SAGE Journals Publishes New Data Revealing NEXGEL’s SilverSeal Hydrogel Dressing Improves Postsurgical Scarring
October 03 2022 - 7:30AM

NEXGEL, Inc. (NASDAQ: NXGL, NXGLW), (“NEXGEL” or the “Company”), a
leading provider of ultra-gentle, high-water-content hydrogel
products for healthcare and consumer applications, announced today
that SAGE Journals’ Scars, Burns and Healing publication, a
peer-reviewed journal that focuses on scar and burns research, has
published new data from a study evaluating the impact of NEXGEL’s
SilverSeal® hydrogel dressing on postoperative scarring and
complications. The results demonstrated significant scar
improvement in patients treated with SilverSeal.
Study Design:
- SilverSeal was used post-surgery as
an antimicrobial dressing to evaluate its ability to aid in the
healing process.
- 40 foot and ankle patients (aged 32
to 66) were included in the double-blind, randomized study with 20
patients in each group.
- Postoperatively, the treatment
group was given a SilverSeal hydrogel dressing, and the control
group was treated with a standard petroleum-based dressing.
- Scarring and complications were
evaluated, and follow-up was performed at two, six and 12 weeks
post-surgery using the Patient and Observer Scar Assessment Scale
(POSAS), which measures a scar’s quality from both the physician
and patient’s perspective.
- Patients were not excluded in the
study based on gender, age or comorbidity.
Results:
- The treatment group demonstrated
statistically significant improvements in the POSAS observer score
and observer opinion at six and 12 weeks (p<0.001).
- Patients in the treatment group
reported significantly reduced pain, compared with the control
group, at 12 weeks (p<0.001).
- Patients reported that itching at
the surgical site declined over time for both groups (p<0.001),
with significantly less itching reported by the treatment group
(p=0.027).
- The scar area considerably
decreased for the treatment group compared to the control group at
six and 12 weeks (p≤0.002).
- Neither group experienced
post-operative complications.
“Surgical incisions all result in scar
formation, some more pronounced than others,” said Dr. Stephen
Brigido, Section Chief of Foot and Ankle Reconstruction and
Fellowship Director at the Orthopedic Institute at Lehigh Valley
Hospital. “As skin heals and scar formation occurs, patients often
experience discomfort such as sharp pain, burning and itching.
After 12 weeks of observing these patient groups, we saw noteworthy
differences when comparing the control group with the treatment
group. We found that patients treated with NEXGEL’s SilverSeal
hydrogel dressing experienced significantly less negative symptoms
and regained their skin functionality quickly. We also found that
the overall scar area significantly decreased in patients who used
SilverSeal.”
Dr. Brigido continued, “The most important
result we observed was a significant decrease in skin and scar
incisional pain experienced by patients in the hydrogel group,
which was observed throughout the study. As a surgeon, this
suggests a marked change in how patients undergoing surgery should
be treated. These results are promising for not only for foot and
ankle surgery but also for healing all postsurgical incisions.”
The study took place at Lehigh Valley Orthopedic
Institute.
About NEXGEL, INC.
NEXGEL is a leading provider of ultra-gentle,
high-water-content hydrogels for healthcare and consumer
applications. Based in Langhorne, Pa., the Company has developed
and manufactured electron-beam, cross-linked hydrogels for over two
decades. Alongside its strategic partners, NEXGEL has formulated
more than 200 different combinations to bring natural ingredients
to gentle skin patches that can be worn for long periods of time
with little to no irritation.
Forward-Looking Statement
This press release contains "forward-looking
statements" within the meaning of Section 27A of the Securities Act
of 1933, as amended, and Section 21E of the Securities Exchange Act
of 1934, as amended (the "Exchange Act") (which Sections were
adopted as part of the Private Securities Litigation Reform Act of
1995). Statements preceded by, followed by or that otherwise
include the words "believe," "anticipate," "estimate," "expect,"
"intend," "plan," "project," "prospects," "outlook," and similar
words or expressions, or future or conditional verbs, such as
"will," "should," "would," "may," and "could," are generally
forward-looking in nature and not historical facts. These
forward-looking statements involve known and unknown risks,
uncertainties and other factors which may cause the Company's
actual results, performance, or achievements to be materially
different from any anticipated results, performance, or
achievements for many reasons including the impact of the COVID-19
pandemic. The Company disclaims any intention to, and undertakes no
obligation to, revise any forward-looking statements, whether as a
result of new information, a future event, or otherwise. For
additional risks and uncertainties that could impact the Company's
forward-looking statements, please see the Company's Annual Report
on Form 10-K for the year ended December 31, 2021, including but
not limited to the discussion under "Risk Factors" therein, which
the Company filed with the SEC and which may be viewed
at http://www.sec.gov/.
Investor Contact:Valter
PintoKCSA Strategic
Communications212.896.1254valter@kcsa.com
Media Contacts:Raquel Cona /
Michaela FawcettKCSA Strategic Communications212.896.1204 /
978.995.4683rcona@kcsa.com / mfawcett@kcsa.com
###
NexGel (NASDAQ:NXGL)
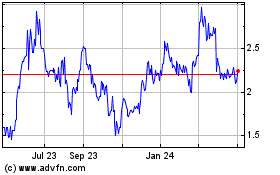
Historical Stock Chart
From Mar 2024 to Apr 2024
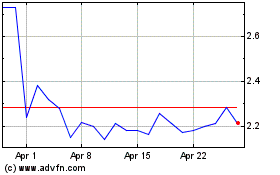
NexGel (NASDAQ:NXGL)
Historical Stock Chart
From Apr 2023 to Apr 2024