Genprex Begins Next Phase of Drug Branding Program
July 31 2019 - 8:30AM
Business Wire
Company submits non-proprietary drug name
selections to United States Adopted Names Council
Genprex, Inc. (NASDAQ: GNPX), a clinical-stage gene therapy
company, today announced it has initiated the first phase of
branding its lead drug candidate, Oncoprex™ immunogene therapy, and
has completed the creation and submission of non-proprietary drug
name selections to the American Medical Association’s United States
Adopted Names (USAN) Council.
Upon receiving feedback from the USAN Council, which is expected
later this year, Genprex will submit its non-proprietary drug name
selections to the World Health Organization for International
Nonproprietary Names (INN) status. The company expects to obtain an
approved non-proprietary drug name by mid-year 2020.
“Developing and obtaining regulatory approval of a
non-proprietary drug name is a requirement for bringing our lead
drug candidate to market,” said Rodney Varner, Chairman and Chief
Executive Officer of Genprex. “We’re one step closer to achieving
this regulatory milestone. An approved non-proprietary drug name
will also make our lead drug candidate more widely recognizable to
clinicians and physicians and most importantly, to patients living
with non-small cell lung cancer.”
The development of non-proprietary and proprietary drug names is
based on regulatory requirements by several governing bodies,
including the American Medical Association’s USAN Council, the
World Health Organization’s INN Programme, the European Medicines
Agency (EMA) and the U.S. Food and Drug Administration (FDA).
Non-proprietary drug names are typically designated based on the
active ingredient or molecular composition of the drug. Specific to
gene therapy, the nomenclature for products produced by insertion
of genetic material into a vector where altered genetic material is
administered to patients as a biologic drug is based on the drug
candidate’s gene composition and the vector used to deliver the
gene, thus requiring a two-word nomenclature scheme.
Upon creation and submission of the non-proprietary name
selections, each governing body reviews the drug name submissions
based on several criteria and guidelines, including similarity or
conflicts with existing drug names and trademarks, prefixes that
imply certain meanings or chemical elements, specific letter
combinations and usage, and international language pronunciation
considerations among other requirements.
About Genprex, Inc.
Genprex, Inc. is a clinical stage gene therapy company
developing potentially life-changing technologies for cancer
patients, based upon a unique proprietary technology platform,
including Genprex’s initial product candidate, Oncoprex™ immunogene
therapy for non-small cell lung cancer (NSCLC). Genprex’s platform
technologies are designed to administer cancer fighting genes by
encapsulating them into nanoscale hollow spheres called
nanovesicles, which are then administered intravenously and taken
up by tumor cells where they express proteins that are missing or
found in low quantities. Oncoprex has a multimodal mechanism of
action whereby it interrupts cell signaling pathways that cause
replication and proliferation of cancer cells, re-establishes
pathways for apoptosis, or programmed cell death, in cancer cells,
and modulates the immune response against cancer cells. Oncoprex
has also been shown to block mechanisms that create drug
resistance. For more information, please visit the company’s web
site at www.genprex.com or follow Genprex on Twitter, Facebook and
LinkedIn.
Forward-Looking Statements
Statements contained in this press release regarding matters
that are not historical facts are “forward-looking statements”
within the meaning of the Private Securities Litigation Reform Act
of 1995. Because such statements are subject to risks and
uncertainties, actual results may differ materially from those
expressed or implied by such forward-looking statements. Such
statements include, but are not limited to, statements regarding
the effects of Oncoprex on cancer, and statements regarding
Genprex’s anticipated receipt of a non-proprietary drug name,
including the timing of receipt of such a name. Risks that
contribute to the uncertain nature of the forward-looking
statements include risks relating to the effects of Oncoprex, alone
and in combination with immunotherapies, and risks relating to the
criteria and timing of granting non-proprietary names for drug
candidates. Other risks and uncertainties associated with Genprex
and its lead product candidate Oncoprex are described more fully
under the caption “Risk Factors” and elsewhere in our filings and
reports with the United States Securities and Exchange Commission.
All forward-looking statements contained in this press release
speak only as of the date on which they were made. We undertake no
obligation to update such statements to reflect events that occur
or circumstances that exist after the date on which they were
made.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190731005084/en/
Genprex, Inc. (877) 774-GNPX (4679)
Investor Relations GNPX Investor Relations (877) 774-GNPX
(4679) ext. #2 investors@genprex.com
Media Contact Genprex Media Relations Kalyn Dabbs (877)
774-GNPX (4679) ext. #3 media@genprex.com
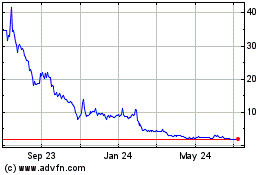
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Mar 2024 to Apr 2024
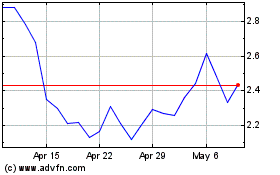
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Apr 2023 to Apr 2024