer
Agent and Registrar
The transfer agent and registrar for our common
stock is American Stock Transfer & Trust Company, LLC. Its address is 6201 15th Avenue, Brooklyn, New York 11219.
DESCRIPTION OF DEBT SECURITIES
We may issue debt securities from time to time, in one or more
series, as either senior or subordinated debt or as senior or subordinated convertible debt. While the terms we have summarized
below will apply generally to any debt securities that we may offer under this prospectus, we will describe the particular terms
of any debt securities that we may offer in more detail in the applicable prospectus supplement. The terms of any debt securities
offered under a prospectus supplement may differ from the terms described below. Unless the context requires otherwise, whenever
we refer to the indenture, we also are referring to any supplemental indentures that specify the terms of a particular series of
debt securities.
We will issue the debt securities under the indenture that we
will enter into with the trustee named in the indenture. The indenture will be qualified under the Trust Indenture Act of 1939,
as amended, or the Trust Indenture Act. We have filed the form of indenture as an exhibit to the registration statement of which
this prospectus is a part, and supplemental indentures and forms of debt securities containing the terms of the debt securities
being offered will be filed as exhibits to the registration statement of which this prospectus is a part or will be incorporated
by reference from reports that we file with the SEC.
The following summary of material provisions of the debt securities
and the indenture is subject to, and qualified in its entirety by reference to, all of the provisions of the indenture applicable
to a particular series of debt securities. We urge you to read the applicable prospectus supplements and any related free writing
prospectuses related to the debt securities that we may offer under this prospectus, as well as the complete indenture that contains
the terms of the debt securities.
General
The indenture does not limit the amount of debt securities that
we may issue. It provides that we may issue debt securities up to the principal amount that we may authorize and may be in any
currency or currency unit that we may designate. Except for the limitations on consolidation, merger and sale of all or substantially
all of our assets contained in the indenture, the terms of the indenture do not contain any covenants or other provisions designed
to give holders of any debt securities protection against changes in our operations, financial condition or transactions involving
us.
We may issue the debt securities issued under the indenture
as “discount securities,” which means they may be sold at a discount below their stated principal amount. These debt
securities, as well as other debt securities that are not issued at a discount, may be issued with “original issue discount,”
or OID, for U.S. federal income tax purposes because of interest payment and other characteristics or terms of the debt securities.
Material U.S. federal income tax considerations applicable to debt securities issued with OID will be described in more detail
in any applicable prospectus supplement.
We will describe in the applicable prospectus supplement the
terms of the series of debt securities being offered, including:
|
|
·
|
the title of the series of debt securities;
|
|
|
·
|
any limit upon the aggregate principal amount that may be issued;
|
|
|
·
|
the maturity date or dates;
|
|
|
·
|
the form of the debt securities of the series;
|
|
|
·
|
the applicability of any guarantees;
|
|
|
·
|
whether or not the debt securities will be secured or unsecured, and
the terms of any secured debt;
|
|
|
·
|
whether the debt securities rank as senior debt, senior subordinated
debt, subordinated debt or any combination thereof, and the terms of any subordination;
|
|
|
·
|
if the price (expressed as a percentage of the aggregate principal
amount thereof) at which such debt securities will be issued is a price other than the principal amount thereof, the portion of
the principal amount thereof payable upon declaration of acceleration of the maturity thereof, or if applicable, the portion of
the principal amount of such debt securities that is convertible into another security or the method by which any such portion
shall be determined;
|
|
|
·
|
the interest rate or rates, which may be fixed or variable, or the
method for determining the rate or rates and the date interest will begin to accrue, the dates interest will be payable and the
regular record dates for interest payment dates or the method for determining such dates;
|
|
|
·
|
our right, if any, to defer payment of interest and the maximum length
of any such deferral period;
|
|
|
·
|
if applicable, the date or dates after which, or the period or periods
during which, and the price or prices at which, we may, at our option, redeem the series of debt securities pursuant to any optional
or provisional redemption provisions and the terms of those redemption provisions;
|
|
|
·
|
the date or dates, if any, on which, and the price or prices at which
we are obligated, pursuant to any mandatory sinking fund or analogous fund provisions or otherwise, to redeem, or at the holder’s
option to purchase, the series of debt securities and the currency or currency unit in which the debt securities are payable;
|
|
|
·
|
the denominations in which we will issue the series of debt securities,
if other than denominations of $1,000 and any integral multiple thereof;
|
|
|
·
|
any and all terms, if applicable, relating to any auction or remarketing
of the debt securities of that series and any security for our obligations with respect to such debt securities and any other terms
which may be advisable in connection with the marketing of debt securities of that series;
|
|
|
·
|
whether the debt securities of the series shall be issued in whole
or in part in the form of a global security or securities, the terms and conditions, if any, upon which such global security or
securities may be exchanged in whole or in part for other individual securities, and the depositary for such global security or
securities;
|
|
|
·
|
if applicable, the provisions relating to conversion or exchange of
any debt securities of the series and the terms and conditions upon which such debt securities will be so convertible or exchangeable,
including the conversion or exchange price, as applicable, or how it will be calculated and may be adjusted, any mandatory or optional
(at our option or the holders’ option) conversion or exchange features, the applicable conversion or exchange period and
the manner of settlement for any conversion or exchange;
|
|
|
·
|
if other than the full principal amount thereof, the portion of the
principal amount of debt securities of the series which shall be payable upon declaration of acceleration of the maturity thereof;
|
|
|
·
|
additions to or changes in the covenants applicable to the particular
debt securities being issued, including, among others, the consolidation, merger or sale covenant;
|
|
|
·
|
additions to or changes in the events of default with respect to the
securities and any change in the right of the trustee or the holders to declare the principal, premium, if any, and interest, if
any, with respect to such securities to be due and payable;
|
|
|
·
|
additions to or changes in or deletions of the provisions relating
to covenant defeasance and legal defeasance;
|
|
|
·
|
additions to or changes in the provisions relating to satisfaction
and discharge of the indenture;
|
|
|
·
|
additions to or changes in the provisions relating to the modification
of the indenture both with and without the consent of holders of debt securities issued under the indenture;
|
|
|
·
|
the currency of payment of debt securities if other than U.S. dollars
and the manner of determining the equivalent amount in U.S. dollars;
|
|
|
·
|
whether interest will be payable in cash or additional debt securities
at our or the holders’ option and the terms and conditions upon which the election may be made;
|
|
|
·
|
the terms and conditions, if any, upon which we will pay amounts in
addition to the stated interest, premium, if any and principal amounts of the debt securities of the series to any holder that
is not a “United States person” for federal tax purposes;
|
|
|
·
|
any restrictions on transfer, sale or assignment of the debt securities
of the series; and
|
|
|
·
|
any other specific terms, preferences, rights or limitations of, or
restrictions on, the debt securities, any other additions or changes in the provisions of the indenture, and any terms that may
be required by us or advisable under applicable laws or regulations.
|
Conversion or Exchange Rights
We will set forth in the applicable prospectus supplement the
terms on which a series of debt securities may be convertible into or exchangeable for our common stock or our other securities.
We will include provisions as to settlement upon conversion or exchange and whether conversion or exchange is mandatory, at the
option of the holder or at our option. We may include provisions pursuant to which the number of shares of our common stock or
our other securities that the holders of the series of debt securities receive would be subject to adjustment.
Consolidation, Merger or Sale
Unless we provide otherwise in the prospectus supplement applicable
to a particular series of debt securities, the indenture will not contain any covenant that restricts our ability to merge or consolidate,
or sell, convey, transfer or otherwise dispose of our assets as an entirety or substantially as an entirety. However, any successor
to or acquirer of such assets (other than a subsidiary of ours) must assume all of our obligations under the indenture or the debt
securities, as appropriate.
Events of Default under the Indenture
Unless we provide otherwise in the prospectus supplement applicable
to a particular series of debt securities, the following are events of default under the indenture with respect to any series of
debt securities that we may issue:
|
|
·
|
if we fail to pay any installment of interest on any series of debt
securities, as and when the same shall become due and payable, and such default continues for a period of 90 days; provided, however,
that a valid extension of an interest payment period by us in accordance with the terms of any indenture supplemental thereto shall
not constitute a default in the payment of interest for this purpose;
|
|
|
·
|
if we fail to pay the principal of, or premium, if any, on any series
of debt securities as and when the same shall become due and payable whether at maturity, upon redemption, by declaration or otherwise,
or in any payment required by any sinking or analogous fund established with respect to such series; provided, however, that a
valid extension of the maturity of such debt securities in accordance with the terms of any indenture supplemental thereto shall
not constitute a default in the payment of principal or premium, if any;
|
|
|
·
|
if we fail to observe or perform any other covenant or agreement contained
in the debt securities or the indenture, other than a covenant specifically relating to another series of debt securities, and
our failure continues for 90 days after we receive written notice of such failure, requiring the same to be remedied and stating
that such is a notice of default thereunder, from the trustee or holders of at least 25% in aggregate principal amount of the outstanding
debt securities of the applicable series; and
|
|
|
·
|
if specified events of bankruptcy, insolvency or reorganization occur.
|
If an event of default with respect to debt securities of any
series occurs and is continuing, other than an event of default specified in the last bullet point above, the trustee or the holders
of at least 25% in aggregate principal amount of the outstanding debt securities of that series, by notice to us in writing, and
to the trustee if notice is given by such holders, may declare the unpaid principal of, premium, if any, and accrued interest,
if any, due and payable immediately. If an event of default specified in the last bullet point above occurs with respect to us,
the principal amount of and accrued interest, if any, of each issue of debt securities then outstanding shall be due and payable
without any notice or other action on the part of the trustee or any holder.
The holders of a majority in principal amount of the outstanding
debt securities of an affected series may waive any default or event of default with respect to the series and its consequences,
except defaults or events of default regarding payment of principal, premium, if any, or interest, unless we have cured the default
or event of default in accordance with the indenture. Any waiver shall cure the default or event of default.
Subject to the terms of the indenture, if an event of default
under an indenture shall occur and be continuing, the trustee will be under no obligation to exercise any of its rights or powers
under such indenture at the request or direction of any of the holders of the applicable series of debt securities, unless such
holders have offered the trustee reasonable indemnity. The holders of a majority in principal amount of the outstanding debt securities
of any series will have the right to direct the time, method and place of conducting any proceeding for any remedy available to
the trustee, or exercising any trust or power conferred on the trustee, with respect to the debt securities of that series, provided
that:
|
|
·
|
the direction so given by the holder is not in conflict with any law
or the applicable indenture; and
|
|
|
·
|
subject to its duties under the Trust Indenture Act, the trustee need
not take any action that might involve it in personal liability or might be unduly prejudicial to the holders not involved in the
proceeding.
|
A holder of the debt securities of any series will have the
right to institute a proceeding under the indenture or to appoint a receiver or trustee, or to seek other remedies only if:
|
|
·
|
the holder has given written notice to the trustee of a continuing
event of default with respect to that series;
|
|
|
·
|
the holders of at least 25% in aggregate principal amount of the outstanding
debt securities of that series have made written request;
|
|
|
·
|
such holders have offered to the trustee indemnity satisfactory to
it against the costs, expenses and liabilities to be incurred by the trustee in compliance with the request; and
|
|
|
·
|
the trustee does not institute the proceeding, and does not receive
from the holders of a majority in aggregate principal amount of the outstanding debt securities of that series other conflicting
directions within 90 days after the notice, request and offer.
|
These limitations do not apply to a suit instituted by a holder
of debt securities if we default in the payment of the principal, premium, if any, or interest on, the debt securities.
We will periodically file statements with the trustee regarding
our compliance with specified covenants in the indenture.
Modification of Indenture; Waiver
We and the trustee may change an indenture
without the consent of any holders with respect to specific matters:
|
|
·
|
to cure any ambiguity, defect or inconsistency in the indenture or
in the debt securities of any series;
|
|
|
·
|
to comply with the provisions described above under “Description
of Debt Securities—Consolidation, Merger or Sale;”
|
|
|
·
|
to provide for uncertificated debt securities in addition to or in
place of certificated debt securities;
|
|
|
·
|
to add to our covenants, restrictions, conditions or provisions such
new covenants, restrictions, conditions or provisions for the benefit of the holders of all or any series of debt securities, to
make the occurrence, or the occurrence and the continuance, of a default in any such additional covenants, restrictions, conditions
or provisions an event of default or to surrender any right or power conferred upon us in the indenture;
|
|
|
·
|
to add to, delete from or revise the conditions, limitations, and
restrictions on the authorized amount, terms, or purposes of issue, authentication and delivery of debt securities, as set forth
in the indenture;
|
|
|
·
|
to make any change that does not adversely affect the interests of
any holder of debt securities of any series in any material respect;
|
|
|
·
|
to provide for the issuance of and establish the form and terms and
conditions of the debt securities of any series as provided above under “Description of Debt Securities—General”
to establish the form of any certifications required to be furnished pursuant to the terms of the indenture or any series of debt
securities, or to add to the rights of the holders of any series of debt securities;
|
|
|
·
|
to evidence and provide for the acceptance of appointment under any
indenture by a successor trustee; or
|
|
|
·
|
to comply with any requirements of the SEC in connection with the
qualification of any indenture under the Trust Indenture Act.
|
In addition, under the indenture, the rights of holders of a
series of debt securities may be changed by us and the trustee with the written consent of the holders of at least a majority in
aggregate principal amount of the outstanding debt securities of each series that is affected. However, unless we provide otherwise
in the prospectus supplement applicable to a particular series of debt securities, we and the trustee may make the following changes
only with the consent of each holder of any outstanding debt securities affected:
|
|
·
|
extending the fixed maturity of any debt securities of any series;
|
|
|
·
|
reducing the principal amount, reducing the rate of or extending the
time of payment of interest, or reducing any premium payable upon the redemption of any series of any debt securities; or
|
|
|
·
|
reducing the percentage of debt securities, the holders of which are
required to consent to any amendment, supplement, modification or waiver.
|
Discharge
Each indenture provides that we can elect to be discharged from
our obligations with respect to one or more series of debt securities, except for specified obligations, including obligations
to:
|
|
·
|
register the transfer or exchange of debt securities of the series;
|
|
|
·
|
replace stolen, lost or mutilated debt securities of the series;
|
|
|
·
|
pay principal of and premium and interest on any debt securities of
the series;
|
|
|
·
|
maintain paying agencies;
|
|
|
·
|
hold monies for payment in trust;
|
|
|
·
|
recover excess money held by the trustee;
|
|
|
·
|
compensate and indemnify the trustee; and
|
|
|
·
|
appoint any successor trustee.
|
In order to exercise our rights to be discharged, we must deposit
with the trustee money or government obligations sufficient to pay all the principal of, any premium, if any, and interest on,
the debt securities of the series on the dates payments are due.
Form, Exchange and Transfer
We will issue the debt securities of each series only in fully
registered form without coupons and, unless we provide otherwise in the applicable prospectus supplement, in denominations of $1,000
and any integral multiple thereof. The indenture provides that we may issue debt securities of a series in temporary or permanent
global form and as book-entry securities that will be deposited with, or on behalf of, The Depository Trust Company, or DTC, or
another depositary named by us and identified in the applicable prospectus supplement with respect to that series. To the extent
the debt securities of a series are issued in global form and as book-entry, a description of terms relating to any book-entry
securities will be set forth in the applicable prospectus supplement.
At the option of the holder, subject to the terms of the indenture
and the limitations applicable to global securities described in the applicable prospectus supplement, the holder of the debt securities
of any series can exchange the debt securities for other debt securities of the same series, in any authorized denomination and
of like tenor and aggregate principal amount.
Subject to the terms of the indenture and the limitations applicable
to global securities set forth in the applicable prospectus supplement, holders of the debt securities may present the debt securities
for exchange or for registration of transfer, duly endorsed or with the form of transfer endorsed thereon duly executed if so required
by us or the security registrar, at the office of the security registrar or at the office of any transfer agent designated by us
for this purpose. Unless otherwise provided in the debt securities that the holder presents for transfer or exchange, we will impose
no service charge for any registration of transfer or exchange, but we may require payment of any taxes or other governmental charges.
We will name in the applicable prospectus supplement the security
registrar, and any transfer agent in addition to the security registrar, that we initially designate for any debt securities. We
may at any time designate additional security registrars or transfer agents or rescind the designation of any security registrars
or transfer agent or approve a change in the office through which any security registrars or transfer agent acts, except that we
will be required to maintain a security registrar and a transfer agent in each place of payment for the debt securities of each
series.
If we elect to redeem the debt securities of any series, we
will not be required to:
|
|
·
|
issue, register the transfer of, or exchange any debt securities of
that series during a period beginning at the opening of business 15 days before the day of mailing of a notice of redemption
of any debt securities that may be selected for redemption and ending at the close of business on the day of the mailing; or
|
|
|
·
|
register the transfer of or exchange any debt securities so selected
for redemption, in whole or in part, except the unredeemed portion of any debt securities we are redeeming in part.
|
Information Concerning the Trustee
The trustee, other than during the occurrence and continuance
of an event of default under an indenture, undertakes to perform only those duties as are specifically set forth in the applicable
indenture. Upon an event of default under an indenture, the trustee must use the same degree of care as a prudent person would
exercise or use in the conduct of his or her own affairs. Subject to this provision, the trustee is under no obligation to exercise
any of the powers given it by the indenture at the request of any holder of debt securities unless it is offered reasonable security
and indemnity against the costs, expenses and liabilities that it might incur.
Payment and Paying Agents
Unless we otherwise indicate in the applicable prospectus supplement,
we will make payment of the interest on any debt securities on any interest payment date to the person in whose name the debt securities,
or one or more predecessor securities, are registered at the close of business on the regular record date for the interest.
We will pay principal of and any premium and interest on the
debt securities of a particular series at the office of the paying agents designated by us, except that unless we otherwise indicate
in the applicable prospectus supplement, we will make interest payments by check that we will mail to the holder or by wire transfer
to certain holders. Unless we otherwise indicate in the applicable prospectus supplement, we will designate the corporate trust
office of the trustee as our sole paying agent for payments with respect to debt securities of each series. We will name in the
applicable prospectus supplement any other paying agents that we initially designate for the debt securities of a particular series.
We may at any time rescind the designation or any paying agent or approve a change in the office through which any paying agent
acts, except that we will be required to maintain a paying agent in each place of payment for the debt securities of a particular
series.
All money we pay to a paying agent or the trustee for the payment
of the principal of or any premium or interest on any debt securities that remains unclaimed at the end of two years after such
principal, premium or interest has become due and payable will be repaid to us, and the holder of the debt security thereafter
may look only to us for payment thereof.
Governing Law
The indenture and the debt securities will be governed by and
construed in accordance with the internal laws of the State of New York, except to the extent that the Trust Indenture Act of 1939
is applicable.
DESCRIPTION OF WARRANTS
The following description, together with the additional information
we may include in any applicable prospectus supplement and in any related free writing prospectus that we may authorize to be distributed
to you, summarizes the material terms and provisions of the warrants that we may offer under this prospectus, which may consist
of warrants to purchase common stock, preferred stock or debt securities and may be issued in one or more series. Warrants may
be offered independently or in combination with common stock, preferred stock or debt securities offered by any prospectus supplement.
While the terms we have summarized below will apply generally to any warrants that we may offer under this prospectus, we will
describe the particular terms of any series of warrants in more detail in the applicable prospectus supplement. The following description
of warrants will apply to the warrants offered by this prospectus unless we provide otherwise in the applicable prospectus supplement.
The applicable prospectus supplement for a particular series of warrants may specify different or additional terms.
We have filed forms of the warrant agreements and forms of warrant
certificates containing the terms of the warrants that may be offered as exhibits to the registration statement of which this prospectus
is a part. We will file as exhibits to the registration statement of which this prospectus is a part, or will incorporate by reference
from reports that we file with the SEC, the form of warrant and/or the warrant agreement and warrant certificate, as applicable,
that contain the terms of the particular series of warrants we are offering, and any supplemental agreements, before the issuance
of such warrants. The following summaries of material terms and provisions of the warrants are subject to, and qualified in their
entirety by reference to, all the provisions of the form of warrant and/or the warrant agreement and warrant certificate, as applicable,
and any supplemental agreements applicable to a particular series of warrants that we may offer under this prospectus. We urge
you to read the applicable prospectus supplement related to the particular series of warrants that we may offer under this prospectus,
as well as any related free writing prospectus, and the complete form of warrant and/or the warrant agreement and warrant certificate,
as applicable, and any supplemental agreements, that contain the terms of the warrants.
General
We will describe in the applicable prospectus supplement the
terms of the series of warrants being offered, including, to the extent applicable:
|
|
·
|
the offering price and aggregate number of warrants offered;
|
|
|
·
|
the currency for which the warrants may be purchased;
|
|
|
·
|
the designation and terms of the securities with which the warrants are issued and the number of warrants issued with each
such security or each principal amount of such security;
|
|
|
·
|
in the case of warrants to purchase debt securities, the principal amount of debt securities purchasable upon exercise of one
warrant and the price at, and currency in which, this principal amount of debt securities may be purchased upon such exercise;
|
|
|
·
|
in the case of warrants to purchase common stock or preferred stock, the number of shares of common stock or preferred stock,
as the case may be, purchasable upon the exercise of one warrant and the price at which these shares may be purchased upon such
exercise;
|
|
|
·
|
the effect of any merger, consolidation, sale or other disposition of our business on the warrant agreements and the warrants;
|
|
|
·
|
the terms of any rights to redeem or call the warrants;
|
|
|
·
|
any provisions for changes to or adjustments in the exercise price or number of securities issuable upon exercise of the warrants;
|
|
|
·
|
the dates on which the right to exercise the warrants will commence and expire;
|
|
|
·
|
the manner in which the warrant agreements and warrants may be modified;
|
|
|
·
|
a discussion of material or special U.S. federal income tax considerations of holding or exercising the warrants;
|
|
|
·
|
the terms of the securities issuable upon exercise of the warrants; and
|
|
|
·
|
any other specific terms, preferences, rights or limitations of or restrictions on the warrants.
|
Before exercising their warrants, holders of warrants will not
have any of the rights of holders of the securities purchasable upon such exercise, including:
|
|
·
|
in the case of warrants to purchase common stock or preferred stock, the right to receive dividends, if any, or, payments upon
our liquidation, dissolution or winding up or to exercise voting rights, if any; or
|
|
|
·
|
in the case of warrants to purchase debt securities, the right to receive payments of principal of, or premium, if any, or
interest on, the debt securities purchasable upon exercise or to enforce covenants in the applicable indenture.
|
Exercise of Warrants
Each warrant will entitle the holder to purchase the securities
that we specify in the applicable prospectus supplement at the exercise price that we describe in the applicable prospectus supplement.
The warrants may be exercised as set forth in the prospectus supplement relating to the warrants offered. Unless we otherwise specify
in the applicable prospectus supplement, warrants may be exercised at any time up to the specified time on the expiration date
that we set forth in the applicable prospectus supplement. After the close of business on the expiration date, unexercised warrants
will become void.
Upon receipt of payment and the warrant or warrant certificate,
as applicable, properly completed and duly executed at the corporate trust office of the warrant agent, if any, or any other office,
including ours, indicated in the prospectus supplement, we will, as soon as practicable, issue and deliver the securities purchasable
upon such exercise. If less than all of the warrants (or the warrants represented by such warrant certificate) are exercised, a
new warrant or a new warrant certificate, as applicable, will be issued for the remaining warrants.
Governing Law
Unless we provide otherwise in the applicable prospectus supplement,
the warrants and warrant agreements will be governed by and construed in accordance with the laws of the State of New York.
Enforceability of Rights by Holders of Warrants
Each warrant agent, if any, will act solely as our agent under
the applicable warrant agreement and will not assume any obligation or relationship of agency or trust with any holder of any warrant.
A single bank or trust company may act as warrant agent for more than one issue of warrants. A warrant agent will have no duty
or responsibility in case of any default by us under the applicable warrant agreement or warrant, including any duty or responsibility
to initiate any proceedings at law or otherwise, or to make any demand upon us. Any holder of a warrant may, without the consent
of the related warrant agent or the holder of any other warrant, enforce by appropriate legal action its right to exercise, and
receive the securities purchasable upon exercise of, its warrants.
LEGAL OWNERSHIP OF SECURITIES
We can issue securities in registered form or in the form of
one or more global securities. We describe global securities in greater detail below. We refer to those persons who have securities
registered in their own names on the books that we or any applicable trustee, depositary or warrant agent maintain for this purpose
as the “holders” of those securities. These persons are the legal holders of the securities. We refer to those persons
who, indirectly through others, own beneficial interests in securities that are not registered in their own names, as “indirect
holders” of those securities. As we discuss below, indirect holders are not legal holders, and investors in securities issued
in book-entry form or in street name will be indirect holders.
Book-Entry Holders
We may issue securities in book-entry form only, as we will
specify in the applicable prospectus supplement. This means securities may be represented by one or more global securities registered
in the name of a financial institution that holds them as depositary on behalf of other financial institutions that participate
in the depositary’s book-entry system. These participating institutions, which are referred to as participants, in turn,
hold beneficial interests in the securities on behalf of themselves or their customers.
Only the person in whose name a security is registered is recognized
as the holder of that security. Securities issued in global form will be registered in the name of the depositary or its participants.
Consequently, for securities issued in global form, we will recognize only the depositary as the holder of the securities, and
we will make all payments on the securities to the depositary. The depositary passes along the payments it receives to its participants,
which in turn pass the payments along to their customers who are the beneficial owners. The depositary and its participants do
so under agreements they have made with one another or with their customers; they are not obligated to do so under the terms of
the securities.
As a result, investors in a global security will not own securities
directly. Instead, they will own beneficial interests in a global security, through a bank, broker or other financial institution
that participates in the depositary’s book-entry system or holds an interest through a participant. As long as the securities
are issued in global form, investors will be indirect holders, and not legal holders, of the securities.
Street Name Holders
We may terminate a global security or issue securities in non-global
form. In these cases, investors may choose to hold their securities in their own names or in “street name.” Securities
held by an investor in street name would be registered in the name of a bank, broker or other financial institution that the investor
chooses, and the investor would hold only a beneficial interest in those securities through an account he or she maintains at that
institution.
For securities held in street name, we or any applicable trustee
or depositary will recognize only the intermediary banks, brokers and other financial institutions in whose names the securities
are registered as the holders of those securities, and we or any such trustee or depositary will make all payments on those securities
to them. These institutions pass along the payments they receive to their customers who are the beneficial owners, but only because
they agree to do so in their customer agreements or because they are legally required to do so. Investors who hold securities in
street name will be indirect holders, not legal holders, of those securities.
Legal Holders
Our obligations, as well as the obligations of any applicable
trustee or any third party employed by us or a trustee, run only to the legal holders of the securities. We do not have obligations
to investors who hold beneficial interests in global securities, in street name or by any other indirect means. This will be the
case whether an investor chooses to be an indirect holder of a security or has no choice because we are issuing the securities
only in global form.
For example, once we make a payment or give a notice to the
holder, we have no further responsibility for the payment or notice even if that holder is required, under agreements with its
participants or customers or by law, to pass it along to the indirect holders but does not do so. Similarly, we may want to obtain
the approval of the holders to amend an indenture, to relieve us of the consequences of a default or of our obligation to comply
with a particular provision of the indenture or for other purposes. In such an event, we would seek approval only from the holders,
and not the indirect holders, of the securities. Whether and how the holders contact the indirect holders is up to the holders.
Special Considerations for Indirect Holders
If you hold securities through a bank, broker or other financial
institution, either in book-entry form or in street name, you should check with your own institution to find out:
|
|
·
|
how it handles securities payments and notices;
|
|
|
·
|
whether it imposes fees or charges;
|
|
|
·
|
how it would handle a request for the holders’ consent, if ever required;
|
|
|
·
|
whether and how you can instruct it to send you securities registered in your own name so you can be a holder, if that is permitted
in the future;
|
|
|
·
|
how it would exercise rights under the securities if there were a default or other event triggering the need for holders to
act to protect their interests; and
|
|
|
·
|
if the securities are in book-entry form, how the depositary’s rules and procedures will affect these matters.
|
Global Securities
A global security is a security that represents one or any other
number of individual securities held by a depositary. Generally, all securities represented by the same global securities will
have the same terms.
Each security issued in book-entry form will be represented
by a global security that we issue to, deposit with and register in the name of a financial institution or its nominee that we
select. The financial institution that we select for this purpose is called the depositary. Unless we specify otherwise in the
applicable prospectus supplement, DTC will be the depositary for all securities issued in book-entry form.
A global security may not be transferred to or registered in
the name of anyone other than the depositary, its nominee or a successor depositary, unless special termination situations arise.
We describe those situations below under the section titled “Special Situations When a Global Security Will Be Terminated.”
As a result of these arrangements, the depositary, or its nominee, will be the sole registered owner and holder of all securities
represented by a global security, and investors will be permitted to own only beneficial interests in a global security. Beneficial
interests must be held by means of an account with a broker, bank or other financial institution that in turn has an account with
the depositary or with another institution that does. Thus, an investor whose security is represented by a global security will
not be a legal holder of the security, but only an indirect holder of a beneficial interest in the global security.
If the prospectus supplement for a particular security indicates
that the security will be issued in global form only, then the security will be represented by a global security at all times unless
and until the global security is terminated. If termination occurs, we may issue the securities through another book-entry clearing
system or decide that the securities may no longer be held through any book-entry clearing system.
Special Considerations for Global Securities
The rights of an indirect holder relating to a global security
will be governed by the account rules of the investor’s financial institution and of the depositary, as well as general laws
relating to securities transfers. We do not recognize an indirect holder as a holder of securities and instead deal only with the
depositary that holds the global security.
If securities are issued only in the form of a global security,
an investor should be aware of the following:
|
|
·
|
an investor cannot cause the securities to be registered in his or her name, and cannot obtain non-global certificates for
his or her interest in the securities, except in the special situations we describe below;
|
|
|
·
|
an investor will be an indirect holder and must look to his or her own bank or broker for payments on the securities and protection
of his or her legal rights relating to the securities, as we describe above;
|
|
|
·
|
an investor may not be able to sell interests in the securities to some insurance companies and to other institutions that
are required by law to own their securities in non-book-entry form;
|
|
|
·
|
an investor may not be able to pledge his or her interest in a global security in circumstances where certificates representing
the securities must be delivered to the lender or other beneficiary of the pledge in order for the pledge to be effective;
|
|
|
·
|
the depositary’s policies, which may change from time to time, will govern payments, transfers, exchanges and other matters
relating to an investor’s interest in a global security;
|
|
|
·
|
we and any applicable trustee have no responsibility for any aspect of the depositary’s actions or for its records of
ownership interests in a global security, nor do we or any applicable trustee supervise the depositary in any way;
|
|
|
·
|
the depositary may, and we understand that DTC will, require that those who purchase and sell interests in a global security
within its book-entry system use immediately available funds, and your bank or broker may require you to do so as well; and
|
|
|
·
|
financial institutions that participate in the depositary’s book-entry system, and through which an investor holds its
interest in a global security, may also have their own policies affecting payments, transfers, exchanges, notices and other matters
relating to the securities.
|
There may be more than one financial intermediary in the chain
of ownership for an investor. We do not monitor and are not responsible for the actions of any of those intermediaries.
Special Situations When a Global Security Will Be Terminated
In a few special situations described below, the global security
will terminate and interests in it will be exchanged for physical certificates representing those interests. After that exchange,
the choice of whether to hold securities directly or in street name will be up to the investor. Investors must consult their own
banks or brokers to find out how to have their interests in securities transferred to their own name, so that they will be direct
holders. We have described the rights of holders and street name investors above.
Unless we provide otherwise in the applicable prospectus supplement,
the global security will terminate when the following special situations occur:
|
|
·
|
if the depositary notifies us that it is unwilling, unable or no longer qualified to continue as depositary for that global
security and we do not appoint another institution to act as depositary within 90 days;
|
|
|
·
|
if we notify any applicable trustee that we wish to terminate that global security; or
|
|
|
·
|
if an event of default has occurred with regard to securities represented by that global security and has not been cured or
waived.
|
The applicable prospectus supplement may also list additional
situations for terminating a global security that would apply only to the particular series of securities covered by the prospectus
supplement. When a global security terminates, the depositary, and not we or any applicable trustee, is responsible for deciding
the names of the institutions that will be the initial direct holders.
PLAN OF DISTRIBUTION
We may sell the securities from time to time pursuant to underwritten
public offerings, direct sales to the public, “at the market” offerings, negotiated transactions, block trades or a
combination of these methods. We may sell the securities to or through underwriters or dealers, through agents, or directly to
one or more purchasers. We may distribute securities from time to time in one or more transactions:
|
|
·
|
at a fixed price or prices, which may be changed;
|
|
|
·
|
at market prices prevailing at the time of sale;
|
|
|
·
|
at prices related to such prevailing market prices; or
|
A prospectus supplement or supplements (and any related free
writing prospectus that we may authorize to be provided to you) will describe the terms of the offering of the securities, including,
to the extent applicable:
|
|
·
|
the name or names of the underwriters or agents, if any;
|
|
|
·
|
the purchase price of the securities or other consideration therefor, and the proceeds, if any, we will receive from the sale;
|
|
|
·
|
any over-allotment or other options under which underwriters may purchase additional securities from us;
|
|
|
·
|
any agency fees or underwriting discounts and other items constituting agents’ or underwriters’ compensation;
|
|
|
·
|
any public offering price;
|
|
|
·
|
any discounts or concessions allowed or reallowed or paid to dealers; and
|
|
|
·
|
any securities exchange or market on which the securities may be listed.
|
Only underwriters named in the prospectus supplement will be
underwriters of the securities offered by the prospectus supplement.
If underwriters are used in the sale, they will acquire the
securities for their own account and may resell the securities from time to time in one or more transactions at a fixed public
offering price or at varying prices determined at the time of sale. The obligations of the underwriters to purchase the securities
will be subject to the conditions set forth in the applicable underwriting agreement. We may offer the securities to the public
through underwriting syndicates represented by managing underwriters or by underwriters without a syndicate. Subject to certain
conditions, the underwriters will be obligated to purchase all of the securities offered by the prospectus supplement, other than
securities covered by any over-allotment or other option. Any public offering price and any discounts or concessions allowed or
reallowed or paid to dealers may change from time to time. We may use underwriters with whom we have a material relationship. We
will describe in the prospectus supplement, naming the underwriter, the nature of any such relationship.
We may sell securities directly or through agents we designate
from time to time. We will name any agent involved in the offering and sale of securities and we will describe any commissions
we will pay the agent in the prospectus supplement. Unless the prospectus supplement states otherwise, our agent will act on a
best-efforts basis for the period of its appointment.
We may authorize agents or underwriters to solicit offers by
certain types of institutional investors to purchase securities from us at the public offering price set forth in the prospectus
supplement pursuant to delayed delivery contracts providing for payment and delivery on a specified date in the future. We will
describe the conditions to these contracts and the commissions we must pay for solicitation of these contracts in the prospectus
supplement.
We may provide agents and underwriters with indemnification
against civil liabilities, including liabilities under the Securities Act, or contribution with respect to payments that the agents
or underwriters may make with respect to these liabilities. Agents and underwriters may engage in transactions with, or perform
services for, us in the ordinary course of business.
All securities we may offer, other than common stock, will be
new issues of securities with no established trading market. Any underwriters may make a market in these securities, but will not
be obligated to do so and may discontinue any market making at any time without notice. We cannot guarantee the liquidity of the
trading markets for any securities.
Any underwriter may engage in over-allotment, stabilizing transactions,
short-covering transactions and penalty bids in accordance with Regulation M under the Exchange Act. Over-allotment involves sales
in excess of the offering size, which create a short position. Stabilizing transactions permit bids to purchase the underlying
security so long as the stabilizing bids do not exceed a specified maximum price. Syndicate-covering or other short-covering transactions
involve purchases of the securities, either through exercise of the over-allotment option or in the open market after the distribution
is completed, to cover short positions. Penalty bids permit the underwriters to reclaim a selling concession from a dealer when
the securities originally sold by the dealer are purchased in a stabilizing or covering transaction to cover short positions. Those
activities may cause the price of the securities to be higher than it would otherwise be. If commenced, the underwriters may discontinue
any of the activities at any time.
Any underwriters or agents that are qualified market makers
on The Nasdaq Capital Market may engage in passive market making transactions in the common stock on The Nasdaq Capital Market
in accordance with Regulation M under the Exchange Act, during the business day prior to the pricing of the offering, before the
commencement of offers or sales of the common stock. Passive market makers must comply with applicable volume and price limitations
and must be identified as passive market makers. In general, a passive market maker must display its bid at a price not in excess
of the highest independent bid for such security; if all independent bids are lowered below the passive market maker’s bid,
however, the passive market maker’s bid must then be lowered when certain purchase limits are exceeded. Passive market making
may stabilize the market price of the securities at a level above that which might otherwise prevail in the open market and, if
commenced, may be discontinued at any time.
In compliance with guidelines of the Financial Industry Regulatory
Authority, or FINRA, the maximum consideration or discount to be received by any FINRA member or independent broker dealer may
not exceed 8% of the aggregate amount of the securities offered pursuant to this prospectus and any applicable prospectus supplement.
LEGAL MATTERS
Unless otherwise indicated in the applicable prospectus supplement,
the validity of the securities offered by this prospectus and any supplement thereto, will be passed upon for us by Cooley LLP.
Additional legal matters may be passed upon for us or any underwriters, dealers or agents, by counsel that we will name in the
applicable prospectus supplement.
EXPERTS
The consolidated
financial statements of Outlook Therapeutics, Inc. as of September 30, 2018 and 2017, and for the years then ended, have been incorporated
by reference in this prospectus in reliance upon the report of KPMG LLP, independent registered public accounting firm, incorporated
by reference herein, and upon the authority of said firm as experts in accounting and auditing. The audit report covering the September
30, 2018 consolidated financial statements contains an explanatory paragraph that states that the Company has incurred recurring
losses and negative cash flows from operations and has an accumulated deficit at September 30, 2018 of $216.3 million,
$13.5 million of senior secured notes that may become due in fiscal 2019 and $4.6 million of unsecured indebtedness, $1.0 million
of which is due on demand, and $3.6 million of which matures December 22, 2018, that raise substantial doubt about the Company’s
ability to continue as a going concern. The consolidated financial statements do not include any adjustments that might result
from the outcome of that uncertainty.
WHERE YOU CAN FIND MORE INFORMATION
This prospectus is part of a registration statement we filed
with the SEC. This prospectus does not contain all of the information set forth in the registration statement and the exhibits
to the registration statement. For further information with respect to us and the securities we are offering under this prospectus,
we refer you to the registration statement and the exhibits and schedules filed as a part of the registration statement. You should
rely only on information contained in this prospectus or incorporated by reference into this prospectus. We have not authorized
any person to provide you with different information. We are not making an offer of these securities in any state where the offer
is not permitted. You should not assume that the information in this prospectus is accurate as of any date other than the date
on the front page of this prospectus, regardless of the time of delivery of this prospectus or any sale of the securities offered
by this prospectus.
We file annual, quarterly and current reports, proxy statements
and other information with the SEC. Our SEC filings are available to the public at the SEC’s website at http://www.sec.gov.
We maintain a website at http://www.outlooktherapeutics.com.
Information contained in or accessible through our website does not constitute a part of this prospectus.
INCORPORATION OF CERTAIN INFORMATION
BY REFERENCE
The SEC allows us to incorporate by reference the information
we file with it, which means that we can disclose important information to you by referring you to another document that we have
filed separately with the SEC. You should read the information incorporated by reference because it is an important part of this
prospectus. Information in this prospectus supersedes information incorporated by reference that we filed with the SEC prior to
the date of this prospectus, while information that we file later with the SEC will automatically update and supersede the information
in this prospectus. We incorporate by reference into this prospectus and the registration statement of which this prospectus is
a part the information and documents listed below that we have filed with the SEC (Commission File No. 001-37759):
|
|
·
|
our Annual Report on Form 10-K for the fiscal year ended September 30,
2018, filed with the SEC on December 18, 2018;
|
|
|
·
|
our Quarterly Reports on Form 10-Q for the quarters ended December 31, 2018 and March 31, 2019, filed with the SEC on February 14, 2019 and May 15, 2019, respectively;
|
|
|
·
|
our Current Reports on Form 8-K, filed with the SEC on October 26, 2018, October 30, 2018, November 9, 2018, December 6, 2018, December 18, 2018, January 22, 2019, March 5, 2019, March 18, 2019, April 5, 2019 and April 26, 2019, to the extent the information in such reports is filed and
not furnished; and
|
|
|
·
|
the description of our common stock set forth in our registration
statement on Form 8-A, filed with the SEC on April 29, 2016, as amended on May 11, 2016, including any further amendments
thereto or reports filed for the purposes of updating this description.
|
We also incorporate by reference any future filings (other than
Current Reports furnished under Item 2.02 or Item 7.01 of Form 8-K and exhibits filed on such form that are related to
such items unless such Form 8-K expressly provides to the contrary) made with the SEC pursuant to Sections 13(a), 13(c),
14 or 15(d) of the Exchange Act, including those made after the date of the initial filing of the registration statement of which
this prospectus is a part and prior to effectiveness of such registration statement, until we file a post-effective amendment that
indicates the termination of the offering of the common stock made by this prospectus and will become a part of this prospectus
from the date that such documents are filed with the SEC. Information in such future filings updates and supplements the information
provided in this prospectus. Any statements in any such future filings will automatically be deemed to modify and supersede any
information in any document we previously filed with the SEC that is incorporated or deemed to be incorporated herein by reference
to the extent that statements in the later filed document modify or replace such earlier statements.
We will furnish without charge to each person, including any
beneficial owner, to whom a prospectus is delivered, upon written or oral request, a copy of any or all of the documents incorporated
by reference into this prospectus but not delivered with the prospectus, including exhibits that are specifically incorporated
by reference into such documents. You should direct any requests for documents to Outlook Therapeutics, Inc., Attention: Corporate
Secretary, 7 Clarke Drive, Cranbury, New Jersey 08512. Our phone number is (609) 619-3990. You may also view the documents that
we file with the SEC and incorporate by reference in this prospectus on our corporate website at www.outlooktherapeutics.com. The
information on our website is not incorporated by reference and is not a part of this prospectus.

8,407,411 Shares of Common Stock
PROSPECTUS Supplement
H.C. Wainwright &
Co.
June 22, 2020
Outlook Therapeutics (NASDAQ:OTLK)
Historical Stock Chart
From Mar 2024 to Apr 2024
Outlook Therapeutics (NASDAQ:OTLK)
Historical Stock Chart
From Apr 2023 to Apr 2024