Nemaura Medical Plans Head-to-head Study with Major Glucose Monitoring System
February 25 2020 - 9:17AM
Business Wire
Studies planned to compare sugarBEAT® and
major continuous glucose monitor sensor, seeking to position
sugarBEAT® as both a possible alternative and complementary device
for people with diabetes
Nemaura Medical, Inc. (NASDAQ: NMRD) (“Nemaura” or the
“Company”), a medical technology company focused on developing
micro-systems-based wearable diagnostic devices and currently
commercializing sugarBEAT® its non-invasive and flexible continuous
glucose monitor (“CGM”), together with BEAT®diabetes, a planned
health subscription service designed to help people with Type 2
diabetes and prediabetes through personalized lifestyle coaching,
today, announced it’s planning to initiate a user study comparing
sugarBEAT® directly against a highly successful major incumbent CGM
sensor with the goal of positioning sugarBEAT® as a non-invasive,
daily/flexible, cost-effective alternative to traditional,
expensive invasive CGM.
“Our decision to go head-to-head vs. a hugely successful CGM
sensor was based on positive feedback we received from recent
meetings with public health insurers in key territories in Europe,”
stated Dr. Faz Chowdhury, CEO of Nemaura. “We believe that most
people with diabetes do not currently use any continuous glucose
monitoring system due to the high costs and the invasiveness of
current products. We believe that sugarBEAT® changes this paradigm
and is the first non-invasive CGM to provide the [masses] an option
for daily monitoring whenever they choose at an affordable price
point. We believe that this level of flexibility is a better option
than any alternative CGM devices with either 7 day, 10 day, or 14
day sensor wear time currently available.”
“We believe that sugarBEAT®’s flexibility empowers users with
very powerful trend data at a lower cost compared to current CGM’s,
which we believe will encourage broad adoption of the system”.
Several studies are planned over the course of this year
designed to demonstrate the effectiveness of sugarBEAT® to increase
Time-In-Range, thus reducing HbA1C with intermittent
(non-consecutive days) use over a few days per week or even month.
Initial results from studies comparing the various market leading
CGM devices indicates sugarBEAT’s® accuracy (MARD mean absolute
relative difference), to be on par with the major incumbent
invasive CGM sensors, using single finger-stick blood glucose meter
calibration in real-life settings.
The first phase of the commercial launch of sugarBEAT® began in
the UK after the Company received CE Mark approval in 2019. In this
initial phase, devices were supplied to a limited cohort of users
while the Company sought to ramp up its manufacturing
operations.
The UK licensee of sugarBEAT®, DB Ethitronix, are currently
finalizing the launch of online sales of sugarBEAT®. Concurrently,
Nemaura is finalizing the launch of its subscription-based service,
BEAT®diabetes, aimed at reversing Type 2 diabetes and preventing
prediabetes from the onset of diabetes.
About Nemaura Medical, Inc.:
Nemaura Medical, Inc. (NMRD), is a medical technology company
commercializing BEAT®diabetes, a health subscription service
designed to help people with diabetes and prediabetes better manage
diabetes and reverse Type 2 diabetes or prevent diabetes through
1-on-1 lifestyle coaching and behavior driven by real time
continuous glucose monitoring (CGM) and daily glucose trend data
provided by sugarBEAT®, a non-invasive and flexible CGM.
For more information visit: www.NemauraMedical.com.
Cautionary Statement Regarding Forward-Looking
Statements:
The statements in this press release that are not historical
facts, and may constitute forward-looking statements that are based
on current expectations and are subject to risks and uncertainties
that could cause actual future results to differ materially from
those expressed or implied by such statements. Those risks and
uncertainties include, but are not limited to, risks related to
regulatory approvals and the success of Nemaura’s ongoing studies,
including the safety and efficacy of Nemaura’s sugarBEAT® system,
the failure of future development and preliminary marketing
efforts, Nemaura’s ability to secure additional commercial
partnering arrangements, risks and uncertainties relating to
Nemaura and its partners’ ability to develop, market and sell
sugarBEAT®, the availability of substantial additional equity or
debt capital to support its research, development and product
commercialization activities, and the success of its research,
development, regulatory approval, marketing and distribution plans
and strategies, including those plans and strategies related to
sugarBEAT®. These and other risks and uncertainties are identified
and described in more detail in Nemaura’s filings with the United
States Securities and Exchange Commission, including, without
limitation, its Annual Report on Form 10-K for the most recently
completed fiscal year, its Quarterly Reports on Form 10-Q, and its
Current Reports on Form 8-K. Nemaura undertakes no obligation to
publicly update or revise any forward-looking statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200225005660/en/
Dave Gentry, CEO RedChip Companies Office: 1.800.RED.CHIP
(733.2447) Cell: 407.491.4498 dave@redchip.com
Nemaura Medical (NASDAQ:NMRD)
Historical Stock Chart
From Mar 2024 to Apr 2024
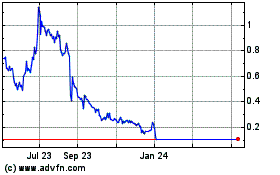
Nemaura Medical (NASDAQ:NMRD)
Historical Stock Chart
From Apr 2023 to Apr 2024