Genprex Strengthens Scientific Advisory Board with Appointment of Lead Researcher of University of Pittsburgh, George K. Gitt...
November 10 2020 - 8:17AM
Business Wire
Inventor of the Company’s licensed diabetes
gene therapy technology to provide leadership, strategy for
diabetes program
Dr. Gittes was recipient of a $2.59 million
grant from the National Institutes of Health (“NIH”) National
Institute of Diabetes and Digestive and Kidney Diseases for his
groundbreaking gene therapy research
Genprex, Inc. (“Genprex” or the “Company”) (NASDAQ: GNPX), a
clinical-stage gene therapy company focused on developing
life-changing therapies for patients with cancer and diabetes,
today announced the addition of George K. Gittes, MD to its
Scientific Advisory Board (SAB). Dr. Gittes is the inventor of the
Company’s licensed diabetes gene therapy technology that is
currently in development, and serves as the Chief of Pediatric
Surgery and Surgeon-in-Chief Emeritus at the UPMC Children’s
Hospital of Pittsburgh.
In February 2020, Genprex signed an exclusive license agreement
with the University of Pittsburgh for Dr. Gittes’ diabetes gene
therapy technology. Type 1 and Type 2 diabetes together affect
approximately 30.3 million people in the U.S., or about 9 percent
of the U.S. population. By replacing or repairing damaged or
missing genes, gene therapy offers a potential cure for patients
suffering from diabetes, as opposed to merely treating
symptoms.
As a member of Genprex’s SAB, Dr. Gittes will leverage his
highly impressive expertise to provide strategic support and drive
strategy for the Company’s diabetes clinical development
program.
“We are delighted to have Dr. Gittes join our Scientific
Advisory Board as Genprex continues to advance its diabetes
program,” said Rodney Varner, Genprex’s President and Chief
Executive Officer. “It is imperative that we rely on the guidance
and direction of esteemed individuals with a wealth of expertise in
research and gene therapy, and I expect that Genprex will benefit
greatly from Dr. Gittes’ experience. He will play an instrumental
role in our efforts to bring new hope to patients who suffer from
the devastating complications of this chronic illness.”
In addition to his role as Chief of Pediatric Surgery and
Surgeon-in-Chief Emeritus at UPMC Children’s Hospital of
Pittsburgh, one of the busiest pediatric surgical programs in the
U.S., he was appointed Director of the Richard King Mellon
Foundation Institute for Pediatric Research and Co-Scientific
director at UPMC Children's Hospital in 2018. Prior to UPMC, Dr.
Gittes served as the Director of Surgical Research at Children’s
Mercy Hospital in Kansas City and held the Thomas M. Holder and
Keith W. Ashcraft Chair in Pediatric Surgical Research. During his
time in Kansas City, he also was elected to the position of
President of the Society of University of Surgeons.
He is an active member of numerous professional and scientific
society memberships, including the American Society of Clinical
Investigators, the American Surgical Association, the American
Diabetes Association and the Association of American Physicians.
His research has been published in several peer-reviewed scientific
publications, and he is the recipient of several research grants,
including a recent $2.59 million grant awarded by the National
Institutes of Health (NIH) National Institute of Diabetes and
Digestive and Kidney Diseases to assist his ongoing preclinical
research for important proof-of-principle non-human primate studies
of his diabetes gene therapy.
About Genprex, Inc.
Genprex, Inc. is a clinical-stage gene therapy company focused
on developing life-changing therapies for patients with cancer and
diabetes. Genprex’s technologies are designed to administer
disease-fighting genes to provide new therapies for large patient
populations with cancer and diabetes who currently have limited
treatment options. Genprex works with world-class institutions and
collaborators to develop drug candidates to further its pipeline of
gene therapies in order to provide novel treatment approaches. The
Company’s lead product candidate, REQORSA™ immunogene therapy drug
(quaratusugene ozeplasmid), is being evaluated as a treatment for
non-small cell lung cancer (NSCLC). REQORSA has a multimodal
mechanism of action that has been shown to interrupt cell signaling
pathways that cause replication and proliferation of cancer cells;
re-establish pathways for apoptosis, or programmed cell death, in
cancer cells; and modulate the immune response against cancer
cells. REQORSA has also been shown to block mechanisms that create
drug resistance. In January 2020, the U.S. Food and Drug
Administration granted Fast Track Designation for REQORSA for NSCLC
in combination therapy with osimertinib (AstraZeneca’s Tagrisso®)
for patients with EFGR mutations whose tumors progressed after
treatment with osimertinib alone. For more information, please
visit the Company’s web site at www.genprex.com or follow Genprex
on Twitter, Facebook and LinkedIn.
Forward-Looking Statements
Statements contained in this press release regarding matters
that are not historical facts are "forward-looking statements"
within the meaning of the Private Securities Litigation Reform Act
of 1995. Because such statements are subject to risks and
uncertainties, actual results may differ materially from those
expressed or implied by such forward-looking statements. Such
statements include, but are not limited to, statements regarding
the effect of Genprex’s product candidates, alone and in
combination with other therapies, on cancer and diabetes, regarding
potential, current and planned clinical trials, regarding the
Company’s future growth and financial status and regarding our
commercial partnerships and intellectual property licenses. Risks
that contribute to the uncertain nature of the forward-looking
statements include the presence and level of the effect of our
product candidates, alone and in combination with other therapies,
on cancer; the timing and success of our clinical trials and
planned clinical trials of REQORSA™, alone and in combination with
targeted therapies and/or immunotherapies, and whether our other
potential product candidates, including GPX-002, our gene therapy
in diabetes, advance into clinical trials; the success of our
strategic partnerships, including those relating to manufacturing
of our product candidates; the timing and success at all of
obtaining FDA approval of REQORSA and our other potential product
candidates including whether we receive or benefit from fast track
or similar regulatory designations; costs associated with
developing our product candidates, whether we identify and succeed
in acquiring other technologies and whether patents will ever be
issued under patent applications that are the subject of our
license agreements or otherwise. These and other risks and
uncertainties are described more fully under the caption “Risk
Factors” and elsewhere in our filings and reports with the United
States Securities and Exchange Commission. All forward-looking
statements contained in this press release speak only as of the
date on which they were made. We undertake no obligation to update
such statements to reflect events that occur or circumstances that
exist after the date on which they were made.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20201110005159/en/
Genprex, Inc. (877) 774-GNPX (4679) Investor
Relations GNPX Investor Relations (877) 774-GNPX (4679) ext. #2
investors@genprex.com Media Contact Genprex Media Relations
Kalyn Dabbs (877) 774-GNPX (4679) ext. #3 media@genprex.com
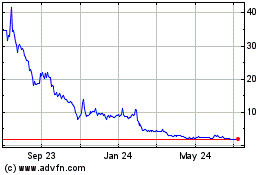
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Mar 2024 to Apr 2024
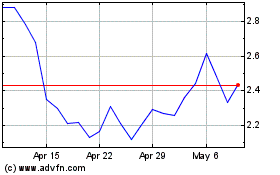
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Apr 2023 to Apr 2024