Alnylam Announces Recipients of Second Annual Advocacy for Impact Grants Program
June 02 2020 - 8:00AM
Business Wire
− Program Recognizes Seven Patient Advocacy
Groups Addressing Unmet Needs Among Three Rare Disease Communities
−
Alnylam Pharmaceuticals, Inc. (Nasdaq: ALNY), the leading RNAi
therapeutics company, announced today that seven patient advocacy
groups will receive supportive funding as part of the company’s
second annual Advocacy for Impact grants program. The global
competitive grants program aims to inspire patient advocacy groups
to develop solutions that address critical unmet needs among the
hereditary transthyretin-mediated (hATTR) amyloidosis, acute
hepatic porphyria (AHP) and primary hyperoxaluria type 1 (PH1) rare
disease communities.
In its inaugural year, Advocacy for Impact recognized the
efforts of seven patient advocacy groups around the world,
providing grants totaling nearly $250,000.00 to fund new
initiatives in support of hATTR amyloidosis and AHP. This year,
seven patient advocacy groups in five countries across three
continents will receive grants totaling $270,000.00. At their core,
these initiatives are designed to increase disease awareness and
access to diagnosis, offer education to patients, caregivers and
healthcare providers, and improve patient care.
“We are pleased to demonstrate our commitment to supporting
patient communities through the Advocacy for Impact grants program
for the second year in a row,” said Tiffany Patrick, Head of Global
Patient Advocacy and Engagement at Alnylam. “Those impacted by rare
diseases often face unique challenges due to the complexity of
their conditions. Through Advocacy for Impact, we hope to inspire
innovative thinking that brings high-impact initiatives to diverse
communities and geographies, and ultimately, improves the lives of
rare disease patients.”
2019-2020 Advocacy for Impact grant recipients include:
- Balearic Association for Andrade Disease (Asociación Balear
de la Enfermedad de Andrade [ABEA]), Spain – ABEA will develop
a hATTR amyloidosis learning program using digital training
capsules to raise awareness of the condition and provide education
on disease management.
- American Porphyria Foundation (APF), USA – The APF will
develop an engaging educational tool utilizing the grand rounds
format, “Porphyria Grand Rounds,” to educate healthcare
professionals about AHP and shine a spotlight on the patient
experience.
- Amyloidosis Alliance, France – The alliance will create
a comprehensive toolbox, inclusive of digital platforms, videos,
webinars, templates and documents, for use by new or developing
patient groups to support their structural development, ultimately
helping to establish a strong international network of hATTR
amyloidosis organizations.
- Amyloidosis New Zealand Trust, New Zealand – The trust
will organize and host a conference open to healthcare
professionals and patients, so that the healthcare community can
hear directly from patients to better understand the
multidisciplinary nature of hATTR amyloidosis; findings will help
to establish a National Amyloidosis Centre for New Zealand.
- French Association in the Fight Against Amyloidosis
(Association Française contre l'Amylose), France – The
association will work closely with healthcare professionals to
develop a set of educational tools aimed at optimizing disease
management and patient care.
- The Oxalosis & Hyperoxaluria Foundation (OHF), USA –
The OHF will partner with hospitals to create Centers of Excellence
that will increase access to optimal multidisciplinary clinical
care and services for those impacted by all forms of hyperoxaluria,
including PH1.
- UK ATTR Amyloidosis Patients' Association (UKATPA), UK –
UKATPA will organize informational sessions for patients and their
families in up to five different cities outside of London to
provide disease education, and create social opportunities for
those in the community to meet one another and develop supportive
local relationships.
The 2019-2020 Advocacy for Impact grants program was open to
patient advocacy groups around the world requesting funding for up
to $50,000 for new projects focused on supporting the hATTR
amyloidosis, AHP and/or PH1 communities in one of the following
ways:
- Increase disease awareness and access to diagnosis
- Offer education to patients, families, caregivers, healthcare
providers and/or public
- Improve patient care
Applications were reviewed by a committee comprised of internal
experts from Alnylam and external experts with experience in
nonprofits and rare disease. Grant recipients were determined based
on a clear identification of an unmet need, an effective plan of
execution, the level of expected impact within the target rare
disease community and a strong strategy to measure success. In
light of the COVID-19 pandemic, initiatives that include in-person
events and activities are subject to change.
For additional information about the Advocacy for Impact grants
program, please visit our website.
About Alnylam Pharmaceuticals
Alnylam (Nasdaq: ALNY) is leading the translation of RNA
interference (RNAi) into a whole new class of innovative medicines
with the potential to transform the lives of people afflicted with
rare genetic, cardio-metabolic, hepatic infectious, and central
nervous system (CNS)/ocular diseases. Based on Nobel Prize-winning
science, RNAi therapeutics represent a powerful, clinically
validated approach for the treatment of a wide range of severe and
debilitating diseases. Founded in 2002, Alnylam is delivering on a
bold vision to turn scientific possibility into reality, with a
robust RNAi therapeutics platform. Alnylam’s commercial RNAi
therapeutic products are ONPATTRO® (patisiran), approved in the
U.S., EU, Canada, Japan, Brazil, and Switzerland, and GIVLAARI®
(givosiran), approved in the U.S and the EU. Alnylam has a deep
pipeline of investigational medicines, including six product
candidates that are in late-stage development. Alnylam is executing
on its “Alnylam 2020” strategy of building a multi-product,
commercial-stage biopharmaceutical company with a sustainable
pipeline of RNAi-based medicines to address the needs of patients
who have limited or inadequate treatment options. Alnylam is
headquartered in Cambridge, MA.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200602005193/en/
Alnylam Pharmaceuticals, Inc. Christine Regan Lindenboom
(Investors and Media)
617-682-4340
Joshua Brodsky
(Investors) 617-551-8276
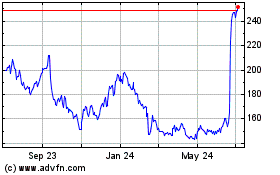
Alnylam Pharmaceuticals (NASDAQ:ALNY)
Historical Stock Chart
From Mar 2024 to Apr 2024
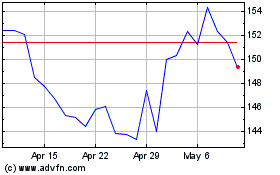
Alnylam Pharmaceuticals (NASDAQ:ALNY)
Historical Stock Chart
From Apr 2023 to Apr 2024