Filed
Pursuant to Rule 424(b)(5)
Registration
No. 333-226988
PROSPECTUS
SUPPLEMENT
(to
Prospectus dated August 30, 2018)
Up
to $20,000,000 of Common Stock
1,084,266
Shares of Common Stock
This
prospectus supplement relates to the issuance and sale of up to $20,000,000 of shares of our common stock, par value $0.001 per
share (“common stock”), that we may issue to Lincoln Park Capital Fund, LLC (“Lincoln Park”) from time
to time under a Purchase Agreement that we entered into with Lincoln Park on July 30, 2020 (the “Purchase Agreement”),
and an additional 1,084,266 shares of common stock that are being issued to Lincoln Park as a commitment fee for entering into
the Purchase Agreement.
The
shares offered include:
|
|
●
|
up
to $20,000,000 of shares of common stock we may sell to Lincoln Park from time to time, at our sole discretion, in accordance
with the Purchase Agreement; and
|
|
|
|
|
|
|
●
|
1,084,266
shares of common stock being issued to Lincoln Park as commitment shares in consideration for entering into the Purchase Agreement,
valued at $0.60 per share. We will not receive any cash proceeds from the issuance of these commitment shares.
|
This
prospectus supplement and the accompanying prospectus also cover the resale of these shares by Lincoln Park to the public.
Our
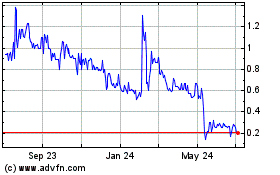
common stock is listed on the Nasdaq Global Select Market under the symbol “ADXS.” The last reported sale price of
our common stock on July 29, 2020 was $0.60 per share.
Investing
in our common stock involves a high degree of risk. See “Risk Factors” beginning on page S-6 of this prospectus supplement
and in the documents that are incorporated by reference into this prospectus supplement and the accompanying prospectus.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or
passed upon the adequacy or accuracy of this prospectus supplement and the accompanying prospectus. Any representation to the
contrary is a criminal offense.
The
date of this prospectus supplement is July 30, 2020
Table
of Contents
ABOUT
THIS PROSPECTUS SUPPLEMENT
This
prospectus supplement and the accompanying prospectus relate to the offering of our common stock. You should read this prospectus
supplement, the accompanying prospectus, the documents incorporated by reference into this prospectus supplement and the accompanying
prospectus, and any free writing prospectus that we may authorize for use in connection with this offering in their entirety before
making an investment decision. You should also read and consider the information in the documents to which we have referred you
in the section of this prospectus supplement entitled “Where You Can Find More Information” and “Incorporation
by Reference.” These documents contain important information that you should consider when making your investment decision.
This
document is in two parts. The first part is this prospectus supplement, which describes the specific terms of this offering and
certain other matters and also adds to and updates information contained in the accompanying prospectus and the documents incorporated
by reference herein or therein. The second part, the accompanying prospectus, including the documents incorporated by reference
into the accompanying prospectus, provides more general information. Generally, when we refer to this prospectus, we are referring
to the combined document consisting of this prospectus supplement and the accompanying prospectus. To the extent there is a conflict
between the information contained in this prospectus supplement and the information contained in the accompanying prospectus or
any document incorporated by reference herein or therein that was filed with the Securities and Exchange Commission (the “SEC”)
before the date of this prospectus supplement, you should rely on the information in this prospectus supplement; provided that
if any statement in one of these documents is inconsistent with a statement in another document having a later date — for
example, a document incorporated by reference in the accompanying prospectus —the statement in the document having the later
date modifies or supersedes the earlier statement.
Unless
otherwise stated, all references in this prospectus supplement to “we,” “us,” “our,” “Advaxis,”
the “Company” and similar designations refer to Advaxis, Inc. This prospectus supplement, the accompanying prospectus
and the information incorporated by reference herein and therein contain trademarks, service marks and trade names of Advaxis,
Inc., including our name and logo. Other trademarks, service marks and trade names referred to in this prospectus supplement or
the accompanying prospectus or the information incorporated by reference herein and therein are the property of their respective
owners.
We
are not making offers to sell or solicitations to buy our common stock in any jurisdiction in which an offer or solicitation is
not authorized or in which the person making that offer or solicitation is not qualified to do so or to anyone to whom it is unlawful
to make an offer or solicitation. You should assume that the information in this prospectus supplement, the accompanying prospectus
or any related free writing prospectus is accurate only as of the date on the front of the document and that any information that
we have incorporated by reference is accurate only as of the date of the document incorporated by reference, regardless of the
time of delivery of this prospectus supplement or any related free writing prospectus, or any sale of a security.
CAUTIONARY
NOTE REGARDING FORWARD LOOKING STATEMENTS
This
prospectus supplement, the accompanying prospectus and the other documents we have filed with the SEC that are incorporated herein
by reference contain forward-looking statements that involve substantial risks and uncertainties. Any statements, other than statements
of historical facts, contained in this prospectus supplement, the accompanying prospectus or the information incorporated by reference
herein or therein may be deemed to be forward-looking statements. In some cases, these forward-looking statements can be identified
by the use of forward-looking terminology, including the terms “believes,” “estimates,” “anticipates,”
“expects,” “plans,” “intends,” “may,” “could,” “might,”
“will,” “should,” “approximately” or, in each case, their negative or other variations thereon
or comparable terminology, although not all forward-looking statements contain these words. Such forward-looking statements include
statements about our intentions, beliefs, projections, outlook, analyses or current expectations concerning, among other things,
our ongoing and planned discovery and development of drug candidates, the strength and breadth of our intellectual property, our
ongoing and planned preclinical studies and clinical trials, the timing of and our ability to make regulatory filings and obtain
and maintain regulatory approvals for our product candidates, the degree of clinical utility of our product candidates, particularly
in specific patient populations, expectations regarding clinical trial data, our results of operations, financial condition, liquidity,
prospects, growth and strategies, the length of time that we will be able to continue to fund our operating expenses and capital
expenditures, our expected financing needs and sources of financing, the industry in which we operate and the trends that may
affect the industry or us.
By
their nature, forward-looking statements involve risks and uncertainties because they relate to events, competitive dynamics,
and healthcare, regulatory and scientific developments and depend on the economic circumstances that may or may not occur in the
future or may occur on longer or shorter timelines than anticipated. Although we believe that we have a reasonable basis for each
forward-looking statement contained in this prospectus supplement, we caution you that forward-looking statements are not guarantees
of future performance and they may not be predictive of results or developments in future periods.
Forward-looking
statements involve risks and uncertainties that could cause actual results or outcomes to differ materially from those expressed
therein. We express our estimates, expectations, beliefs, and projections in good faith and believe them to have a reasonable
basis. However, we make no assurances that management’s estimates, expectations, beliefs, or projections will be achieved
or accomplished. We have identified the following important factors that could cause actual results to differ materially from
those discussed in our forward-looking statements. These may be in addition to other factors and matters discussed in “Risk
Factors,” beginning on page S-6 of this prospectus; the sections entitled “Risk Factors” and Management’s
Discussion and Analysis of Financial Condition and Results of our Annual Report on Form 10-K for the fiscal year ended October
31, 2019 (as filed with the SEC on December 31, 2019, and amended by Amendment No. 1 thereto on Form 10-K/A filed on January 21,
2020 and by Amendment No. 2 thereto on Form 10-K/A filed on February 28, 2020, as so amended the “2019 Form 10-K/A”),
and as updated from time to time in the Company’s filings with the SEC. These factors include:
|
|
●
|
the
success and timing of our clinical trials, including patient accrual;
|
|
|
●
|
our
ability to obtain and maintain regulatory approval or reimbursement of our product candidates for marketing;
|
|
|
●
|
our
ability to obtain the appropriate labeling of our products under any regulatory approval;
|
|
|
●
|
potential
effects of the coronavirus (COVID-19) pandemic on our business, financial condition, liquidity and results of operations,
and our ability to continue operations in the same manner as previously conducted prior to the macroeconomic effects of the
COVID-19 pandemic;
|
|
|
●
|
our
ability to develop and commercialize our products;
|
|
|
●
|
the
successful development and implementation of our sales and marketing campaigns;
|
|
|
●
|
a
change of key scientific or management personnel;
|
|
|
●
|
the
size and growth of the potential markets for our product candidates and our ability to serve those markets;
|
|
|
●
|
our
ability to successfully compete in the potential markets for our product candidates, if commercialized;
|
|
|
●
|
regulatory
developments in the United States and other countries;
|
|
|
●
|
the
rate and degree of market acceptance of any of our product candidates;
|
|
|
●
|
new
products, product candidates or new uses for existing products or technologies introduced or announced by our competitors
and the timing of these introductions or announcements;
|
|
|
●
|
market
conditions in the pharmaceutical and biotechnology sectors;
|
|
|
●
|
our
available cash;
|
|
|
●
|
the
accuracy of our estimates regarding expenses, future revenues, capital requirements and needs for additional financing;
|
|
|
●
|
our
ability to obtain additional funding;
|
|
|
●
|
our
ability to obtain and maintain intellectual property protection for our product candidates;
|
|
|
●
|
the
success and timing of our preclinical studies, including investigational new drug (“IND”) enabling studies;
|
|
|
●
|
the
ability of our product candidates to successfully perform in clinical trials and to resolve any clinical holds that may occur;
|
|
|
●
|
our
ability to obtain and maintain approval of our product candidates for trial initiation;
|
|
|
●
|
our
ability to manufacture and the performance of third-party manufacturers;
|
|
|
●
|
our
ability to identify license and collaboration partners and to maintain existing relationships;
|
|
|
●
|
the
performance of our clinical research organizations, clinical trial sponsors, clinical trial investigators and collaboration
partners for any clinical trials we conduct;
|
|
|
●
|
our
ability to successfully implement our strategy; and
|
|
|
●
|
our
ability to maintain the listing of our common stock on the Nasdaq Global Select Market.
|
This
prospectus supplement includes statistical and other industry and market data that we obtained from industry publications and
research, surveys and studies conducted by third-parties. Industry publications and third-party research, surveys and studies
generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee
the accuracy or completeness of such information. While we believe these industry publications and third-party research, surveys
and studies are reliable, we have not independently verified such data.
PROSPECTUS
SUPPLEMENT SUMMARY
This
summary highlights information contained elsewhere or incorporated by reference in this prospectus supplement and the accompanying
prospectus and in the documents we incorporate by reference. This summary does not contain all of the information that you should
consider before deciding to invest in our common stock. You should read this entire prospectus supplement and the accompanying
prospectus carefully, including the “Risk Factors’’ section contained in this prospectus supplement and
our consolidated financial statements and the related notes and the other documents incorporated by reference herein, as well
as the information included in any free writing prospectus that we have authorized for use in connection with this offering.
Our
Business
We
are a clinical-stage biotechnology company focused on the development and commercialization of proprietary Listeria monocytogenes
(“Lm”)-based antigen delivery products. We are using our Lm platform directed against tumor-specific targets in order
to engage the patient’s immune system to destroy tumor cells. Through a license from the University of Pennsylvania, we
have exclusive access to this proprietary formulation of attenuated Lm called Lm TechnologyTM. Our proprietary approach
is designed to deploy a unique mechanism of action that redirects the immune system to attack cancer in three distinct ways:
|
|
●
|
Alerting
and training the immune system by activating multiple pathways in Antigen-Presenting Cells with the equivalent of multiple
adjuvants;
|
|
|
●
|
Attacking
the tumor by generating a strong, cancer-specific T cell response; and
|
|
|
●
|
Breaking
down tumor protection through suppression of the protective cells in the tumor microenvironment that shields the tumor from
the immune system. This enables the activated T cells to begin working to attack the tumor cells.
|
Our
proprietary Lm platform technology has demonstrated clinical activity in several of our programs and has been dosed in over 470
patients across multiple clinical trials and in various tumor types. We believe that Lm Technology immunotherapies can complement
and address significant unmet needs in the current oncology treatment landscape. Specifically, our product candidates have the
potential to work synergistically with other immunotherapies, including checkpoint inhibitors, while having a generally well-tolerated
safety profile.
Recent
Developments
COVID-19
Recent Developments
We
are monitoring the novel coronavirus (“COVID-19”) pandemic and are taking steps intended to mitigate the potential
risks to us posed by COVID-19. This is a rapidly evolving situation, which has disrupted economic activity as well as the capital
markets, and these disruptions could continue for a prolonged period of time or become more severe. We continue to assess the
situation and the potential impact it will have on our business, we believe it is likely that we may encounter a slowdown in recruiting
patients for our studies. We cannot predict the extent or duration of the pandemic or its effects on the global, national, or
local economy, the capital markets, or our suppliers, operations, including our clinical trials, or our financial results. We
are continuing to monitor developments with respect to the pandemic and its effects and intend to take additional measures as
we believe are warranted, including for the safety of our patients and employees.
Strategic
Transactions
As
a matter of course, we are reviewing strategic transactions and alternatives and there can be no assurance that we will be successful
in identifying or completing any strategic transactions, that any such strategic transaction will result in additional value for
our stockholders or that the process will not have an adverse impact on our business. These transactions could include, but are
not limited to, collaboration agreements, co-development agreements, strategic mergers, reverse mergers, the issuance or buyback
of public shares, or the purchase or sale of specific assets, in addition to other potential actions aimed at increasing stockholder
value. There can be no assurance that the review of strategic transactions will result in the identification or consummation of
any transaction. Our Board of Directors may also determine that our most effective strategy is to continue to effectuate our current
business plan. Any potential transaction would be dependent upon a number of factors that may be beyond our control, including,
but not limited to, market conditions, industry trends, the interest of third parties in our business and the availability of
financing to potential buyers on reasonable terms. No decision has been made with respect to any transaction.
Company
Information
We
were originally incorporated in the State of Colorado on June 5, 1987 under the name Great Expectations, Inc. We were a publicly-traded
“shell” company without any business until November 12, 2004 when we acquired Advaxis, Inc., a Delaware corporation,
through a Share Exchange and Reorganization Agreement, dated as of August 25, 2004, which we refer to as the Share Exchange, by
and among Advaxis, the stockholders of Advaxis and us. As a result of the Share Exchange, Advaxis became our wholly-owned subsidiary
and our sole operating company. On December 23, 2004, we amended and restated our articles of incorporation and changed our name
to Advaxis, Inc. On June 6, 2006, our stockholders approved the reincorporation of our company from Colorado to Delaware by merging
the Colorado entity into our wholly-owned Delaware subsidiary. Our date of inception, for financial statement purposes, is March
1, 2002 and we were uplisted to Nasdaq in 2014.
Our
principal executive offices and manufacturing facility are located at 305 College Road East, Princeton, New Jersey 08540 and our
telephone number is (609) 452-9813. We maintain a corporate website at www.advaxis.com which contains descriptions of our technology,
our product candidates and the development status of each drug. We make available free of charge through our Internet website
our annual reports on Form 10-K, quarterly reports on Form 10-Q and current reports on Form 8-K, and any amendments to these reports,
as soon as reasonably practicable after we electronically file such material with, or furnish such material to, the SEC. We are
not including the information on our website as a part of, nor incorporating it by reference into, this prospectus. The SEC maintains
a website that contains annual, quarterly, and current reports, proxy statements, and other information that issuers (including
us) file electronically with the SEC. The SEC’s website address is www.sec.gov.
The
Offering
|
Common
stock offered by us (1)
|
|
●
|
Up
to $20,000,000 of shares of common stock we may sell to Lincoln Park from time to time, at our sole discretion, in accordance
with the Purchase Agreement; and
|
|
|
|
|
|
|
|
|
●
|
1,084,266
shares of common stock being issued to Lincoln Park as commitment shares in consideration for entering into the Purchase Agreement,
valued at $0.60 per share. We will not receive any cash proceeds from the issuance of these commitment shares.
|
|
|
|
|
|
|
Shares
of common stock to be outstanding immediately after this offering (2)
|
|
Up
to 73,336,710 shares of common stock, assuming an average sales price of $0.60 per share (the last reported sales price of
our common stock on the Nasdaq Global Select Market on July 29, 2020) for the $20,000,000 of shares of common stock
we may sell to Lincoln Park from time to time, including 1,084,266 shares being issued to Lincoln Park as commitment shares
in consideration for entering into the Purchase Agreement. The actual number of shares issued and outstanding will vary depending
on the sale prices of shares sold to Lincoln Park in this offering.
|
|
|
|
|
|
|
Use
of proceeds
|
|
We
intend to use the net proceeds from this offering to advance our ADXS-HOT program and for general corporate purposes.
See
“Use of Proceeds” on page S-10 of this prospectus supplement for a more complete description of the intended
use of proceeds from this offering.
|
|
|
|
|
|
|
Risk
factors
|
|
Investing
in our common stock involves significant risks. Please read the information contained in or incorporated by reference under
the heading “Risk Factors” beginning on page S-6 of this prospectus supplement, and under similar headings in
documents that are filed after the date hereof and incorporated by reference into this prospectus supplement and the accompanying
prospectus.
|
|
|
|
|
|
|
Nasdaq
Global Select Market symbol
|
|
“ADXS.”
|
|
(1)
|
The
Purchase Agreement prohibits us from issuing or selling to Lincoln Park under the Purchase Agreement (i) in excess of 12,326,314
shares of our common stock (the “Exchange Cap”), unless we obtain stockholder approval to issue shares in
excess of the Exchange Cap or the average price of all applicable sales of our common stock to Lincoln Park under the Purchase
Agreement equal or exceed the lower of (i) the Nasdaq Official Closing Price immediately preceding the execution of this Agreement
or (ii) the average of the five Nasdaq Official Closing Prices for the common stock immediately preceding the execution
of this Agreement, as adjusted in accordance with the rules of the Principal Market, and (ii) any shares of our common stock
if those shares, when aggregated with all other shares of our common stock then beneficially owned by Lincoln Park and its
affiliates would result in Lincoln Park and its affiliates having beneficial ownership of more than 9.99% of the then total
outstanding shares of our common stock (the “Beneficial Ownership Cap”).
|
|
(2)
|
The
number of shares of common stock to be outstanding immediately after this offering is based on 60,252,444 shares of common
stock outstanding as of April 30, 2020. The number of shares outstanding as of April 30, 2020 excludes:
|
|
|
●
|
5,398,226
shares of our common stock reserved for issuance upon the exercise of outstanding warrants at a weighted average exercise
price of $1.16 per share;
|
|
|
|
|
|
|
●
|
5,818
shares of our common stock reserved for issuance upon settlement of restricted stock units;
|
|
|
|
|
|
|
●
|
252,296
shares of our common stock reserved for issuance upon the
exercise of outstanding stock options at a weighted average exercise price of $73.50 per share; and
|
|
|
|
|
|
|
●
|
210,787
shares of our common stock reserved for future awards under our 2015 Incentive Plan.
|
Except
as otherwise indicated, all information in this prospectus supplement assumes no exercise or forfeiture of the outstanding options
or remaining warrants or settlement of restricted stock units after April 30, 2020. In addition, on May 4, 2020, we amended our
2015 Incentive Plan to increase the number of shares available for issuance pursuant to awards granted thereunder from 877,744
shares to 6,000,000 shares of common stock.
Agreement
with Lincoln Park Capital Fund, LLC
On
July 30, 2020, we entered into a Purchase Agreement with Lincoln Park, pursuant to which, upon the terms and subject to the conditions
and limitations set forth therein, we have the right to sell to Lincoln Park up to $20,000,000 of shares of our common stock at
our discretion as described below.
As
consideration for entering into the Purchase Agreement, we issued 1,084,266 shares of our common stock to Lincoln Park as a commitment
fee (the “Commitment Shares”). We will not receive any cash proceeds from the issuance of the Commitment Shares.
We
are filing this prospectus supplement to cover the offer and sale of (i) up to $20,000,000 of shares of our common stock, subject
to the conditions and limitations in the Purchase Agreement, and (ii) the 1,084,266 Commitment Shares.
Over
the 36-month term of the Purchase Agreement, up to an aggregate amount of $20,000,000 (subject to certain limitations) of shares
of common stock, we have the right, but not the obligation, from time to time, in our sole discretion and subject to certain
conditions, including that the closing price of our common stock is not below $0.10 per share, to direct Lincoln Park to
purchase up to 1,000,000 shares (the “Regular Purchase Share Limit”) of our common stock (each such
purchase, a “Regular Purchase”). Lincoln Park’s maximum obligation under any single Regular Purchase will not
exceed $1,000,000, unless we mutually agree to increase the maximum amount of such Regular Purchase. The purchase price for shares
of common stock to be purchased by Lincoln Park under a Regular Purchase will be the equal to the lower of (in each case,
subject to the adjustments described in the Purchase Agreement): (i) the lowest sale price for our common stock on the
applicable purchase date, and (ii) the arithmetic average of the three lowest sale prices for our common stock during the
ten trading days prior to the purchase date.
Pursuant
to the Purchase Agreement, Lincoln Park has agreed to an initial purchase of 3,510,527 shares of our common stock for an aggregate
purchase price of $1,999,999.89.
If
we direct Lincoln Park to purchase the maximum number of shares of common stock we then may sell in a Regular Purchase,
then in addition to such Regular Purchase, and subject to certain conditions and limitations in the Purchase Agreement, we may
direct Lincoln Park to make an “accelerated purchase” of an additional amount of common stock that may not
exceed the lesser of (i) 300% of the number of shares purchased pursuant to the corresponding Regular Purchase and (ii) 30% of
the total number of shares of our common stock traded during a specified period on the applicable purchase date as set
forth in the Purchase Agreement. Under certain circumstances and in accordance with the Purchase Agreement, the Company may direct
Lincoln Park to purchase shares in multiple accelerated purchases on the same trading day.
The
Purchase Agreement does not limit our ability to raise capital from other sources at our sole discretion, except that we may not
enter into any equity line or similar transaction for 36 months.
During
an event of default as set forth in the Purchase Agreement, all of which are outside the control of Lincoln Park, shares of our
common stock cannot be sold by us or purchased by Lincoln Park under the terms of the Purchase Agreement.”
We may at any time, in our sole discretion, terminate the Purchase Agreement without fee, penalty or cost, upon one trading day
written notice. In the event of bankruptcy proceedings by or against us, the Purchase Agreement will automatically terminate without
action of any party.
The
above description of the Purchase Agreement is qualified in its entirety by reference to the Purchase Agreement, which will be
filed with the SEC and incorporated by reference into this prospectus supplement.
Amount
of Potential Proceeds to be Received under the Purchase Agreement
Under
the Purchase Agreement, we may sell shares of common stock having an aggregate offering price of up to $20,000,000 to Lincoln
Park from time to time. The number of shares ultimately offered for sale to Lincoln Park in this offering is dependent upon the
number of shares we elect to sell to Lincoln Park under the Purchase Agreement. The following table sets forth the amount of proceeds
we would receive from Lincoln Park from the sale of shares at varying purchase prices:
|
Assumed Average Purchase
Price
|
|
|
Number of Shares to be
Issued in this Offering at the Assumed Average Purchase Price(1)(2)
|
|
|
Percentage of Outstanding
Shares After Giving Effect to the Additional Purchased Shares Issued to Lincoln Park(3)
|
|
|
Proceeds from the Sale
of Shares Under the Purchase Agreement Registered in this Offering
|
|
|
$
|
0.40
|
|
|
|
11,242,048
|
|
|
|
15.2
|
%
|
|
$
|
4,496,819
|
|
|
$
|
0.60
|
(4)
|
|
|
11,242,048
|
|
|
|
15.2
|
%
|
|
$
|
6,745,229
|
|
|
$
|
0.80
|
|
|
|
25,000,000
|
|
|
|
28.5
|
%
|
|
$
|
20,000,000
|
|
|
$
|
1.00
|
|
|
|
20,000,000
|
|
|
|
24.2
|
%
|
|
$
|
20,000,000
|
|
|
$
|
2.00
|
|
|
|
10,000,000
|
|
|
|
13.8
|
%
|
|
$
|
20,000,000
|
|
(1)
Includes the total number of shares of common stock to be purchased under the Purchase Agreement (but not Commitment Shares) that
we would have sold under the Purchase Agreement at the corresponding assumed purchase price set forth in the adjacent column,
up to the aggregate purchase price of $20,000,000.
(2)
The Purchase Agreement prohibits us from issuing or selling to Lincoln Park under the Purchase Agreement (i) shares of our common
stock in excess of the Exchange Cap, unless we obtain stockholder approval to issue shares in excess of the Exchange Cap or the
average price of all applicable sales of our common stock to Lincoln Park under the Purchase Agreement equal or exceed the lower
of (i) the Nasdaq Official Closing Price immediately preceding the execution of this Agreement or (ii) the average of the five
Nasdaq Official Closing Prices for the common stock immediately preceding the execution of this Agreement, as adjusted in accordance
with the applicable rules of the Nasdaq Capital Market, and (ii) any shares of our common stock if those shares, when aggregated
with all other shares of our common stock then beneficially owned by Lincoln Park and its affiliates, would exceed the Beneficial
Ownership Cap.
(3)
The denominator is based on 61,631,579 shares outstanding as of July 29, 2020, the 1,084,266 shares being issued to Lincoln Park
and the number of shares set forth in the adjacent column that we would have sold to Lincoln Park. The numerator is based on the
additional number of shares which we may issue to Lincoln Park under the Purchase Agreement, which are the subject of this offering
at the corresponding assumed purchase price set forth in the adjacent column.
(4)
The closing sale price of the common stock on July 29, 2020.
RISK
FACTORS
Investment
in our common stock involves risks. Before deciding whether to invest in our common stock, you should consider carefully the risk
factors discussed below and those contained in the section entitled “Risk Factors” contained in our 2019 Form 10-K/A,
as well as any amendment or update to our risk factors reflected in our subsequent filings with the SEC. If any of the risks or
uncertainties described in our SEC filings actually occurs, our business, financial condition, results of operations or cash flow
could be materially and adversely affected. This could cause the trading price of our common stock to decline, resulting in a
loss of all or part of your investment. The risks and uncertainties we have described are not the only ones we face. Additional
risks and uncertainties not presently known to us or that we currently deem immaterial may also affect our business operations.
Risks
Related to this Offering
Our
stock price can be volatile, which increases the risk of litigation, and may result in a significant decline in the value of your
investment.
The
stock market in general and the market for biopharmaceutical companies in particular have experienced extreme volatility that
has often been unrelated to the operating performance of particular companies. As a result of this volatility, you may not be
able to sell your common stock at or above the price at which it was acquired. The market price for our common stock may be influenced
by many factors, including:
|
|
●
|
changes
in the market valuations, stock market prices and trading volumes of similar companies;
|
|
|
●
|
actual
or anticipated changes in our net loss or fluctuations in our operating results or in the expectations of securities analysts;
|
|
|
●
|
the
issuance of new equity securities pursuant to a future offering, including potential issuances of preferred stock;
|
|
|
●
|
sales
of large blocks of our stock;
|
|
|
●
|
failure
of our common stock or warrants to be listed or quoted on the Nasdaq Stock Market, NYSE Amex Equities or other national market
system;
|
|
|
●
|
additions
or departures of key personnel;
|
|
|
●
|
changes
in the regulatory status of our immunotherapies, including results of our pre-clinical and clinical trials;
|
|
|
●
|
regulatory
developments in the United States and other countries;
|
|
|
●
|
positive
and negative changes in relationships with partners;
|
|
|
●
|
announcements
of new products or technologies, commercial relationships or other events by us or our competitors;
|
|
|
●
|
changes
in accounting principles;
|
|
|
●
|
discussion
of us or our stock price by the financial and scientific press and in online investor communities;
|
|
|
●
|
market
conditions and events in the pharmaceutical and biotech sectors; and
|
|
|
●
|
general
economic, industry, and market conditions.
|
These
broad market and industry factors may materially affect the market price of our common stock, regardless of our development and
operating performance. In the past, following periods of volatility in the market price of a company’s securities, securities
class-action litigation has often been instituted against that company. Due to the volatility of our stock price, we have been
and may be the target of securities litigation in the future. Securities litigation could result in substantial costs and divert
management’s in the future attention and resources from our business.
You
may experience immediate and substantial dilution in the book value per share of the common stock you purchase in the offering.
Because
the prices per share of our common stock being offered may be higher than the book value per share of our common stock, you may
suffer immediate substantial dilution in the net tangible book value of the common stock if you purchase in this offering. See
the section entitled “Dilution” below for a more detailed discussion of the dilution you may incur if you purchase
common stock in this offering.
In
addition, in order to raise additional capital, we may in the future offer, issue or sell additional shares of common stock or
other securities convertible into or exchangeable for shares of our common stock. We cannot assure you that we will be able to
sell shares or other securities in any other transaction at a price per share or that have an exercise price or conversion price
per share that is equal to or greater than the prices for the common stock purchased by investors in this offering, and investors
purchasing shares or other securities in the future could have rights superior to existing stockholders. If we sell shares or
other securities in any future transaction at a price per share or that have an exercise price or conversion price per share that
is less than the price you pay for shares of common stock in this offering, you will experience dilution.
Moreover,
if outstanding warrants or options are exercised at prices per share that are less than the price you pay in this offering, or
shares of common stock issue upon the vesting of outstanding restricted stock units, or if we issue options or warrants to purchase,
or securities convertible into or exchangeable for, shares of our common stock in the future at a price per share that is less
than the price you pay in this offering and such options, warrants or other securities are exercised, converted or exchanged,
you will experience further dilution.
The
actual number of shares we will issue to Lincoln Park under the Purchase Agreement, at any one time or in total, is uncertain.
Subject
to certain limitations in the Purchase Agreement with Lincoln Park and compliance with applicable law, we have the discretion
to deliver purchase notices to Lincoln Park at any time throughout the term of the Purchase Agreement. The number of shares that
are bought by Lincoln Park after delivering a purchase notice will fluctuate based on the market price of the common stock during
the sales period and limits we set with Lincoln Park.
Future
sales or other issuances of our common stock could depress the market for our common stock.
Sales
of a substantial number of shares of our common stock, or the perception by the market that those sales could occur, could cause
the market price of our common stock to decline or could make it more difficult for us to raise funds through the sale of equity
in the future.
Future
issuances of common stock could further depress the market for our common stock. We expect to continue to incur drug development
and selling, general and administrative costs, and to satisfy our funding requirements, we will need to sell additional equity
securities, which may include sales of significant amounts of common stock to strategic investors, and which common stock may
be subject to registration rights and warrants with anti-dilutive protective provisions. The sale or the proposed sale of substantial
amounts of our common stock or other equity securities in the public markets or in private transactions may adversely affect the
market price of our common stock and our stock price may decline substantially. Our stockholders may experience substantial dilution
and a reduction in the price that they are able to obtain upon sale of their shares. Also, new equity securities issued may have
greater rights, preferences or privileges than our existing common stock. In addition, we have a significant number of shares
of restricted stock, restricted stock units, stock options and warrants outstanding. To the extent that outstanding stock options
or warrants have been or may be exercised or other shares issued, investors purchasing our common stock in this offering may experience
further dilution.
If
we make one or more significant acquisitions in which the consideration includes stock or other securities, our stockholders’
holdings may be significantly diluted. In addition, stockholders’ holdings may also be diluted if we enter into arrangements
with third parties permitting us to issue shares of common stock in lieu of certain cash payments upon the achievement of milestones.
We
do not intend to pay cash dividends.
We
have not declared or paid any cash dividends on our common stock, and we do not anticipate declaring or paying cash dividends
for the foreseeable future. Any future determination as to the payment of cash dividends on our common stock will be at our Board
of Directors’ discretion and will depend on our financial condition, operating results, capital requirements and other factors
that our Board of Directors considers to be relevant.
You
may experience future dilution as a result of future equity offerings.
To
raise additional capital, we may in the future offer additional shares of our common stock or other securities convertible into
or exchangeable for our common stock at prices that may not be the same as the prices per share in this offering. We may sell
shares or other securities in any other offering at a price per share that is less than the prices per share paid by investors
in this offering, and investors purchasing shares of our common stock or other securities in the future could have rights superior
to existing stockholders. The price per share at which we sell additional shares of our common stock, or securities convertible
or exchangeable into common stock, in future transactions may be higher or lower than the prices per share paid by investors in
this offering.
Our
management will have broad discretion to use the net proceeds from this offering and our investment of these proceeds pending
any such use may not yield a favorable return.
Our
management will have broad discretion as to the use of the net proceeds from any offering by us and could use them for purposes
other than those contemplated at the time of this offering. Accordingly, you will be relying on the judgment of our management
with regard to the use of these net proceeds, and you will not have the opportunity, as part of your investment decision, to assess
whether the proceeds are being used appropriately. It is possible that the proceeds will be invested in a way that does not yield
a favorable, or any, return for Advaxis.
We
will require additional capital funding, the receipt of which may impair the value of our common stock.
Our
future capital requirements depend on many factors, including our research, development, sales and marketing activities. We will
need to raise additional capital through public or private equity or debt offerings or through arrangements with strategic partners
or other sources in order to continue to develop our drug candidates. There can be no assurance that additional capital will be
available when needed or on terms satisfactory to us, if at all. To the extent we raise additional capital by issuing equity securities,
our stockholders may experience substantial dilution and the new equity securities may have greater rights, preferences or privileges
than our existing common stock.
Our
common stock may be delisted from the Nasdaq Global Select Market if we fail to regain compliance with continued listing standards.
On
April 8, 2020, we received a written notification from the Nasdaq Stock Market (“Nasdaq”) indicating that we were
not in compliance with Nasdaq Listing Rule 5450(a)(1) because the closing bid price for our common stock had closed below $1.00
per share for the previous 30 consecutive business days. The notification of noncompliance has no immediate effect on the listing
or trading of our common stock on the Nasdaq Global Select Market under the symbol “ADXS,” and we are currently monitoring
the closing bid price of our common stock and evaluating our alternatives, if appropriate, to regain compliance with this rule.
In accordance with Nasdaq’s listing rules, we would typically have 180 calendar days from the date of such notice, or until
October 5, 2020, to regain compliance with the minimum closing bid price requirement. However, on April 17, 2020, we received
an additional written notification from Nasdaq indicating that, due to extraordinary market conditions, Nasdaq has filed an immediately
effective rule change with the SEC that tolls the compliance period for the bid-price requirement through June 30, 2020. Accordingly,
upon expiration of the tolling period and beginning on July 1, 2020, we will then have 172 calendar days (which is the balance
of days we had to regain compliance under the previously effective compliance period) from July 1, 2020, or until December 21,
2020, to regain compliance with the bid-price requirement. To regain compliance, the closing bid price of our common stock must
meet or exceed $1.00 per share for a minimum of 10 consecutive business days, unless the staff of the Nasdaq exercises its discretion
to extend this 10 business day period.
As
a matter of course, we explore and evaluate strategic transactions for our company. We may not be successful in identifying or
completing any strategic transaction and any such strategic transaction completed may not yield additional value for stockholders.
As
a matter of course, we are reviewing strategic transactions and alternatives and there can be no assurance that we will be successful
in identifying or completing any strategic transactions, that any such strategic transaction will result in additional value for
our stockholders or that the process will not have an adverse impact on our business. These transactions could include, but are
not limited to, collaboration agreements, co-development agreements, strategic mergers, reverse mergers, the issuance or buyback
of public shares, or the purchase or sale of specific assets, in addition to other potential actions aimed at increasing stockholder
value. There can be no assurance that the review of strategic transactions will result in the identification or consummation of
any transaction. Our Board of Directors may also determine that our most effective strategy is to continue to effectuate our current
business plan. The process of reviewing strategic transactions may be time consuming and disruptive to our business operations
and, if we are unable to effectively manage the process, our business, financial condition and results of operations could be
adversely affected. We could incur substantial expenses associated with identifying and evaluating potential strategic alternatives.
No decision has been made with respect to any transaction and we cannot assure you that we will be able to identify and undertake
any transaction that allows our shareholders to realize an increase in the value of their common stock or provide any guidance
on the timing of such action, if any.
We
also cannot assure you that any potential strategic transaction or other alternative transaction, if identified, evaluated and
consummated, will provide greater value to our stockholders than that reflected in the current price of our common stock. Any
potential transaction would be dependent upon a number of factors that may be beyond our control, including, but not limited to,
market conditions, industry trends, the interest of third parties in our business and the availability of financing to potential
buyers on reasonable terms. We do not intend to comment regarding the evaluation of strategic alternatives until such time as
our Board of Directors has determined the outcome of the process or otherwise has deemed that disclosure is appropriate or required
by applicable law. As a consequence, perceived uncertainties related to our future may result in the loss of potential business
opportunities and volatility in the market price of our common stock and may make it more difficult for us to attract and retain
qualified personnel and business partners.
Risks
Related to our Business and Operations
A
global health crisis such as a pandemic, epidemic or outbreak of an infectious disease, such as COVID-19, may materially and adversely
affect our business and operations.
On
March 11, 2020, the World Health Organization declared the outbreak of COVID-19 a pandemic. The COVID-19 pandemic is affecting
the United States and global economies and has affected, and may continue to affect, our operations and those of third parties
on which we rely, including by causing disruptions in our raw material supply and the manufacturing of our product candidates.
In addition, the COVID-19 pandemic has affected the operations of the U.S. Food and Drug Administration (the “FDA”)
and other health authorities, which can result in delays of reviews and approvals, including with respect to our product candidates.
The evolving COVID-19 pandemic has, and may continue to, directly or indirectly affect the pace of enrollment in our clinical
trials as patients may avoid or may not be able to travel to healthcare facilities and physicians’ offices unless due to
a health emergency and clinical trial staff can no longer get to the clinic. Additionally, such facilities and offices have been
and may continue to be required to focus limited resources on non-clinical trial matters, including treatment of COVID-19 patients,
thereby decreasing availability, in whole or in part, for clinical trial services. In addition, employee disruptions and remote
working environments related to the COVID-19 pandemic and the federal, state and local responses to such virus, could materially
affect the efficiency and pace with which we work and develop our product candidates and the manufacturing of our product candidates.
In addition, COVID-19 infection of our workforce could result in a temporary disruption in our business activities, including
manufacturing and other functions. Further, while the potential economic impact brought by, and the duration of, the COVID-19
pandemic is difficult to assess or predict, the impact of the COVID-19 pandemic on the global financial markets may reduce our
ability to access capital, which could negatively affect our short-term and long-term liquidity. Additionally, the stock market
has been unusually volatile during the COVID-19 outbreak and such volatility may continue. The ultimate impact of the COVID-19
pandemic is highly uncertain and subject to change. We do not yet know the full extent of potential delays or impacts on our business,
financing or clinical trial activities, or on healthcare systems or the global economy as a whole. However, these effects could
have a material impact on our liquidity, capital resources, operations and business and those of the third parties on which we
rely.
USE
OF PROCEEDS
We
estimate the net proceeds from this offering will be approximately $19.9 million after deducting our estimated offering expenses.
We
currently intend to use the net proceeds, if any, from this offering to advance our ADXS-HOT program
and for general corporate purposes.
We
may also use a portion of the net proceeds to acquire or invest in complementary businesses, products and technologies. Although
we currently have no specific agreements, commitments or understandings with respect to any acquisition or investment, we evaluate
acquisition and investment opportunities and may engage in related discussions with other companies from time to time.
As
of the date of this prospectus supplement, we cannot specify with certainty all of the particular uses for the net proceeds to
us from this offering. Accordingly, our management will have broad discretion in the timing and application of these proceeds.
Pending application of the net proceeds as described above, we may invest the net proceeds of this offering in a variety of capital
preservation investments, including but not limited to short-term, interest-bearing investment grade securities, money market
accounts, certificates of deposit and direct or guaranteed obligations of the U.S. government.
DIVIDEND
POLICY
We
have never declared or paid any cash dividends on our common stock and do not anticipate paying any cash dividends in the foreseeable
future. Any future determination to pay dividends will be at the discretion of our Board of Directors and will depend on our financial
condition, operating results, capital requirements and other factors that our Board of Directors considers to be relevant.
DILUTION
If
you invest in our common stock, your ownership interest will be diluted to the extent of the difference between the price per
share you pay in this offering and the net tangible book value per share of our common stock immediately after this offering.
The net tangible book value of our common stock as of April 30, 2020 was approximately $31.3 million, or approximately $0.52 per
share of common stock based upon 60,252,444 shares outstanding. Net tangible book value per share is equal to our total tangible
assets, less our total liabilities, divided by the total number of shares outstanding as of April 30, 2020.
After giving effect to the initial
issuance and sale to Lincoln Park of 3,510,527 shares of our common stock at an offering price of approximately $0.57 per share
and the issuance of the Commitment Shares, each in accordance with the Purchase Agreement, and after deducting commissions
and estimated offering expenses payable by us, our as adjusted net tangible book value as of April 30, 2020 would have been approximately
$33.3 million, or $0.51 per share of common stock. This represents an immediate decrease in net tangible
book value of $0.01 per share to our existing stockholders and an immediate dilution of $0.06 per share to new investors
in this offering. The following table illustrates this calculation on a per share basis.
|
Offering price per share
|
|
|
|
|
|
$
|
0.57
|
|
|
Net tangible book value per share as of April 30, 2020
|
|
$
|
0.52
|
|
|
|
|
|
|
Increase in net
tangible book value per share attributable to the offering
|
|
$
|
0.01
|
|
|
|
|
|
|
As adjusted net
tangible book value per share as of April 30, 2020, after giving effect to this offering
|
|
|
|
|
|
$
|
0.51
|
|
|
Dilution per share to the purchaser in the offering
|
|
|
|
|
|
$
|
0.06
|
|
The
number of shares of common stock to be outstanding immediately after this offering is based on 60,252,444 shares of common stock
outstanding as of April 30, 2020. The number of shares outstanding as of April 30, 2020 excludes:
|
|
●
|
5,398,226
shares of our common stock reserved for issuance upon the exercise of outstanding warrants at a weighted average exercise
price of $1.16 per share;
|
|
|
|
|
|
|
●
|
5,818
shares of our common stock reserved for issuance upon settlement of restricted stock units;
|
|
|
|
|
|
|
●
|
252,296
shares of our common stock reserved for issuance upon the
exercise of outstanding stock options at a weighted average exercise price of $73.50 per share; and
|
|
|
|
|
|
|
●
|
210,787
shares of our common stock reserved for future awards under our 2015 Incentive Plan.
|
Except
as otherwise indicated, all information in this prospectus supplement assumes no exercise or forfeiture of the outstanding options
or remaining warrants or settlement of restricted stock units after April 30, 2020. In addition, on May 4, 2020, we amended our
2015 Incentive Plan to increase the number of shares available for issuance pursuant to awards granted thereunder from 877,744
shares to 6,000,000 shares of common stock.
PLAN
OF DISTRIBUTION
This
prospectus supplement and the accompanying prospectus relate to the issuance and sale of up to $20,000,000 of shares of our common
stock that we may issue to Lincoln Park from time to time under a Purchase Agreement that we entered into with Lincoln Park
on July 30, 2020, and an additional 1,084,266 shares of common stock that are being issued to Lincoln Park as Commitment
Shares for entering into the Purchase Agreement. This prospectus supplement and the accompanying prospectus also cover the resale
of these shares by Lincoln Park to the public.
Pursuant
to the Purchase Agreement, over the 36-month term of the Purchase Agreement, up to an aggregate amount of $20,000,000 (subject
to certain limitations) of shares of our common stock, we have the right, but not the obligation, from time to time, in
our sole discretion and subject to certain conditions, to direct Lincoln Park to purchase up to the Regular Purchase Share Limit
in a Regular Purchase. Lincoln Park’s maximum obligation under any single Regular Purchase will not exceed $1,000,000, unless
we mutually agree to increase the maximum amount of such Regular Purchase. The purchase price for shares of common stock
to be purchased by Lincoln Park under a Regular Purchase will be the equal to the lower of (in each case, subject to the adjustments
described in the Purchase Agreement): (i) the lowest sale price for our common stock on the applicable purchase date, and
(ii) the arithmetic average of the three lowest sale prices for our common stock during the ten trading days prior to the
purchase date.
If
we direct Lincoln Park to purchase the maximum number of shares of common stock we then may sell in a Regular Purchase,
then in addition to such Regular Purchase, and subject to certain conditions and limitations in the Purchase Agreement, we may
direct Lincoln Park to make an “accelerated purchase” of an additional amount of common stock that may not
exceed the lesser of (i) 300% of the number of shares purchased pursuant to the corresponding Regular Purchase and (ii) 30% of
the total number of shares of our common stock traded during a specified period on the applicable purchase date as set
forth in the Purchase Agreement. Under certain circumstances and in accordance with the Purchase Agreement, the Company may direct
Lincoln Park to purchase shares in multiple accelerated purchases on the same trading day.
We
will control the timing and amount of any sales of our common stock to Lincoln Park. There is no upper limit on the price
per share that Lincoln Park must pay for our common stock under the Purchase Agreement.
As
consideration for entering into the Purchase Agreement, the Company issued to Lincoln Park 1,084,266 shares of common stock.
The Company will not receive any cash proceeds from the issuance of these shares. Pursuant
to the Purchase Agreement, Lincoln Park has agreed to an initial purchase of 3,510,527 shares of our common stock for an aggregate
purchase price of $1,999,999.89.
We
may at any time, in our sole discretion terminate the Purchase Agreement without fee, penalty or cost, upon one trading day written
notice. In the event of bankruptcy proceedings by or against us, the Purchase Agreement will automatically terminate without action
of any party.
We
may suspend the sale of shares to Lincoln Park pursuant to this prospectus supplement for certain periods of time for certain
reasons, including if this prospectus supplement is required to be supplemented or amended to include additional material information.
Lincoln
Park is an “underwriter” within the meaning of Section 2(a)(11) of the Securities Act. Lincoln Park has informed us
that it will use an unaffiliated broker-dealer to effectuate all sales, if any, of the common stock that it may purchase from
us pursuant to the Purchase Agreement. Such sales will be made at prices and at terms then prevailing or at prices related to
the then current market price. Each such unaffiliated broker-dealer will be an underwriter within the meaning of Section 2(a)(11)
of the Securities Act. Lincoln Park has informed us that each such broker-dealer will receive commissions from Lincoln Park that
will not exceed customary brokerage commissions.
We
know of no existing arrangements between Lincoln Park and any other stockholder, broker, dealer, underwriter, or agent relating
to the sale or distribution of the shares offered by this Prospectus. At the time a particular offer of shares is made, a prospectus
supplement, if required, will be distributed that will set forth the names of any agents, underwriters, or dealers and any compensation
from the selling stockholder, and any other required information.
We
will pay all of the expenses incident to the registration, offering, and sale of the shares to Lincoln Park. We have agreed to
indemnify Lincoln Park and certain other persons against certain liabilities in connection with the offering of shares of common
stock offered hereby, including liabilities arising under the Securities Act or, if such indemnity is unavailable, to contribute
amounts required to be paid in respect of such liabilities.
Insofar
as indemnification for liabilities arising under the Securities Act may be permitted to our directors, officers, and controlling
persons, we have been advised that in the opinion of the SEC this indemnification is against public policy as expressed in the
Securities Act and is therefore unenforceable.
Lincoln
Park represented to us that at no time prior to the date of the Purchase Agreement has Lincoln Park or its agents, representatives
or affiliates engaged in or effected, in any manner whatsoever, directly or indirectly, any short sale (as such term is defined
in Rule 200 of Regulation SHO of the Exchange Act) of our common stock or any hedging transaction. Lincoln Park agreed
that during the term of the Purchase Agreement, it, its agents, representatives or affiliates will not enter into or effect, directly
or indirectly, any of the foregoing transactions.
We
have advised Lincoln Park that it is required to comply with Regulation M promulgated under the Exchange Act. With certain exceptions,
Regulation M precludes the selling stockholder, any affiliated purchasers, and any broker-dealer or other person who participates
in the distribution from bidding for or purchasing, or attempting to induce any person to bid for or purchase any security which
is the subject of the distribution until the entire distribution is complete. Regulation M also prohibits any bids or purchases
made in order to stabilize the price of a security in connection with the distribution of that security. All of the foregoing
may affect the marketability of the shares offered by this prospectus supplement.
LEGAL
MATTERS
Certain
legal matters with respect to the securities offered by this prospectus supplement will be passed upon for us by Morgan, Lewis
& Bockius LLP, Princeton, NJ. Certain legal matters will be passed upon for Lincoln Park by K&L Gates LLP, Miami, FL.
EXPERTS
The
financial statements of Advaxis, Inc. incorporated in this prospectus supplement by reference to the 2019 Form 10-K/A have been
so incorporated in reliance upon the report of Marcum LLP, an independent registered public accounting firm, and upon the authority
of said firm as experts in accounting and auditing.
WHERE
YOU CAN FIND MORE INFORMATION
We
file annual, quarterly and current reports, proxy statements, and other information with the SEC. We file these documents with
the SEC electronically. You can access the electronic versions of these filings on the SEC’s website found at www.sec.gov.
You can also obtain copies of materials we file with the SEC, free of charge, from our website found at www.advaxis.com. Information
contained on our website does not constitute part of this prospectus supplement or the accompanying prospectus. Our stock is quoted
on the Nasdaq Global Select Market under the symbol “ADXS.”
INCORPORATION
OF CERTAIN INFORMATION BY REFERENCE
The
SEC allows us to “incorporate by reference” the information we file with them which means that we can disclose important
information to you by referring you to those documents instead of having to repeat the information in this prospectus supplement
and accompanying prospectus. The information incorporated by reference is considered to be part of this prospectus supplement
and accompanying prospectus, and later information that we file with the SEC will automatically update and supersede this information.
We incorporate by reference the documents listed below and any future filings made with the SEC under Sections 13(a), 13(c), 14
or 15(d) of the Exchange Act between the date of this prospectus supplement and the termination of the offering (other than, unless
otherwise specifically indicated, current reports furnished under Item 2.02 or Item 7.01 of Form 8-K and exhibits filed on such
form that are related to such items):
|
|
●
|
our
Annual Report on Form 10-K for the fiscal year ended October 31, 2019 (as filed with the SEC on December 31, 2019, and amended
by Amendment No. 1 thereto on Form 10-K/A filed on January 21, 2020 and by Amendment No. 2 thereto on Form 10-K/A filed on
February 28, 2020);
|
|
|
|
|
|
|
●
|
our
Quarterly Reports on Form 10-Q for the fiscal quarter ended January 31, 2020 filed with the SEC on March 13, 2020, and for
the fiscal quarter ended April 30, 2020, filed on June 11, 2020;
|
|
|
|
|
|
|
●
|
our
Current Reports on Form 8-K filed with the SEC on January 7, 2020, January 16, 2020, January 23, 2020, February 13, 2020,
February 14, 2020, February 20, 2020, February 27, 2020, April 10, 2020, April 20, 2020, May 5, 2020, May 8, 2020 and August
3, 2020; and
|
|
|
|
|
|
|
●
|
the
description of our common stock, par value $0.001 per share, contained in our Registration Statement on Form 8-A, filed with
the SEC on October 15, 2013, and all amendments and reports updating such description.
|
We
will provide to each person, including any beneficial owner, to whom a copy of this prospectus supplement and the related prospectus
is delivered, a copy of any or all of the information that we have incorporated by reference into this prospectus supplement and
the related prospectus, but not delivered with this prospectus supplement and the related prospectus. We will provide this information
upon written or oral request at no cost to the requester. You may request this information by contacting our corporate headquarters
at the following address: 305 College Road East, Princeton, New Jersey 08540, Attn: Molly Henderson, or by calling (609) 452-9813.
PROSPECTUS

$250,000,000
Advaxis,
Inc.
Common
Stock
Preferred
Stock
Debt
Securities
Warrants
Units
We
may offer and sell up to $250,000,000 in the aggregate of any combination of the securities described in this prospectus, from
time to time in one or more offerings. We may offer these securities separately or together
in units.
We
may offer and sell the securities described in this prospectus and any prospectus supplement to or through one or more underwriters,
dealers and agents, or directly to purchasers, or through a combination of these methods. If any underwriters, dealers or agents
are involved in the sale of any of the securities, their names and any applicable purchase price, fee, commission or discount
arrangement between or among them will be set forth, or will be calculable from the information set forth, in the applicable prospectus
supplement. See the sections of this prospectus entitled “About this Prospectus” and “Plan of Distribution”
for more information. No securities may be sold without delivery of this prospectus and the applicable prospectus supplement describing
the method and terms of the offering of such securities.
This
prospectus provides a general description of the securities we may offer. Each time we offer and sell securities, we will provide
specific terms of the securities offered in a supplement to this prospectus. The prospectus supplement may also add, update or
change information contained in this prospectus. You should read this prospectus and the applicable prospectus supplement carefully
before you invest in any securities being offered. This prospectus may not be used to consummate a sale of securities unless accompanied
by the applicable prospectus supplement.
Our
common stock is traded on the NASDAQ Global Select Market under the symbol “ADXS.” On August 22, 2018, the per share
closing price of our common stock as reported on the NASDAQ Global Select Market was $1.47 per share. We will provide information
in any applicable prospectus supplement regarding any listing of securities other than shares of our common stock on any securities
exchange.
Investing
in our securities involves certain risks. See “Risk Factors” on page 3 of this prospectus and any similar section
contained in the applicable prospectus supplement concerning factors you should consider before investing in our securities.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or
determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The
date of this prospectus is August 30, 2018
Table
of Contents
ABOUT
THIS PROSPECTUS
This
prospectus is part of a registration statement that we filed with the Securities and Exchange Commission, or the SEC, using a
“shelf” registration process. Under this shelf registration process, we may from time to time offer and sell any combination
of the securities described in this prospectus in one or more offerings for an aggregate initial offering price of up to $250,000,000.
Each time we sell securities, we will provide a prospectus supplement to this prospectus that contains specific information about
the terms of such offering. The prospectus supplement may also add, update or change information contained in this prospectus.
Before purchasing any securities, you should carefully read both this prospectus and any prospectus supplement, together with
the additional information incorporated into this prospectus or described under the heading “Where You Can Find More Information.”
You
should rely only on the information contained or incorporated by reference in this prospectus and any prospectus supplement. We
have not authorized any other person to provide you with different information. If anyone provides you with different or inconsistent
information, you should not rely on it. We will not make an offer to sell securities in any jurisdiction where the offer or sale
is not permitted. You should assume that the information appearing in this prospectus, as well as information we previously filed
with the SEC and have incorporated by reference, is accurate as of the date on the front cover of this prospectus only, or when
such document was filed with the SEC. Our business, financial condition, results of operations and prospects may have changed
since the relevant date.
We
will not use this prospectus to offer and sell securities unless it is accompanied by a prospectus supplement that more fully
describes the terms of the offering.
When
we refer to “Advaxis,” “we,” “our,” “us” and the “Company” in this
prospectus, we mean Advaxis, Inc., unless otherwise specified.
We
own various U.S. federal and foreign trademark registrations and applications, as well as pending and unregistered trademarks
and service marks. All trademarks or trade names referred to in this prospectus are the property of their respective owners. Solely
for convenience, the trademarks and trade names in this prospectus may be referred to without the ® and ™
symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest
extent under applicable law, their rights thereto.
WHERE
YOU CAN FIND MORE INFORMATION
We
file reports with the Securities and Exchange Commission, or the SEC, annually using Form 10-K, quarterly reports on Form 10-Q
and current reports on Form 8-K. You may read and copy any such reports and amendments thereto at the SEC’s Public Reference
Room at 100 F Street, N.E., Washington, D.C. 20549. Please call the SEC at 1-800-SEC-0330 for information on the Public Reference
Room. Additionally, the SEC maintains a website that contains annual, quarterly, and current reports, proxy statements, and other
information that issuers (including us) file electronically with the SEC. The SEC’s website address is www.sec.gov. You
can also obtain copies of materials we file with the SEC from our Internet website found at www.advaxis.com. The information on
our website, however, is not, and should not be deemed to be, a part of this prospectus or any prospectus supplement. We have
included our website address as an inactive textual reference only. Our stock is quoted on the NASDAQ Global Select Market under
the symbol “ADXS.”
INCORPORATION
OF CERTAIN INFORMATION BY REFERENCE
The
SEC allows us to “incorporate by reference” into this prospectus the information we file with the SEC. This means
that we can disclose important information to you by referring you to those documents without restating that information in this
document. The information incorporated by reference into this prospectus is considered to be part of this prospectus, and information
we file with the SEC pursuant to Section 13(a), 13(c), 14 or 15(d) of the Securities Exchange Act of 1934, as amended, or the
Exchange Act, including those made on or after the date of the initial filing of the registration statement of which this prospectus
is a part and prior to the effectiveness of such registration statement, will be deemed to be incorporated by reference into this
prospectus and will automatically update and supersede the information contained in this prospectus and documents listed below.
We incorporate by reference into this prospectus the documents listed below, except to the extent information in those documents
differs from information contained in this prospectus, and any future filings made by us with the SEC under Sections 13(a), 13(c),
14 or 15(d) of the Exchange Act prior to the termination of the offering, including exhibits (however, unless specifically indicated,
we do not incorporate by reference, whether listed below or filed in the future, current reports furnished under Item 2.02 or
Item 7.01 of Form 8-K or related exhibits furnished pursuant to Item 9.01 of Form 8-K):
|
|
●
|
our
Annual Report on Form 10-K for the fiscal year ended October 31, 2017 filed with the
Commission on December 21, 2017;
|
|
|
●
|
the
portions of our Definitive Proxy Statement on Schedule 14A filed with the Commission
on February 6, 2018 that are incorporated by reference into our Annual Report on Form
10-K for the fiscal year ended October 31, 2017;
|
|
|
●
|
our
Quarterly Reports on Form 10-Q filed with the Commission on March 12, 2018 and June 7,
2018;
|
|
|
●
|
our
Current Reports on Form 8-K filed with the Commission on December 20, 2017, February
15, 2018, February 21, 2018, February 22, 2018, March 12, 2018, March 21, 2018, April
23, 2018, June 6, 2018, June 8, 2018, July 10, 2018, July 13, 2018, July 17, 2018 and
July 30, 2018; and
|
|
|
●
|
the
description of our common stock, par value $0.001 per share, contained in our Registration
Statement on Form 8-A, filed with the Commission on October 15, 2013 and under the caption
“Description of Securities” in the Registrant’s prospectus, dated as
of October 15, 2013, forming a part of the Registration Statement on Form S-1 (Registration
No. 333-188637) filed with the Commission, including any amendments or reports filed
for the purpose of updating such description.
|
We
will provide to each person, including any beneficial owner, to whom a copy of this prospectus is delivered, a copy of any or
all of the information that we have incorporated by reference into this prospectus. We will provide this information upon written
or oral request at no cost to the requester. You may request this information by contacting our corporate headquarters at the
following address: 305 College Road East, Princeton, New Jersey 08540, Attn: Molly Henderson, or by calling (609) 452-9813.
RISK
FACTORS
Investment
in our common stock involves risks. You should consider carefully the risk factors set forth below as well as those contained
in the section entitled “Risk Factors” contained in our Annual Report on Form
10-K for the year ended October 31, 2017, as filed with the SEC on December 21, 2017, which is incorporated herein by reference
in its entirety, as well as any amendment or update to our risk factors reflected in subsequent filings with the SEC incorporated
by reference into this prospectus and other information contained in the applicable prospectus supplement. If any of the risks
or uncertainties described in our SEC filings actually occurs, our business, financial condition, results of operations or cash
flow could be materially and adversely affected. This could cause the trading price of our common stock to decline, resulting
in a loss of all or part of your investment. The risks and uncertainties we have described are not the only ones facing our company.
Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also affect our business
operations.
Risks
Related to Our Common Stock
Our
stock price can be volatile, which increases the risk of litigation, and may result in a significant decline in the value of your
investment.
The
trading price of our common stock is likely to be highly volatile and subject to wide fluctuations in price in response to various
factors, many of which are beyond our control and may not be related to our operating performance. These fluctuations could cause
you to lose part or all of your investment in our common stock. These factors include, but are not limited to, the following:
|
|
●
|
price and volume
fluctuations in the overall stock market from time to time;
|
|
|
●
|
changes in the market
valuations, stock market prices and trading volumes of similar companies;
|
|
|
●
|
actual or anticipated
changes in our net loss or fluctuations in our operating results or in the expectations of securities analysts;
|
|
|
●
|
the issuance of
new equity securities pursuant to a future offering, including potential issuances of preferred stock;
|
|
|
●
|
general economic
conditions and trends;
|
|
|
●
|
positive and negative
events relating to healthcare and the overall pharmaceutical and biotech sector;
|
|
|
●
|
major catastrophic
events;
|
|
|
●
|
sales of large blocks
of our stock;
|
|
|
●
|
additions or departures
of key personnel;
|
|
|
●
|
changes in the regulatory
status of our immunotherapies, including results of our pre-clinical and clinical trials;
|
|
|
●
|
positive and negative
changes in relationships with partners;
|
|
|
●
|
events affecting
the University of Pennsylvania or any of our other current or future collaborators;
|
|
|
●
|
announcements of
new products or technologies, commercial relationships or other events by us or our competitors;
|
|
|
●
|
regulatory developments
in the United States and other countries;
|
|
|
●
|
failure of our common
stock or warrants to be listed or quoted on the Nasdaq Stock Market, NYSE Amex Equities or other national market system;
|
|
|
●
|
changes in accounting
principles; and
|
|
|
●
|
discussion of us
or our stock price by the financial and scientific press and in online investor communities.
|
In
addition, equity markets in general, and the market for biotechnology and life sciences companies in particular, have experienced
extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of companies
traded in those markets. These broad market and industry factors may materially affect the market price of our common stock, regardless
of our development and operating performance. In the past, following periods of volatility in the market price of a company’s
securities, securities class-action litigation has often been instituted against that company. Due to the volatility of our stock
price, we have been and may be the target of securities litigation in the future. Securities litigation could result in substantial
costs and divert management’s in the future attention and resources from our business.
Future
sales or other issuances of our common stock could depress the market for our common stock.
Sales of a substantial number of shares of our common
stock, or the perception by the market that those sales could occur, could cause the market price of our common stock to decline
or could make it more difficult for us to raise funds through the sale of equity in the future.
In
connection with future offerings, we and our directors and executive officers may enter into lock-up agreements for certain periods
of time following such offerings. See “Plan of Distribution” below. Upon expiration or earlier release of the lock-up,
we and our directors and executive officers may sell shares into the market, which could adversely affect the market price of
shares of our common stock.
Future
issuances of common stock could further depress the market for our common stock. We expect to continue to incur drug development
and selling, general and administrative costs, and to satisfy our funding requirements, we will need to sell additional equity
securities, which may be subject to registration rights and warrants with anti-dilutive protective provisions. The sale or the
proposed sale of substantial amounts of our common stock or other equity securities in the public markets may adversely affect
the market price of our common stock and our stock price may decline substantially. Our stockholders may experience substantial
dilution and a reduction in the price that they are able to obtain upon sale of their shares. Also, new equity securities issued
may have greater rights, preferences or privileges than our existing common stock. In addition, we have a significant number of
shares of restricted stock, restricted stock units, stock options and warrants outstanding. To the extent that outstanding stock
options or warrants have been or may be exercised or other shares issued, investors in our common stock may experience further
dilution.
If
we make one or more significant acquisitions in which the consideration includes stock or other securities, our stockholders’
holdings may be significantly diluted. In addition, stockholders’ holdings may also be diluted if we enter into arrangements
with third parties permitting us to issue shares of common stock in lieu of certain cash payments upon the achievement of milestones.
We
do not intend to pay cash dividends.
We
have not declared or paid any cash dividends on our common stock, and we do not anticipate declaring or paying cash dividends
for the foreseeable future. Any future determination as to the payment of cash dividends on our common stock will be at our Board
of Directors’ discretion and will depend on our financial condition, operating results, capital requirements and other factors
that our Board of Directors considers to be relevant.
Certain
anti-takeover provisions in our charter documents and Delaware law could make a third-party acquisition of us difficult. This
could limit the price investors might be willing to pay in the future for our common stock.
Provisions
in our amended and restated certificate of incorporation and amended and restated bylaws could have the effect of making it more
difficult for a third party to acquire, or of discouraging a third party from attempting to acquire, or control us. These factors
could limit the price that certain investors might be willing to pay in the future for shares of our common stock. Our amended
and restated certificate of incorporation allows us to issue preferred stock without the approval of our stockholders. The issuance
of preferred stock could decrease the amount of earnings and assets available for distribution to the holders of our common stock
or could adversely affect the rights and powers, including voting rights, of such holders. In certain circumstances, such issuance
could have the effect of decreasing the market price of our common stock. Our amended and restated bylaws also provide our board
of directors with the ability to alter such bylaws without stockholder approval. Any of these provisions could also have the effect
of delaying or preventing a change in control.
Risks
Related to Our Business
We
expect that we will need to raise additional funding to complete the development and commercialization of our product candidates.
This additional financing may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed
may force us to delay, limit, or terminate our product development efforts or other operations.
We estimate
that our current cash, cash equivalents and investments will be sufficient for us to fund our operating expenses and capital expenditure
requirements through 2019. We have based this estimate on assumptions that may prove to be wrong, and we could utilize our available
capital resources sooner than we currently expect. We will need to raise substantial additional capital to complete the development
and commercialization of our product candidates. We will continue to seek funds through equity or debt financings, collaborative
or other arrangements with corporate sources, or through other sources of financing. Adequate additional funding may not be available
to us on acceptable terms, or at all. Any failure to raise capital as and when needed, as a result of insufficient authorized
shares or otherwise, could have a negative impact on our financial condition and on our ability to pursue our business plans and
strategies.
Our
existing and future collaborations are important to our business. If we are unable to establish or manage strategic collaborations,
our revenue and drug development may be limited.
Our
strategy includes eventual substantial reliance upon strategic collaborations for marketing and commercialization of our clinical
product candidates, and we currently rely on strategic collaborations for research, development, marketing and commercialization
for some of our immunotherapies. To date, we have been heavily reliant upon third party outsourcing for our clinical trials execution
and production of drug supplies for use in clinical trials.
In
addition, as discussed below in “Advaxis, Inc.—Cervical Cancer,” we are currently seeking a partner to fund
the development and commercialization of axalimogene filolisbac in cervical cancer. If a partner is not found, subject to ongoing
discussions with our collaboration partners over our obligations with respect the program, we anticipate winding down the program
in a clinically responsible manner. We may incur additional costs in connection with such a wind-down, including in severing our
relationship with our collaboration partners. Some of the costs are indeterminable at this time and there is no guarantee that
we will be able to wind down the program effectively, which could lead to considerable expense and could negatively affect our
financial results.
Establishing
strategic collaborations is difficult and time-consuming. Our discussions with potential collaborators may not lead to the establishment
of collaborations on favorable terms, if at all. For example, potential collaborators may reject collaborations based upon their
assessment of our financial, clinical, regulatory or intellectual property position. Our current collaborations, as well as any
future new collaborations, may never result in the successful development or commercialization of our immunotherapies or the generation
of sales revenue. To the extent that we have entered or will enter into co-promotion or other collaborative arrangements, our
product revenues are likely to be lower than if we directly marketed and sold any products that we may develop.
Management
of our relationships with our collaborators requires:
|
|
●
|
significant time
and effort from our management team;
|
|
|
●
|
financial funding
to support said collaboration;
|
|
|
●
|
coordination of
our research and development programs with the research and development priorities of our collaborators; and
|
|
|
●
|
effective allocation
of our resources to multiple projects.
|
In
addition, our existing collaborations, and any future collaborations we may enter into, may pose a number of risks, including
the fact that:
|
|
●
|
we will not directly
control the amount or timing of resources devoted by our collaborators to activities related to our immunotherapies;
|
|
|
●
|
our collaborators
may not commit sufficient resources to our research and development programs or the commercialization, marketing or distribution
of our immunotherapies;
|
|
|
●
|
our collaborators
may not perform their obligations as expected;
|
|
|
●
|
our collaborators
may fail to comply with applicable regulatory requirements regarding the development, manufacture, distribution or marketing
of a product candidate or product;
|
|
|
●
|
our collaborators
may pursue existing or other development-stage products or alternative technologies in preference to those being developed
in collaboration with us;
|
|
|
●
|
our collaborators
could independently develop, or develop with third parties, products that compete directly or indirectly with our products
and product candidates, or product candidates discovered in collaboration with us may be viewed by our collaborators as competitive
with their own product candidates or products, which in either case may cause our collaborators to cease to devote resources
to us;
|
|
|
●
|
if we fail to make
required milestone or royalty payments to our collaborators or to observe other obligations in our agreements with them, our
collaborators may have the right to terminate those agreements;
|
|
|
●
|
our collaborators
may seek to renegotiate agreements we have entered into, or may disagree with us about the terms and implementation of these
agreements;
|
|
|
●
|
disagreements with
our collaborators, including disagreements over proprietary rights, contract interpretation or the preferred course of development,
might cause delays or terminations of the research, development or commercialization of product candidates, might lead to
additional responsibilities for us with respect to product candidates, or might result in litigation or arbitration, any of
which would be time-consuming and expensive;
|
|
|
●
|
our collaborators
may not properly maintain or defend our intellectual property rights or may use our proprietary information in a manner that
may invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us
to potential litigation; and
|
|
|
●
|
our collaborators
may infringe the intellectual property rights of third parties, which may expose us to litigation and potential liability.
|
If
any of the above risks develop into actual events, it may result in considerable expense to us and could negatively affect our
drug development, revenue and financial results.
We
are subject to numerous risks inherent in conducting clinical trials.
We
outsource the management of our clinical trials to third parties. Agreements with clinical research organizations, clinical investigators
and medical institutions for clinical testing and data management services, place substantial responsibilities on these parties
that, if unmet, could result in delays in, or termination of, our clinical trials. For example, if any of our clinical trial sites
fail to comply with FDA-approved good clinical practices, we may be unable to use the data gathered at those sites. If these clinical
investigators, medical institutions or other third parties do not carry out their contractual duties or regulatory obligations
or fail to meet expected deadlines, or if the quality or accuracy of the clinical data they obtain is compromised due to their
failure to adhere to our clinical protocols or for other reasons, our clinical trials may be extended, delayed or terminated,
and we may be unable to obtain regulatory approval for, or successfully commercialize, our agents. We are not certain that we
will successfully recruit enough patients to complete our clinical trials nor that we will reach our primary endpoints. Delays
in recruitment, lack of clinical benefit or unacceptable side effects would delay or prevent the initiation of future development
of our agents. We or our regulators may suspend or terminate our clinical trials for a number of reasons. We may voluntarily suspend
or terminate our clinical trials if at any time we believe they present an unacceptable risk to the patients enrolled in our clinical
trials or do not demonstrate clinical benefit. In addition, regulatory agencies may order the temporary or permanent discontinuation
of our clinical trials, or place our products on temporary or permanent hold, at any time if they believe that the clinical trials
are not being conducted in accordance with applicable regulatory requirements or that they present an unacceptable safety risk
to the patients enrolled in our clinical trials. For example, our Phase 1/2 trial of axalimogene filolisbac in combination with
durvalumab for the treatment of patients with metastatic squamous or non-squamous carcinoma of the cervix and metastatic HPV-associated
SCCHN was placed on clinical hold by FDA on March 9, 2018, following its review of a safety report regarding a Grade 5 Serious
Adverse Event occurring on February 27, 2018 and involving respiratory failure which followed a sixth combination cycle (11th
dose of axalimogene filolisbac, 21st dose of durvalumab) in the trial. Although this has been a single event in our development
of axalimogene filolisbac to date, we agreed on new guidelines for the early detection and treatment of such rare events with
the FDA that will be implemented for all axalimogene filolisbac programs. As development of axalimogene filolisbac continues
we may find that this event is not as rare as our experience to date indicates, our development of axalimogene filolisbac may
be put on clinical hold by regulatory authorities or terminated voluntarily by us for safety concerns.
Our
clinical trial operations are subject to regulatory inspections at any time. If regulatory inspectors conclude that we or our
clinical trial sites are not in compliance with applicable regulatory requirements for conducting clinical trials, we may receive
reports of observations or warning letters detailing deficiencies, and we will be required to implement corrective actions. If
regulatory agencies deem our responses to be inadequate, or are dissatisfied with the corrective actions we or our clinical trial
sites have implemented, our clinical trials may be temporarily or permanently discontinued, we may be fined, we or our investigators
may be precluded from conducting any ongoing or any future clinical trials, the government may refuse to approve our marketing
applications or allow us to manufacture or market our products, and we may be criminally prosecuted. The lengthy approval process
as well as the unpredictability of future clinical trial results may result in our failing to obtain regulatory approval for our
product candidates, which would materially harm our business, results of operations and prospects.
The
recently enacted tax reform bill could adversely affect our business and financial condition.
On December
22, 2017, President Trump signed into law the “Tax Cuts and Jobs Act,” or the TCJA, which significantly amends the
Internal Revenue Code of 1986. The TCJA, among other things, reduces the corporate tax rate from a top marginal rate of 35% to
a flat rate of 21%, limits the tax deduction for interest expense to 30% of adjusted earnings, eliminates net operating loss carrybacks,
imposes a one-time tax on offshore earnings at reduced rates regardless of whether they are repatriated, allows immediate deductions
for certain new investments instead of deductions for depreciation expense over time, and modifies or repeals many business deductions
and credits (including reducing the business tax credit for certain clinical testing expenses incurred in the testing of certain
drugs for rare diseases or conditions generally referred to as “orphan drugs”). We continue to examine the impact
these changes may have on our business. Notwithstanding the reduction in the corporate income tax rate, the overall impact of
the TCJA is uncertain and our business and financial condition could be adversely affected. The impact of the TCJA on holders
of our common stock is also uncertain and could be adverse. This prospectus does not discuss the TCJA or the manner in which it
might affect us or purchasers of our common stock. We urge our stockholders to consult with their legal and tax advisers with
respect to the TCJA and the potential tax consequences of investing in our common stock.
We
are subject to U.S. and certain foreign export and import controls, sanctions, embargoes, anti-corruption laws, and anti-money
laundering laws and regulations. We can face criminal liability and other serious consequences for violations which can harm our
business.
We
are subject to U.S. export control and economic sanctions laws and regulations and other restrictions on international trade.
As such, we are required to export our technology, products, and services in compliance with those laws and regulations. If we
export our technology, products, or services, the exports may require authorizations, including a license, a license exception
or other appropriate government authorization. In addition, the United States and other governments and their agencies impose
sanctions and embargoes on certain countries, their governments and designated parties, which may prohibit the export of certain
technology, products, and services to such persons altogether.
We
are also subject to the U.S. Foreign Corrupt Practices Act of 1977, as amended, the U.S. domestic bribery statute contained in
18 U.S.C. § 201, the U.S. Travel Act, the USA PATRIOT Act, and possibly other state and national anti-bribery and anti-money
laundering laws in countries in which we conduct activities. Anti-corruption laws are interpreted broadly and prohibit companies
and their employees, third-party intermediaries, and other associated persons from authorizing, promising, offering, providing,
soliciting, or accepting directly or indirectly, improper payments or benefits to or from any person whether in the public or
private sector. We have direct or indirect interactions with officials and employees of government agencies. We can be held liable
for the corrupt or other illegal activities of our employees, representatives, contractors, business partners, and agents, in
violation of U.S. and applicable foreign anti-corruption, export, import, sanctions, or anti-money laundering laws and regulations,
even if we do not explicitly authorize or have actual knowledge of such activities.
Any violation
of the laws and regulations described above may result in substantial civil and criminal fines and penalties, imprisonment, the
loss of export or import privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm,
and other consequences.
SPECIAL
CAUTIONARY NOTE REGARDING FORWARD LOOKING STATEMENTS
This
prospectus includes statements that are, or may be deemed, “forward-looking statements.” In some cases, these forward-looking
statements can be identified by the use of forward-looking terminology, including the terms “believes,” “estimates,”
“anticipates,” “expects,” “plans,” “intends,” “may,” “could,”
“might,” “will,” “should,” “approximately” or, in each case, their negative or
other variations thereon or comparable terminology, although not all forward-looking statements contain these words. They appear
in a number of places throughout this prospectus and include statements regarding our intentions, beliefs, projections, outlook,
analyses or current expectations concerning, among other things, our ongoing and planned discovery and development of drug candidates,
the strength and breadth of our intellectual property, our ongoing and planned preclinical studies and clinical trials, the timing
of and our ability to make regulatory filings and obtain and maintain regulatory approvals for our product candidates, the degree
of clinical utility of our product candidates, particularly in specific patient populations, expectations regarding clinical trial
data, our results of operations, financial condition, liquidity, prospects, growth and strategies, the length of time that we
will be able to continue to fund our operating expenses and capital expenditures, our expected financing needs and sources of
financing, the industry in which we operate and the trends that may affect the industry or us.
By
their nature, forward-looking statements involve risks and uncertainties because they relate to events, competitive dynamics,
and healthcare, regulatory and scientific developments and depend on the economic circumstances that may or may not occur in the
future or may occur on longer or shorter timelines than anticipated. Although we believe that we have a reasonable basis for each
forward-looking statement contained in this prospectus, we caution you that forward-looking statements are not guarantees of future
performance and that our actual results of operations, financial condition and liquidity, and the development of the industry
in which we operate may differ materially from the forward-looking statements contained in this prospectus. In addition, even
if our results of operations, financial condition and liquidity, and the development of the industry in which we operate are consistent
with the forward-looking statements contained in this prospectus, they may not be predictive of results or developments in future
periods.
Some
of the factors that we believe could cause actual results to differ from those anticipated or predicted include:
|
|
●
|
the
success and timing of our clinical trials, including patient accrual;
|
|
|
●
|
our
ability to obtain and maintain regulatory approval and/or reimbursement of our product
candidates for marketing;
|
|
|
●
|
our
ability to obtain the appropriate labeling of our products under any regulatory approval;
|
|
|
●
|
our
plans to develop and commercialize our products;
|
|
|
●
|
the
successful development and implementation of our sales and marketing campaigns;
|
|
|
●
|
the
change of key scientific or management personnel;
|
|
|
●
|
the
size and growth of the potential markets for our product candidates and our ability to
serve those markets;
|
|
|
●
|
our
ability to successfully compete in the potential markets for our product candidates,
if commercialized;
|
|
|
●
|
regulatory
developments in the United States and other countries;
|
|
|
●
|
the
rate and degree of market acceptance of any of our product candidates;
|
|
|
●
|
new
products, product candidates or new uses for existing products or technologies introduced
or announced by our competitors and the timing of these introductions or announcements;
|
|
|
●
|
market
conditions in the pharmaceutical and biotechnology sectors;
|
|
|
●
|
any
stockholder dilution that will result from future capital raising efforts and the exercise
or conversion, as applicable, of our outstanding options and warrants;
|
|
|
●
|
the
accuracy of our estimates regarding expenses, future revenues, capital requirements and
needs for additional financing;
|
|
|
●
|
our
ability to obtain additional funding;
|
|
|
●
|
our
ability to obtain and maintain intellectual property protection for our product candidates;
|
|
|
●
|
the
success and timing of our preclinical studies including investigational new drug application,
or IND, enabling studies;
|
|
|
●
|
the
ability of our product candidates to successfully perform in clinical trials;
|
|
|
●
|
our
ability to establish and manage strategic collaborations;
|
|
|
●
|
our
ability to initiate trials, enroll our trials, obtain and maintain approval of our product
candidates;
|
|
|
●
|
our
ability to manufacture and the performance of third-party manufacturers;
|
|
|
●
|
the
performance of our clinical research organizations, clinical trial sponsors and clinical
trial investigators; and
|
|
|
●
|
our
ability to successfully implement our strategy.
|
Any
forward-looking statements that we make in this prospectus speak only as of the date of such statement, and we undertake no obligation
to update such statements to reflect events or circumstances after the date of this prospectus. You should also read carefully
the factors described in the “Risk Factors” section of this prospectus and of our Annual Report on Form 10-K for the
year ended October 31, 2017, as filed with the SEC on December 21, 2017, or in our subsequent filings with the SEC incorporated
by reference herein to better understand the risks and uncertainties inherent in our business and underlying any forward-looking
statements. As a result of these factors, we cannot assure you that the forward-looking statements in this prospectus will prove
to be accurate.
This
prospectus includes statistical and other industry and market data that we obtained from industry publications and research, surveys
and studies conducted by third-parties. Industry publications and third-party research, surveys and studies generally indicate
that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or
completeness of such information. While we believe these industry publications and third-party research, surveys and studies are
reliable, we have not independently verified such data.
We
qualify all of our forward-looking statements by these cautionary statements. In addition, with respect to all of our forward-looking
statements, we claim the protection of the safe harbor for forward-looking statements contained in the Private Securities Litigation
Reform Act of 1995.
ADVAXIS,
INC.
We
are a late-stage biotechnology company focused on the discovery, development and commercialization of proprietary Listeria
monocytogenes, or Lm, based antigen delivery products. We are using our Lm platform directed against tumor-specific
targets in order to engage the patient’s immune system to destroy tumor cells. Through a license from the University of
Pennsylvania, we have exclusive access to this proprietary formulation of attenuated Lm called Lm Technology. Our
proprietary approach deploys a unique mechanism of action that redirects the immune system to attack cancer in three distinct
ways by:
|
|
●
|
Alerting
and training the immune system by activating multiple pathways in antigen-presenting
cells, or APCs, with the equivalent of multiple adjuvants;
|
|
|
●
|
Attacking
the tumor by generating a strong, cancer-specific T cell response; and
|
|
|
●
|
Breaking
down tumor protection through suppression of the protective cells in the tumor microenvironment
that shields the tumor from the immune system, enabling the activated T cells to begin
working to eliminate the tumor.
|
During
the second fiscal quarter, in an effort to maximize stockholder value and reduce operating expenses, we began assessing the clinical
and commercial viability of our R&D programs in order to determine which were best suited for internal development and which
were better suited for external development opportunities. In particular, we announced plans to take the following actions:
|
|
●
|
Minimize
future investment in cervical cancer and focus on potential partnership opportunities.
While our lead Human Papillomavirus, or HPV, program, axalimogene filolisbac, has shown
meaningful clinical efficacy and supports the manageable safety profile of our Lm
platform in HPV-related cancers, we plan to expand our search for a U.S. and/or European
partner who will take on all development and commercialization activities and costs related
to the HPV program. In the event no partner emerges, we intend to wind down the ongoing
trial in high-risk, locally advanced cervical cancer (AIM2CERV) and not conduct the PD-1
combination trial in metastatic cervical cancer (ADVANCE), which has yet to be initiated.
|
|
|
●
|
Evaluate
cost effective ways to invest in axalimogene filolisbac in head-and-neck cancer through
internal or external partnerships, or both.
|
|
|
●
|
Determine,
in the first quarter of 2019, a path forward for our program related to our ongoing trial
in metastatic prostate cancer with ADXS-PSA in combination with KEYTRUDA®
(pembrolizumab), Merck & Co.’s, or Merck’s, anti PD-1 antibody, which
early clinical data have proven worthy of continued evaluation.
|
|
|
●
|
Increase
internal investment in our ADXS-NEO and ADXS-HOT programs, both of which target neoantigens,
which are antigens encoded by tumor-specific mutated genes, a potentially transformational,
next-generation approach to treating cancer.
|
In
addition, on June 7, 2018, we announced that we would be implementing a reduction in force to align our staffing needs with our
new strategy. The reduction involved the elimination of approximately 24% of our work force to better align our resources with
our strategy outlined above.
ADXS-HOT
We
are currently prioritizing product development in the most prevalent cancers, with the first tumor type to be non-small cell lung
cancer, or NSCLC. On July 30, 2018, we announced the U.S. Food and Drug Administration’s, or FDA’s, allowance of our
ADXS-HOT IND in NSCLC. We plan to commence a first-in-human trial in NSCLC in 2018. We plan to submit additional INDs for the
ADXS-HOT program with prostate cancer in 2018 and bladder cancer in 2019, as well as a fourth ADXS-HOT drug candidate to be selected
from breast, colorectal, ovarian or head and neck cancers.
ADXS-HOT
preclinical data was presented in a poster presentation at the 2018 Annual Meeting of the American Association of Cancer Research,
or AACR. The study, entitled “Targeting Shared Hotspot Cancer Mutations with a Listeria monocytogenes Immunotherapy
Induce Potent Anti-Tumor Immunity” demonstrated that the ADXS-HOT platform could effectively target common (public or shared)
mutations (hotspots) and control tumor growth with both single and multi-target constructs.
ADXS-NEO
On
August 1, 2016, we entered into a global agreement, or the Amgen Agreement, with Amgen Inc., or Amgen, for the development and
commercialization of ADXS-NEO, a novel, preclinical investigational immunotherapy, using our proprietary Listeria monocytogenes
attenuated bacterial vector, which activates a patient’s immune system to respond against unique mutations, or neoepitopes,
contained in and identified from an individual patient’s tumor. Under the terms of the Amgen Agreement, Amgen received an
exclusive worldwide license to develop and commercialize ADXS-NEO. Under the Amgen Agreement, Amgen made an upfront payment to
us of $40 million and purchased an additional $25 million of our common stock. Amgen will fund the clinical development and commercialization
of ADXS-NEO while we will retain manufacturing responsibilities. We will collaborate with Amgen through a joint steering committee
for the development and commercialization of ADXS-NEO. We will also receive development, regulatory and sales milestone payments
of up to $475 million and high single-digit to double-digit royalty payments based on worldwide sales.
Preclinical
findings in our ADXS-NEO program were discussed in poster presentations at the 2018 AACR Annual Meeting. Additionally, portions
of these data were presented by Amgen at a podium presentation during the European Neoantigen Summit 2018.
The
first study, as discussed in a poster presentation at the AACR 2018 Annual Meeting, entitled “Neoantigens that fail to elicit
measurable T cell responses following peptide immunization can control tumor growth when delivered using a Listeria-based immunotherapy
platform,” showed that ADXS-NEO generates T cell responses against neoantigen peptides that control tumor growth, even when
they were identified as “non-immunogenic” using a conventional peptide-adjuvant immunization.
In
the second study, discussed in a poster presentation at the AACR 2018 Annual Meeting entitled “Targeting frameshift mutations
with a Listeria monocytogenes immunotherapy drives neoantigen-specific antitumor immunity in MC38 and CT26 mouse tumor
models,” Our Lm platform was shown to target frameshift mutations and generate T cells to multiple neoantigens per
frameshift in these models. This data highlighted the physical capacity of our Lm platform and its ability to target frameshift
mutations of greater than 90 amino acids, and to generate T cells to multiple neoantigens per frameshift in tumor mouse models.
The
initial tumor types for the ADXS-NEO Phase 1 trial are microsatellite stable colorectal cancer, head and neck cancer, and NSCLC.
On June 11, 2018, we announced that the first patient, being treated for metastatic NSCLC, was dosed in our ADXS-NEO Phase 1 trial.
ADXS-PSA
We
are conducting a Phase 1/2, open-label, multicenter, dose determination and expansion trial in collaboration with Merck evaluating
the safety and efficacy of ADXS-PSA as a monotherapy and in combination with KEYTRUDA® in patients with previously
treated metastatic, castration-resistant prostate cancer. We presented data at the 2018 American Society of Clinical Oncology,
or ASCO, annual meeting. ADXS-PSA was tested alone or in combination with KEYTRUDA in an advanced and heavily pretreated patient
population who had progressed on androgen deprivation therapy. A total of 13 and 37 patients were evaluated on monotherapy and
combination therapy, respectively. Overall, the safety profile was consistent with findings from prior clinical studies using
the Lm platform. Treatment-related adverse events were mostly mild or moderate constitutional symptoms such as fever, chills,
rigors, hypotension, nausea and fatigue, consistent with immune activation and manageable with standard care. There were no new
toxicities observed with the combination therapy. In all treated patients, those who received the combination therapy experienced
the longest overall survival, or OS, at data cut-off. Additional efficacy related data include:
|
|
●
|
Median
overall survival had not been reached in the combination arm after 13 months of follow-up
(95%CI 7.16-NR), and was 7.79 months (95%CI 3.52-11.9) in the monotherapy arm.
|
|
|
●
|
56.8%
of patients on combination therapy and 38.5% of patients on monotherapy did not experience
disease progression.
|
|
|
●
|
The
percentage of patients with prostate-specific antigen, or PSA, declines from baseline
in the combination therapy arm was 40.5%, and 15.4% in the monotherapy arm.
|
|
|
●
|
In
all treated patients, an improvement in survival was observed in patients with PSA declines
from baseline of 50% or greater vs. those with PSA declines of less than 50%. There were
7 patients in the combination arm with 50% or greater declines in PSA from baseline,
and none in the monotherapy arm.
|
HPV
Related Cancers
We
have several programs in HPV-related cancers based on axalimogene filolisbac, an Lm –based antigen delivery product
designed to target cells expressing HPV. Axalimogene filolisbac is currently under investigation in three HPV-associated cancers:
cervical cancer, head and neck cancer, and anal cancer, either as a monotherapy or in combination with other therapies, and has
shown encouraging safety and efficacy in numerous clinical studies to date.
Cervical
Cancer
We
completed a randomized Phase 2 clinical study (Lm -LLO-E7-15), conducted exclusively in India, in 110 women with recurrent/refractory
cervical cancer. The final results showed that 34.9% (38/109) of patients were alive at 12 months, 24.8% (27/109) of patients
were Long-term Survivors, or LTS, alive greater than 18 months. Of the 15 patients consenting to further follow-up beyond 18 months,
12 (or 11%) achieved 24-month OS status (range 24 – 34+ months) at the time of study closure. Axalimogene filolisbac was
found to be well tolerated with the majority of the AEs were mild to moderate in severity (566 of 704 reported adverse events,
or 80.4%) and were not related to study drug (539 of 704 reported AEs, 76.6%). These data were published in the May 2018 edition
of the peer-reviewed International Journal of Gynecological Cancer.
We
also previously reported results from a Phase 2 clinical study (GOG-0265) in 50 patients, which showed a 12-month overall survival
rate (primary efficacy endpoint) of 38% (n=19/50) in women with persistent, recurrent or metastatic carcinoma of the cervix, representing
a 55% improvement over a model-predicted 12-month overall survival rate of 24.5%. As more than half of the women treated in this
study had received multiple prior lines of therapy including with bevacizumab treatment, the 38% 12-month overall survival rate
was unprecedented when compared against historical data. We continue to believe that the results from the GOG-0265 study are clinically
meaningful and provide proof-of-concept that axalimogene filolisbac demonstrated clinical activity in metastatic cervical cancer.
Our
ongoing Phase 3 trial, AIM2CERV or “Advaxis Immunotherapy 2 Prevent Cervical Recurrence, is
evaluating axalimogene filolisbac in patients with high-risk, locally advanced cervical. The study is being conducted under a
Special Protocol Assessment, and has been determined by the FDA to be adequate, well-designed, and suitable for registration if
successful. This study is being conducted in collaboration with the GOG/NRG Oncology, and we have initiated the AIM2CERV study
to support a Biologics License Application submission in the United States and regulatory registration in other territories around
the world.
AIM2CERV
is a double-blind, randomized, placebo-controlled, Phase 3 study of adjuvant axalimogene filolisbac, following primary chemoradiation
treatment of women with high-risk locally advanced cervical cancer, or HRLACC. The primary objective of AIM2CERV is to compare
the disease free survival of axalimogene filolisbac to placebo administered in the adjuvant setting following standard concurrent
chemotherapy and radiotherapy administered with curative intent to patients with HRLACC. Secondary endpoints include examining
overall survival and safety. Our goal is to develop a treatment to prevent or reduce the risk of cervical cancer recurrence after
primary, standard of care, treatment in women who are at high risk of recurrence. The study is active in fourteen countries with
129 sites open to date.
In
February 2018, we submitted a conditional marketing authorization application, or MAA, to the European Medicines Agency’s,
or EMA, Committee for our lead Lm Technology product candidate, axalimogene filolisbac, for the treatment of adult women
who progress beyond first-line therapy of persistent/recurrent metastatic cervical cancer, or PRmCC. The MAA submission was primarily
based on data from the GOG-0265 study, as well as supportive data from other clinical trials evaluating axalimogene filolisbac
and was validated by the EMA in March 2018.
On
July 10, 2018, we announced plans to withdraw our conditional MAA based on EMA feedback following its initial review indicating
the application will likely need additional data to support a conditional approval. We continue to believe the results from the
GOG-0265 study are clinically meaningful and provide proof-of-concept that axalimogene filolisbac demonstrated clinical activity
in metastatic cervical cancer. The withdrawal of this application does not impact the ongoing clinical trials of axalimogene filolisbac.
We are seeking a U.S. and/or European partner to fund the development and commercialization of axalimogene filolisbac in cervical
cancer including the completion of the AIM2CERV study. If a partner is not found, subject to ongoing discussions with our collaboration
partners over our obligations with respect the program, we anticipate winding down the program in a clinically responsible manner.
We may incur additional costs in connection with such a wind-down, including in severing our relationship with our collaboration
partners, some of which are indeterminable at this time and there is no guarantee that we will be able to wind down the program
effectively.
MedImmune
Collaboration
We
have a clinical trial collaboration agreement with MedImmune, the global biologics research and development arm of AstraZeneca,
and are conducting a Phase 1/2, open-label, multicenter, two-part study to evaluate the safety and efficacy of axalimogene filolisbac
in combination with MedImmune’s investigational anti-PD-L1 immune checkpoint inhibitor, durvalumab, as a combination treatment
for patients with metastatic squamous or non-squamous carcinoma of the cervix and metastatic HPV-associated SCCHN. The dose-escalation
part of this study has been completed. We have commenced enrollment in the Part A (20 patients with SCCHN) and B (90 patients
with cervical cancer) expansion phases; however, this trial was placed on clinical hold by FDA on March 9, 2018, following its
review of a safety report regarding a Grade 5 Serious Adverse Event occurring on February 27, 2018 and involving respiratory failure
which followed a sixth combination cycle (11th dose of axalimogene filolisbac, 21st dose of durvalumab) in the trial. Over 430
patients have received axalimogene filolisbac, and approximately 1,259 doses have been delivered across multiple trials in HPV-associated
cancers, to date, and this is the first time we have seen this type of event. New guidelines for the early detection and treatment
of such rare events were agreed to with the FDA and will be implemented for all axalimogene filolisbac programs. Enrollment and
dosing in all other Advaxis and durvalumab clinical programs were not affected by the clinical hold. On July 13, 2018, we announced
that the FDA lifted its clinical hold for this trial.
BMS
Collaboration
We
entered into a clinical development collaboration agreement with Bristol-Myers Squibb to evaluate their PD-1 immune checkpoint
inhibitor, OPDIVO® (nivolumab), in combination with axalimogene filolisbac as a potential treatment option for
women with metastatic cervical cancer. The ADVANCE trial was planned to evaluate this combination regimen in women with persistent,
recurrent or metastatic (squamous or non-squamous cell) carcinoma of the cervix who have failed at least one prior line of systemic
chemotherapy. Under the terms of the agreement, each party would bear its own internal costs and provide its immunotherapy agents.
This trial has not yet been initiated as the Company is seeking a U.S. and/or European partner to fund the cervical cancer program.
If a partner is not found, the study will not be initiated.
Head-and-Neck
Cancer
We
have entered into a clinical trial collaboration agreement with MedImmune to collaborate on a Phase 1/2, open-label, multicenter,
two part trial to evaluate safety and efficacy of axalimogene filolisbac, in combination with durvalumab (MEDI4736), for patients
with metastatic squamous or non-squamous carcinoma of the cervix and metastatic HPV-associated squamous cell carcinoma of the
head and neck, or SCCHN. Part 1 of this trial is complete, and we have commenced enrollment in the Part A (20 patients with SCCHN)
and B (90 patients with cervical cancer) expansion phases; however, this trial was placed on clinical hold as detailed above.
New guidelines for the early detection and treatment of such rare events were agreed to with the FDA and will be implemented for
this combination study. On July 13, 2018, we announced that FDA lifted its clinical hold for this trial.
We
are evaluating opportunities to conduct a capital-efficient trial evaluating axalimogene filolisbac in head-and-neck cancer and
are in discussions with third parties about a potential study.
Company
Information
We
were originally incorporated in the State of Colorado on June 5, 1987 under the name Great Expectations, Inc. We were a publicly-traded
“shell” company without any business until November 12, 2004 when we acquired Advaxis, Inc., a Delaware corporation,
through a Share Exchange and Reorganization Agreement, dated as of August 25, 2004, which we refer to as the Share Exchange, by
and among Advaxis, the stockholders of Advaxis and us. As a result of the Share Exchange, Advaxis became our wholly owned subsidiary
and our sole operating company. On December 23, 2004, we amended and restated our articles of incorporation and changed our name
to Advaxis, Inc. On June 6, 2006, our stockholders approved the reincorporation of our company from Colorado to Delaware by merging
the Colorado entity into our wholly owned Delaware subsidiary. Our date of inception, for financial statement purposes, is March
1, 2002 and we were uplisted to Nasdaq in 2013. Our common stock is traded on the Nasdaq Global Select Market under the symbol
“ADXS.”
Our
principal executive offices are located at 305 College Road East, Princeton, New Jersey 08540 and our telephone number is (609)
452-9813. We maintain a corporate website at www.advaxis.com which contains descriptions of our technology, our product candidates
and the development status of each drug. We are not including the information on our website as a part of, nor incorporating it
by reference into, this prospectus supplement or the accompanying prospectus. For further information regarding us and our financial
information, you should refer to our recent filings with the SEC. See “Where You Can Find More Information” and “Incorporation
of Certain Information by Reference.”
USE
OF PROCEEDS
We
intend to use the net proceeds from the sale of any securities offered under this prospectus for general corporate purposes unless
otherwise indicated in the applicable prospectus supplement. General corporate purposes may include research and development costs,
including the conduct of one or more clinical trials, potential strategic acquisitions of complementary businesses, services or
technologies, expansion of our technology infrastructure and capabilities, working capital, capital expenditures and general corporate
purposes. We may temporarily invest the net proceeds in a variety of capital preservation instruments, including investment grade,
interest bearing instruments and U.S. government securities, until they are used for their stated purpose. We have not determined
the amount of net proceeds to be used specifically for such purposes. As a result, management will retain broad discretion over
the allocation of net proceeds.
RATIO
OF EARNINGS TO FIXED CHARGES
The
following table sets forth our ratio of earnings to fixed charges for the periods shown. You should read this table in conjunction
with the financial statements and notes incorporated by reference in this prospectus. As of the date of this prospectus, we have
no preferred shares outstanding and paid no dividends on preferred shares during the periods indicated. Therefore, the ratios
of earnings to combined fixed charges and preferred dividends are the same as the ratios of earnings to fixed charges presented
below.
|
|
|
|
Years
Ended October 31,
|
|
|
|
|
|
|
|
Nine
Months
Ended
|
|
|
|
|
|
2017
|
|
|
|
2016
|
|
|
|
2015
|
|
|
|
2014
|
|
|
|
2013
|
|
|
|
July
31, 2018
|
|
|
Ratio
of earnings (loss) to fixed charges
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
|
|
N/A
|
|
For
purposes of calculating the ratios in the table above, earnings consist of net loss before income taxes. Fixed charges include
interest expense on indebtedness and an estimate of the interest expense within rental expense.
Due
to our net losses for the years ended October 31, 2017, 2016, 2015, 2014 and 2013 and for
the nine months ended July 31, 2018, earnings were insufficient to cover fixed charges for such periods and we are unable
to disclose a ratio of earnings to fixed charges for such periods. The dollar amount of the deficiency in earnings available for
fixed charges for the years ended October 31, 2017, 2016, 2015, 2014 and 2013 and for the
nine months ended July 31, 2018 was approximately $97.8 million, $76.1 million, $48.6 million, $18.9 million, $20.7 million
and $47.8 million, respectively.
SECURITIES
WE MAY OFFER
We
may issue from time to time, in one or more offerings, the following securities:
|
|
●
|
shares
of common stock;
|
|
|
●
|
shares
of preferred stock;
|
|
|
●
|
warrants
for the purchase of common stock, preferred stock or debt securities; and
|
|
|
●
|
units
consisting of any combination of the other types of securities described in this prospectus.
|
This
prospectus contains summary descriptions of the securities we may offer from time to time. These summary descriptions are not
meant to be complete descriptions of each security. The particular terms of any security, including the offering price and the
net proceeds to us, will be described in the applicable prospectus supplement.
DESCRIPTION
OF CAPITAL STOCK
The
following description of our common stock and preferred stock, together with the additional information we include in any applicable
prospectus supplements, summarizes the material terms and provisions of the common stock and preferred stock that we may offer
under this prospectus. The following description of our capital stock does not purport to be complete and is subject to, and qualified
in its entirety by, our Amended and Restated Certificate of Incorporation and our Amended and Restated Bylaws, which are exhibits
to the registration statement of which this prospectus forms a part, and by applicable law. We refer in this section to our Amended
and Restated Certificate of Incorporation as our “certificate of incorporation”, and we refer to our Amended and Restated
Bylaws as our “bylaws.” The terms of our common stock and preferred stock may also be affected by Delaware law.
Authorized
Capital Stock
Under
our certificate of incorporation, we are authorized to issue a total of 95,000,000 shares of common stock, par value $0.001 per
share, and 5,000,000 shares of “blank check” preferred stock, par value $0.001 per share. As of August 22, 2018, we
had issued and outstanding 52,823,483 shares of our common stock and no shares of preferred stock outstanding. There were approximately
93 holders of record.
Common
Stock
Holders
of our common stock are entitled to one vote for each share held of record on all matters submitted to a vote of shareholders
and do not have any cumulative voting rights. Holders of our common stock are entitled to receive ratably any dividends declared
by the board of directors out of funds legally available for that purpose, subject to any preferential dividend rights of any
outstanding preferred stock. Our common stock has no preemptive rights, conversion rights or other subscription rights or redemption
or sinking fund provisions. In the event of our liquidation, dissolution or winding up, holders of our common stock will be entitled
to share ratably in all assets remaining after payment of all debts and other liabilities and any liquidation preference of any
outstanding preferred stock. All outstanding shares are fully-paid and non-assessable.
Listing
Our
common stock is listed on the NASDAQ Global Select Market under the symbol “ADXS.” On August 22, 2018, the closing
price for our common stock, as reported on the NASDAQ Global Select Market, was $1.47 per share.
Transfer
Agent
The
transfer agent and registrar for our common stock is Continental Stock Transfer and Trust Company, 17 Battery Place, 8th Floor,
New York, NY 10004.
Preferred
Stock
Our
board of directors is authorized, without action by the stockholders, to designate and issue up to an aggregate of 5,000,000 shares
of preferred stock in one or more series. Our board of directors can designate the rights, preferences and privileges of the shares
of each series and any of its qualifications, limitations or restrictions. Our board of directors may authorize the issuance of
preferred stock with voting or conversion rights that could adversely affect the voting power or other rights of the holders of
common stock.
The
issuance of preferred stock, while providing flexibility in connection with possible future financings and acquisitions and other
corporate purposes could, under certain circumstances, have the effect of restricting dividends on our common stock, diluting
the voting power of our common stock, impairing the liquidation rights of our common stock, or delaying, deferring or preventing
a change in control of our company, which might harm the market price of our common stock. See also “Antitakeover Effects
of Delaware Law and Provisions of our Amended and Restated Certificate of Incorporation and Amended and Restated By-laws—Provisions
of our Amended and Restated Certificate of Incorporation and Amended and Restated By-laws—Undesignated preferred stock”
below.
If
a specific series of preferred stock is offered under this prospectus, we will describe the terms of the preferred stock in the
prospectus supplement for such offering and will file a copy of the certificate establishing the terms of the preferred stock
with the SEC. To the extent required, this description will include:
|
|
●
|
the
title and stated value;
|
|
|
●
|
the
number of shares offered, the liquidation preference per share and the purchase price;
|
|
|
●
|
the
liquidation preference per share;
|
|
|
●
|
the
dividend rate(s), period(s) and/or payment date(s), or method(s) of calculation for such
dividends;
|
|
|
●
|
whether
dividends will be cumulative or non-cumulative and, if cumulative, the date from which
dividends will accumulate;
|
|
|
●
|
the
procedures for any auction and remarketing, if any;
|
|
|
●
|
the
provisions for a sinking fund, if any;
|
|
|
●
|
the
provisions for redemption or repurchase, if applicable, and any restrictions on our ability
to exercise those redemption and repurchase rights;
|
|
|
●
|
any
listing of the preferred stock on any securities exchange or market;
|
|
|
●
|
whether
the preferred stock will be convertible into our common stock, and, if applicable, the
conversion price (or how it will be calculated) and conversion period;
|
|
|
●
|
whether
the preferred stock will be exchangeable into debt securities, and, if applicable, the
exchange price (or how it will be calculated) and exchange period;
|
|
|
●
|
voting
rights, if any, of the preferred stock;
|
|
|
●
|
preemptive
rights, if any, of the preferred stock;
|
|
|
●
|
a
discussion of any material and/or special U.S. federal income tax considerations applicable
to the preferred stock;
|
|
|
●
|
restrictions
on transfer, sale or other assignment, if any;
|
|
|
●
|
the
relative ranking and preferences of the preferred stock as to dividend rights and rights
upon liquidation, dissolution or winding up of the affairs of Advaxis; and
|
|
|
●
|
any
material limitations on issuance of any class or series of preferred stock ranking senior
to or on a parity with the series of preferred stock as to dividend rights and rights
upon liquidation, dissolution or winding up of Advaxis.
|
Dividends
Subject
to the dividend rights of the holders of any outstanding series of preferred stock, holders of our common stock are entitled to
receive ratably such dividends and other distributions of cash or any other right or property as may be declared by our board
of directors out of our assets or funds legally available for such dividends or distributions.
Voting
Rights
The
holders of our common stock are entitled to one vote for each share held of record on each matter submitted to a vote of stockholders.
Holders of our common stock do not have a cumulative voting right, which means that the holders of more than one-half of the outstanding
shares of common stock, subject to the rights of the holders of the preferred stock, if any, can elect all of our directors, if
they choose to do so. In this event, the holders of the remaining shares of common stock would not be able to elect any directors.
Except as otherwise required by Delaware law, and subject to the rights of the holders of preferred stock, if any, all stockholder
action is taken by the vote of a majority of the outstanding shares of common stock voting as a single class present at a meeting
of stockholders at which a quorum consisting of one-third of the outstanding shares of common stock is present in person or proxy.
Liquidation
and Dissolution
In
the event of any voluntary or involuntary liquidation, dissolution or winding up of our affairs, holders of common stock would
be entitled to share ratably in our assets that are legally available for distribution to stockholders after payment of liabilities.
If we have any preferred stock outstanding at such time, holders of the preferred stock may be entitled to distributions and/or
liquidation preferences. In either such case, we must pay the applicable distribution to the holders of our preferred stock (if
any) before we may pay distributions to the holders of common stock.
Anti-Takeover
Provisions
Delaware
Law
We
are subject to Section 203 of the Delaware General Corporation Law, or Section 203. This provision generally prohibits a Delaware
corporation from engaging in any business combination with any interested stockholder for a period of three years following the
date the stockholder became an interested stockholder, unless:
|
|
●
|
prior
to such date, the board of directors approved either the business combination or the
transaction that resulted in the stockholder becoming an interested stockholder;
|
|
|
●
|
upon
consummation of the transaction that resulted in the stockholder becoming an interested
stockholder, the interested stockholder owned at least 85% of the voting stock of the
corporation outstanding at the time the transaction commenced, excluding for purposes
of determining the number of shares outstanding those shares owned by persons who are
directors and also officers and by employee stock plans in which employee participants
do not have the right to determine confidentially whether shares held subject to the
plan will be tendered in a tender or exchange offer; or
|
|
|
●
|
on
or subsequent to such date, the business combination is approved by the board of directors
and authorized at an annual meeting or special meeting of stockholders and not by written
consent, by the affirmative vote of at least 66-2/3% of the outstanding voting stock
that is not owned by the interested stockholder.
|
Section
203 defines a business combination to include:
|
|
●
|
any
merger or consolidation involving the corporation and the interested stockholder;
|
|
|
●
|
any
sale, transfer, pledge or other disposition of 10% or more of the assets of the corporation
involving the interested stockholder;
|
|
|
●
|
subject
to certain exceptions, any transaction that results in the issuance or transfer by the
corporation of any stock of the corporation to the interested stockholder;
|
|
|
●
|
any
transaction involving the corporation that has the effect of increasing the proportionate
share of the stock of any class or series of the corporation beneficially owned by the
interested stockholder; or
|
|
|
●
|
the
receipt by the interested stockholder of the benefit of any loans, advances, guarantees,
pledges or other financial benefits provided by or through the corporation.
|
In
general, Section 203 defines an “interested stockholder” as any entity or person beneficially owning 15% or more of
the outstanding voting stock of a corporation, or an affiliate or associate of the corporation and was the owner of 15% or more
of the outstanding voting stock of a corporation at any time within three years prior to the time of determination of interested
stockholder status; and any entity or person affiliated with or controlling or controlled by such entity or person.
These
statutory provisions could delay or frustrate the removal of incumbent directors or a change in control of our company. They could
also discourage, impede, or prevent a merger, tender offer, or proxy contest, even if such event would be favorable to the interests
of stockholders.
Amended
and Restated Certificate of Incorporation and Bylaw Provisions
Our
amended and restated certificate of incorporation and bylaws contain provisions that could have the effect of discouraging potential
acquisition proposals or making a tender offer or delaying or preventing a change in control, including changes a stockholder
might consider favorable. In particular, the certificate of incorporation and bylaws, as applicable, among other things:
|
|
●
|
provide
our board of directors with the ability to alter its bylaws without stockholder approval;
and
|
|
|
●
|
provide
that vacancies on our board of directors may be filled by a majority of directors in
office, although less than a quorum.
|
Such
provisions may have the effect of discouraging a third-party from acquiring us, even if doing so would be beneficial to our stockholders.
These provisions are intended to enhance the likelihood of continuity and stability in the composition of our board of directors
and in the policies formulated by them, and to discourage some types of transactions that may involve an actual or threatened
change in control of our company. These provisions are designed to reduce our vulnerability to an unsolicited acquisition proposal
and to discourage some tactics that may be used in proxy fights. We believe that the benefits of increased protection of our potential
ability to negotiate with the proponent of an unfriendly or unsolicited proposal to acquire or restructure our company outweigh
the disadvantages of discouraging such proposals because, among other things, negotiation of such proposals could result in an
improvement of their terms. However, these provisions could have the effect of discouraging others from making tender offers for
our shares that could result from actual or rumored takeover attempts. These provisions also may have the effect of preventing
changes in our management.
DESCRIPTION
OF DEBT SECURITIES
The
paragraphs below describe the general terms and provisions of the debt securities we may issue. When we offer to sell a particular
series of debt securities, we will describe the specific terms of the securities in a supplement to this prospectus, including
any additional covenants or changes to existing covenants relating to such series. The prospectus supplement also will indicate
whether the general terms and provisions described in this prospectus apply to a particular series of debt securities. You should
read the actual indenture if you do not fully understand a term or the way we use it in this prospectus.
We
may offer senior or subordinated debt securities. Each series of debt securities may have different terms. The senior debt securities
will be issued under one or more senior indentures, dated as of a date prior to such issuance, between us and the trustee identified
in the applicable prospectus supplement, as amended or supplemented from time to time. We will refer to any such indenture throughout
this prospectus as the “senior indenture.” Any subordinated debt securities will be issued under one or more separate
indentures, dated as of a date prior to such issuance, between us and the trustee identified in the applicable prospectus supplement,
as amended or supplemented from time to time. We will refer to any such indenture throughout this prospectus as the “subordinated
indenture” and to the trustee under the senior or subordinated indenture as the “trustee.” The senior indenture
and the subordinated indenture are sometimes collectively referred to in this prospectus as the “indentures.” The
indentures will be subject to and governed by the Trust Indenture Act of 1939, as amended. We included copies of the forms of
the indentures as exhibits to our registration statement and they are incorporated into this prospectus by reference.
If
we issue debt securities at a discount from their principal amount, then, for purposes of calculating the aggregate initial offering
price of the offered securities issued under this prospectus, we will include only the initial offering price of the debt securities
and not the principal amount of the debt securities.
We
have summarized below the material provisions of the indentures and the debt securities, or indicated which material provisions
will be described in the related prospectus supplement. The prospectus supplement relating to any particular securities offered
will describe the specific terms of the securities, which may be in addition to or different from the general terms summarized
in this prospectus. Because the summary in this prospectus and in any prospectus supplement does not contain all of the information
that you may find useful, you should read the documents relating to the securities that are described in this prospectus or in
any applicable prospectus supplement. Please read “Where You Can Find More Information” to find out how you can obtain
a copy of those documents. Except as otherwise indicated, the terms of the indentures are identical. As used under this caption,
the term “debt securities” includes the debt securities being offered by this prospectus and all other debt securities
issued by us under the indentures.
General
The
indentures:
|
|
●
|
do
not limit the amount of debt securities that we may issue;
|
|
|
●
|
allow
us to issue debt securities in one or more series;
|
|
|
●
|
do
not require us to issue all of the debt securities of a series at the same time;
|
|
|
●
|
allow
us to reopen a series to issue additional debt securities without the consent of the
holders of the debt securities of such series; and
|
|
|
●
|
provide
that the debt securities will be unsecured, except as may be set forth in the applicable
prospectus supplement.
|
Unless
we give you different information in the applicable prospectus supplement, the senior debt securities will be unsubordinated obligations
and will rank equally with all of our other senior unsecured and unsubordinated indebtedness. Payments on the subordinated debt
securities will be subordinated to the prior payment in full of all of our senior indebtedness, as described under “Description
of Debt Securities — Subordination” and in the applicable prospectus supplement.
Each
indenture provides that we may, but need not, designate more than one trustee under an indenture. Any trustee under an indenture
may resign or be removed and a successor trustee may be appointed to act with respect to the series of debt securities administered
by the resigning or removed trustee. If two or more persons are acting as trustee with respect to different series of debt securities,
each trustee shall be a trustee of a trust under the applicable indenture separate and apart from the trust administered by any
other trustee. Except as otherwise indicated in this prospectus, any action described in this prospectus to be taken by each trustee
may be taken by each trustee with respect to, and only with respect to, the one or more series of debt securities for which it
is trustee under the applicable indenture.
The
prospectus supplement for each offering will provide the following terms, where applicable:
|
|
●
|
the
title of the debt securities and whether they are senior or subordinated;
|
|
|
●
|
the
aggregate principal amount of the debt securities being offered, the aggregate principal
amount of the debt securities outstanding as of the most recent practicable date and
any limit on their aggregate principal amount, including the aggregate principal amount
of debt securities authorized;
|
|
|
●
|
the
price at which the debt securities will be issued, expressed as a percentage of the principal
and, if other than the principal amount thereof, the portion of the principal amount
thereof payable upon declaration of acceleration of the maturity thereof or, if applicable,
the portion of the principal amount of such debt securities that is convertible into
common stock or other securities of ours or the method by which any such portion shall
be determined;
|
|
|
●
|
if
convertible, the terms on which such debt securities are convertible, including the initial
conversion price or rate and the conversion period and any applicable limitations on
the ownership or transferability of common stock or other securities of ours received
on conversion;
|
|
|
●
|
the
date or dates, or the method for determining the date or dates, on which the principal
of the debt securities will be payable;
|
|
|
●
|
the
fixed or variable interest rate or rates of the debt securities, or the method by which
the interest rate or rates is determined;
|
|
|
●
|
the
date or dates, or the method for determining the date or dates, from which interest will
accrue;
|
|
|
●
|
the
dates on which interest will be payable;
|
|
|
●
|
the
record dates for interest payment dates, or the method by which such dates will be determined;
|
|
|
●
|
the
persons to whom interest will be payable;
|
|
|
●
|
the
basis upon which interest will be calculated if other than that of a 360-day year of
twelve 30-day months;
|
|
|
●
|
any
make-whole amount, which is the amount in addition to principal and interest that is
required to be paid to the holder of a debt security as a result of any optional redemption
or accelerated payment of such debt security, or the method for determining the make-whole
amount;
|
|
|
●
|
the
place or places where the principal of, and any premium or make-whole amount, and interest
on, the debt securities will be payable;
|
|
|
●
|
where
the debt securities may be surrendered for registration of transfer or conversion or
exchange;
|
|
|
●
|
where
notices or demands to or upon us in respect of the debt securities and the applicable
indenture may be served;
|
|
|
●
|
the
times, prices and other terms and conditions upon which we may redeem the debt securities;
|
|
|
●
|
any
obligation we have to redeem, repay or purchase the debt securities pursuant to any sinking
fund or analogous provision or at the option of holders of the debt securities, and the
times and prices at which we must redeem, repay or purchase the debt securities as a
result of such obligation;
|
|
|
●
|
the
currency or currencies in which the debt securities are denominated and payable if other
than U.S. dollars, which may be a foreign currency or units of two or more foreign currencies
or a composite currency or currencies and the terms and conditions relating thereto,
and the manner of determining the equivalent of such foreign currency in U.S. dollars;
|
|
|
●
|
whether
the principal of, and any premium or make-whole amount, or interest on, the debt securities
of the series are to be payable, at our election or at the election of a holder, in a
currency or currencies other than that in which the debt securities are denominated or
stated to be payable, and other related terms and conditions;
|
|
|
●
|
whether
the amount of payments of principal of, and any premium or make-whole amount, or interest
on, the debt securities may be determined according to an index, formula or other method
and how such amounts will be determined;
|
|
|
●
|
whether
the debt securities will be in registered form, bearer form, or both, and (i) if in registered
form, the person to whom any interest shall be payable, if other than the person in whose
name the security is registered at the close of business on the regular record date for
such interest, or (ii) if in bearer form, the manner in which, or the person to whom,
any interest on the security shall be payable if otherwise than upon presentation and
surrender upon maturity;
|
|
|
●
|
any
restrictions applicable to the offer, sale or delivery of securities in bearer form and
the terms upon which securities in bearer form of the series may be exchanged for securities
in registered form of the series and vice versa, if permitted by applicable laws and
regulations;
|
|
|
●
|
whether
any debt securities of the series are to be issuable initially in temporary global form
and whether any debt securities of the series are to be issuable in permanent global
form with or without coupons and, if so, whether beneficial owners of interests in any
such permanent global security may, or shall be required to, exchange their interests
for other debt securities of the series, and the manner in which interest shall be paid;
|
|
|
●
|
the
identity of the depositary for securities in registered form, if such series are to be
issuable as a global security;
|
|
|
●
|
the
date as of which any debt securities in bearer form or in temporary global form shall
be dated if other than the original issuance date of the first security of the series
to be issued;
|
|
|
●
|
the
applicability, if any, of the defeasance and covenant defeasance provisions described
in this prospectus or in the applicable indenture;
|
|
|
●
|
whether
and under what circumstances we will pay any additional amounts on the debt securities
in respect of any tax, assessment or governmental charge and, if so, whether we will
have the option to redeem the debt securities in lieu of making such a payment;
|
|
|
●
|
whether
and under what circumstances the debt securities being offered are convertible into common
stock or other securities of ours, as the case may be, including the conversion price
or rate and the manner or calculation thereof;
|
|
|
●
|
the
circumstances, if any, specified in the applicable prospectus supplement, under which
beneficial owners of interests in the global security may obtain definitive debt securities
and the manner in which payments on a permanent global debt security will be made if
any debt securities are issuable in temporary or permanent global form;
|
|
|
●
|
any
provisions granting special rights to holders of securities upon the occurrence of such
events as specified in the applicable prospectus supplement;
|
|
|
●
|
if
the debt securities of such series are to be issuable in definitive form only upon receipt
of certain certificates or other documents or satisfaction of other conditions, then
the form and/or terms of such certificates, documents or conditions;
|
|
|
●
|
the
name of the applicable trustee and the nature of any material relationship with us or
any of our affiliates, and the percentage of debt securities of the class necessary to
require the trustee to take action;
|
|
|
●
|
any
deletions from, modifications of or additions to our events of default or covenants with
regard to such debt securities and any change in the right of any trustee or any of the
holders to declare the principal amount of any of such debt securities due and payable;
|
|
|
●
|
applicable
CUSIP numbers; and
|
|
|
●
|
any
other terms of such debt securities not inconsistent with the provisions of the applicable
indenture.
|
We
may issue debt securities that provide for less than the entire principal amount thereof to be payable upon declaration of acceleration
of the maturity of the debt securities. We refer to any such debt securities throughout this prospectus as “original issue
discount securities.” The applicable prospectus supplement will describe the U.S. federal income tax consequences and other
relevant considerations applicable to original issue discount securities.
We
also may issue indexed debt securities. Payments of principal of, and premium and interest on, indexed debt securities are determined
with reference to the rate of exchange between the currency or currency unit in which the debt security is denominated and any
other currency or currency unit specified by us, to the relationship between two or more currencies or currency units or by other
similar methods or formulas specified in the prospectus supplement.
Except
as described under “Description of Debt Securities — Merger, Consolidation or Sale of Assets” or as may be set
forth in any prospectus supplement, the debt securities will not contain any provisions that (i) would limit our ability to incur
indebtedness or (ii) would afford holders of debt securities protection in the event of (a) a highly leveraged or similar transaction
involving us, or (b) a change of control or reorganization, restructuring, merger or similar transaction involving us that may
adversely affect the holders of the debt securities. In the future, we may enter into transactions, such as the sale of all or
substantially all of our assets or a merger or consolidation, that may have an adverse effect on our ability to service our indebtedness,
including the debt securities, by, among other things, substantially reducing or eliminating our assets.
Our
governing instruments do not define the term “substantially all” as it relates to the sale of assets. Additionally,
Delaware cases interpreting the term “substantially all” rely upon the facts and circumstances of each particular
case. Consequently, to determine whether a sale of “substantially all” of our assets has occurred, a holder of debt
securities must review the financial and other information that we have disclosed to the public.
We
will provide you with more information in the applicable prospectus supplement regarding any deletions, modifications, or additions
to the events of default or covenants that are described below, including any addition of a covenant or other provision providing
event risk or similar protection.
Payment
Unless
we give you different information in the applicable prospectus supplement, the principal of, and any premium or make-whole amount,
and interest on, any series of the debt securities will be payable at the corporate trust office of the trustee. We will provide
you with the address of the trustee in the applicable prospectus supplement. We may also pay interest by mailing a check to the
address of the person entitled to it as it appears in the applicable register for the debt securities or by wire transfer of funds
to that person at an account maintained within the United States.
All
monies that we pay to a paying agent or a trustee for the payment of the principal of, and any premium or make-whole amount, or
interest on, any debt security will be repaid to us if unclaimed at the end of two years after the obligation underlying payment
becomes due and payable. After funds have been returned to us, the holder of the debt security may look only to us for payment,
without payment of interest for the period which we hold the funds.
Denomination,
Interest, Registration and Transfer
Unless
otherwise described in the applicable prospectus supplement, the debt securities of any series will be issuable in denominations
of $1,000 and integral multiples of $1,000.
Subject
to the limitations imposed upon debt securities that are evidenced by a computerized entry in the records of a depository company
rather than by physical delivery of a note, a holder of debt securities of any series may:
|
|
●
|
exchange
them for any authorized denomination of other debt securities of the same series and
of a like aggregate principal amount and kind upon surrender of such debt securities
at the corporate trust office of the applicable trustee or at the office of any transfer
agent that we designate for such purpose; and
|
|
|
●
|
surrender
them for registration of transfer or exchange at the corporate trust office of the applicable
trustee or at the office of any transfer agent that we designate for such purpose.
|
Every
debt security surrendered for registration of transfer or exchange must be duly endorsed or accompanied by a written instrument
of transfer satisfactory to the applicable trustee or transfer agent. Payment of a service charge will not be required for any
registration of transfer or exchange of any debt securities, but we or the trustee may require payment of a sum sufficient to
cover any tax or other governmental charge payable in connection therewith. If in addition to the applicable trustee, the applicable
prospectus supplement refers to any transfer agent initially designated by us for any series of debt securities, we may at any
time rescind the designation of any such transfer agent or approve a change in the location through which any such transfer agent
acts, except that we will be required to maintain a transfer agent in each place of payment for such series. We may at any time
designate additional transfer agents for any series of debt securities.
Neither
we, nor any trustee, will be required to:
|
|
●
|
issue,
register the transfer of or exchange debt securities of any series during a period beginning
at the opening of business 15 days before the day that the notice of redemption of any
debt securities selected for redemption is mailed and ending at the close of business
on the day of such mailing;
|
|
|
●
|
register
the transfer of or exchange any debt security, or portion thereof, so selected for redemption,
in whole or in part, except the unredeemed portion of any debt security being redeemed
in part; and
|
|
|
●
|
issue,
register the transfer of or exchange any debt security that has been surrendered for
repayment at the option of the holder, except the portion, if any, of such debt security
not to be so repaid.
|
Merger,
Consolidation or Sale of Assets
The
indentures provide that we may, without the consent of the holders of any outstanding debt securities, (i) consolidate with, (ii)
sell, lease or convey all or substantially all of our assets to, or (iii) merge with or into, any other entity provided that:
|
|
●
|
either
we are the continuing entity, or the successor entity, if other than us, assumes the
obligations (a) to pay the principal of, and any premium or make-whole amount, and interest
on, all of the debt securities and (b) to duly perform and observe all of the covenants
and conditions contained in each indenture;
|
|
|
●
|
after
giving effect to the transaction, there is no event of default under the indentures and
no event which, after notice or the lapse of time, or both, would become such an event
of default, occurs and continues; and
|
|
|
●
|
an
officers’ certificate and legal opinion covering such conditions are delivered
to each applicable trustee.
|
Covenants
Existence.
Except as described under “Description of Debt Securities — Merger, Consolidation or Sale of Assets,” the indentures
require us to do or cause to be done all things necessary to preserve and keep in full force and effect our existence, rights
and franchises. However, the indentures do not require us to preserve any right or franchise if we determine that any right or
franchise is no longer desirable in the conduct of our business.
Payment
of taxes and other claims. The indentures require us to pay, discharge or cause to be paid or discharged, before they become
delinquent (i) all taxes, assessments and governmental charges levied or imposed on us, and (ii) all lawful claims for labor,
materials and supplies which, if unpaid, might by law become a lien upon our property. However, we will not be required to pay,
discharge or cause to be paid or discharged any such tax, assessment, charge or claim whose amount, applicability or validity
is being contested in good faith by appropriate proceedings.
Provision
of financial information. The indentures require us to (i) within 15 days of each of the respective dates by which we are
required to file our annual reports, quarterly reports and other documents with the SEC, file with the trustee copies of the annual
report, quarterly report and other documents that we file with the SEC under Section 13 or 15(d) of the Exchange Act, (ii) file
with the trustee and the SEC any additional information, documents and reports regarding compliance by us with the conditions
and covenants of the indentures, as required, (iii) within 30 days after the filing with the trustee, mail to all holders of debt
securities, as their names and addresses appear in the applicable register for such debt securities, without cost to such holders,
summaries of any documents and reports required to be filed by us pursuant to (i) and (ii) above, and (iv) supply, promptly upon
written request and payment of the reasonable cost of duplication and delivery, copies of such documents to any prospective holder.
Additional
covenants. The applicable prospectus supplement will set forth any of our additional covenants relating to any series of debt
securities.
Events
of Default, Notice and Waiver
Unless
the applicable prospectus supplement states otherwise, when we refer to “events of default” as defined in the indentures
with respect to any series of debt securities, we mean:
|
|
●
|
default
in the payment of any installment of interest on any debt security of such series continuing
for 30 days;
|
|
|
●
|
default
in the payment of principal of, or any premium or make-whole amount on, any debt security
of such series for five business days at its stated maturity;
|
|
|
●
|
default
in making any sinking fund payment as required for any debt security of such series for
five business days;
|
|
|
●
|
default
in the performance or breach of any covenant or warranty in the debt securities or in
the indenture by us continuing for 60 days after written notice as provided in the applicable
indenture, but not of a covenant added to the indenture solely for the benefit of a series
of debt securities issued thereunder other than such series;
|
|
|
●
|
a
default under any bond, debenture, note, mortgage, indenture or instrument:
|
|
|
(a)
|
having
an aggregate principal amount of at least $30,000,000; or
|
|
|
(b)
|
under
which there may be issued, secured or evidenced any existing or later created indebtedness
for money borrowed by us, if we are directly responsible or liable as obligor or guarantor,
|
if
the default results in the indebtedness becoming or being declared due and payable prior to the date it otherwise would have,
without such indebtedness having been discharged, or such acceleration having been rescinded or annulled, within 30 days after
notice to the issuing company specifying such default. Such notice shall be given to us by the trustee, or to us and the trustee
by the holders of at least 10% in principal amount of the outstanding debt securities of that series. The written notice shall
specify such default and require us to cause such indebtedness to be discharged or cause such acceleration to be rescinded or
annulled and shall state that such notice is a “Notice of Default” under such indenture;
|
|
●
|
bankruptcy,
insolvency or reorganization, or court appointment of a receiver, liquidator or trustee
of us; and
|
|
|
●
|
any
other event of default provided with respect to a particular series of debt securities.
|
If
an event of default occurs and is continuing with respect to debt securities of any series outstanding, then the applicable trustee
or the holders of 25% or more in principal amount of the debt securities of that series will have the right to declare the principal
amount of all the debt securities of that series to be due and payable. If the debt securities of that series are original issue
discount securities or indexed securities, then the applicable trustee or the holders of 25% or more in principal amount of the
debt securities of that series will have the right to declare the portion of the principal amount as may be specified in the terms
thereof to be due and payable. However, at any time after such a declaration of acceleration has been made, but before a judgment
or decree for payment of the money due has been obtained by the applicable trustee, the holders of at least a majority in principal
amount of outstanding debt securities of such series or of all debt securities then outstanding under the applicable indenture
may rescind and annul such declaration and its consequences if:
|
|
●
|
we
have deposited with the applicable trustee all required payments of the principal, any
premium or make-whole amount, interest and, to the extent permitted by law, interest
on overdue installment of interest, plus applicable fees, expenses, disbursements and
advances of the applicable trustee; and
|
|
|
●
|
all
events of default, other than the non-payment of accelerated principal, or a specified
portion thereof, and any premium or make-whole amount, have been cured or waived.
|
The
indentures also provide that the holders of at least a majority in principal amount of the outstanding debt securities of any
series or of all debt securities then outstanding under the applicable indenture may, on behalf of all holders, waive any past
default with respect to such series and its consequences, except a default:
|
|
●
|
in
the payment of the principal, any premium or make-whole amount, or interest;
|
|
|
●
|
in
respect of a covenant or provision contained in the applicable indenture that cannot
be modified or amended without the consent of the holders of the outstanding debt security
that is affected by the default; or
|
|
|
●
|
in
respect of a covenant or provision for the benefit or protection of the trustee, without
its express written consent.
|
The
indentures require each trustee to give notice to the holders of debt securities within 90 days of a default unless such default
has been cured or waived. However, the trustee may withhold notice if specified persons of such trustee consider such withholding
to be in the interest of the holders of debt securities. The trustee may not withhold notice of a default in the payment of principal,
any premium or interest on any debt security of such series or in the payment of any sinking fund installment in respect of any
debt security of such series.
The
indentures provide that holders of debt securities of any series may not institute any proceedings, judicial or otherwise, with
respect to such indenture or for any remedy under the indenture, unless the trustee fails to act for a period of 60 days after
the trustee has received a written request to institute proceedings in respect of an event of default from the holders of 25%
or more in principal amount of the outstanding debt securities of such series, as well as an offer of indemnity reasonably satisfactory
to the trustee. However, this provision will not prevent any holder of debt securities from instituting suit for the enforcement
of payment of the principal of, and any premium or make-whole amount, and interest on, such debt securities at the respective
due dates thereof.
The
indentures provide that, subject to provisions in each indenture relating to its duties in the case of a default, a trustee has
no obligation to exercise any of its rights or powers at the request or direction of any holders of any series of debt securities
then outstanding under the indenture, unless the holders have offered to the trustee reasonable security or indemnity. The holders
of at least a majority in principal amount of the outstanding debt securities of any series or of all debt securities then outstanding
under an indenture shall have the right to direct the time, method and place of conducting any proceeding for any remedy available
to the applicable trustee, or of exercising any trust or power conferred upon such trustee. However, a trustee may refuse to follow
any direction which:
|
|
●
|
is
in conflict with any law or the applicable indenture;
|
|
|
●
|
may
involve the trustee in personal liability; or
|
|
|
●
|
may
be unduly prejudicial to the holders of debt securities of the series not joining the
proceeding.
|
Within
120 days after the close of each fiscal year, we will be required to deliver to each trustee a certificate, signed by one of our
several specified officers, stating whether or not that officer has knowledge of any default under the applicable indenture. If
the officer has knowledge of any default, the notice must specify the nature and status of the default.
Modification
of the Indentures
The
indentures provide that modifications and amendments may be made only with the consent of the affected holders of a majority in
principal amount of all outstanding debt securities issued under that indenture. However, no such modification or amendment may,
without the consent of the holders of the debt securities affected by the modification or amendment:
|
|
●
|
change
the stated maturity of the principal of, or any premium or make-whole amount on, or any
installment of principal of or interest on, any such debt security;
|
|
|
●
|
reduce
the principal amount of, the rate or amount of interest on, or any premium or make-whole
amount payable on redemption of, any such debt security;
|
|
|
●
|
reduce
the amount of principal of an original issue discount security that would be due and
payable upon declaration of acceleration of the maturity thereof or would be provable
in bankruptcy, or adversely affect any right of repayment of the holder of any such debt
security;
|
|
|
●
|
change
the place of payment or the coin or currency for payment of principal of, or any premium
or make-whole amount, or interest on, any such debt security;
|
|
|
●
|
impair
the right to institute suit for the enforcement of any payment on or with respect to
any such debt security;
|
|
|
●
|
reduce
the percentage in principal amount of any outstanding debt securities necessary to modify
or amend the applicable indenture with respect to such debt securities, to waive compliance
with particular provisions thereof or defaults and consequences thereunder or to reduce
the quorum or voting requirements set forth in the applicable indenture; and
|
|
|
●
|
modify
any of the foregoing provisions or any of the provisions relating to the waiver of particular
past defaults or covenants, except to increase the required percentage to effect such
action or to provide that some of the other provisions may not be modified or waived
without the consent of the holder of such debt security.
|
The
holders of a majority in aggregate principal amount of the outstanding debt securities of each series may, on behalf of all holders
of debt securities of that series, waive, insofar as that series is concerned, our compliance with material restrictive covenants
of the applicable indenture.
We
and our respective trustee may make modifications and amendments of an indenture without the consent of any holder of debt securities
for any of the following purposes:
|
|
●
|
to
evidence the succession of another person to us as obligor under such indenture;
|
|
|
●
|
to
add to our covenants for the benefit of the holders of all or any series of debt securities
or to surrender any right or power conferred upon us in such indenture;
|
|
|
●
|
to
add events of default for the benefit of the holders of all or any series of debt securities;
|
|
|
●
|
to
add or change any provisions of an indenture (i) to change or eliminate restrictions
on the payment of principal of, or premium or make-whole amount, or interest on, debt
securities in bearer form, or (ii) to permit or facilitate the issuance of debt securities
in uncertificated form, provided that such action shall not adversely affect the interests
of the holders of the debt securities of any series in any material respect;
|
|
|
●
|
to
change or eliminate any provisions of an indenture, provided that any such change or
elimination shall become effective only when there are no debt securities outstanding
of any series created prior thereto which are entitled to the benefit of such provision;
|
|
|
●
|
to
secure the debt securities;
|
|
|
●
|
to
establish the form or terms of debt securities of any series;
|
|
|
●
|
to
provide for the acceptance of appointment by a successor trustee or facilitate the administration
of the trusts under an indenture by more than one trustee;
|
|
|
●
|
to
cure any ambiguity, defect or inconsistency in an indenture, provided that such action
shall not adversely affect the interests of holders of debt securities of any series
issued under such indenture; and
|
|
|
●
|
to
supplement any of the provisions of an indenture to the extent necessary to permit or
facilitate defeasance and discharge of any series of such debt securities, provided that
such action shall not adversely affect the interests of the holders of the outstanding
debt securities of any series.
|
Voting
The
indentures provide that in determining whether the holders of the requisite principal amount of outstanding debt securities of
a series have given any request, demand, authorization, direction, notice, consent or waiver under the indentures or whether a
quorum is present at a meeting of holders of debt securities:
|
|
●
|
the
principal amount of an original issue discount security that shall be deemed to be outstanding
shall be the amount of the principal thereof that would be due and payable as of the
date of such determination upon declaration of acceleration of the maturity thereof;
|
|
|
●
|
the
principal amount of any debt security denominated in a foreign currency that shall be
deemed outstanding shall be the U.S. dollar equivalent, determined on the issue date
for such debt security, of the principal amount or, in the case of an original issue
discount security, the U.S. dollar equivalent on the issue date of such debt security
of the amount determined as provided in the preceding bullet point;
|
|
|
●
|
the
principal amount of an indexed security that shall be deemed outstanding shall be the
principal face amount of such indexed security at original issuance, unless otherwise
provided for such indexed security under such indenture; and
|
|
|
●
|
debt
securities owned by us or any other obligor upon the debt securities or by any affiliate
of ours or of such other obligor shall be disregarded.
|
The
indentures contain provisions for convening meetings of the holders of debt securities of a series. A meeting will be permitted
to be called at any time by the applicable trustee, and also, upon request, by us or the holders of at least 25% in principal
amount of the outstanding debt securities of such series, in any such case upon notice given as provided in such indenture. Except
for any consent that must be given by the holder of each debt security affected by the modifications and amendments of an indenture
described above, any resolution presented at a meeting or adjourned meeting duly reconvened at which a quorum is present may be
adopted by the affirmative vote of the holders of a majority of the aggregate principal amount of the outstanding debt securities
of that series represented at such meeting.
Notwithstanding
the preceding paragraph, except as referred to above, any resolution relating to a request, demand, authorization, direction,
notice, consent, waiver or other action that may be made, given or taken by the holders of a specified percentage, which is less
than a majority of the aggregate principal amount of the outstanding debt securities of a series, may be adopted at a meeting
or adjourned meeting duly reconvened at which a quorum is present by the affirmative vote of such specified percentage.
Any
resolution passed or decision taken at any properly held meeting of holders of debt securities of any series will be binding on
all holders of such series. The quorum at any meeting called to adopt a resolution, and at any reconvened meeting, will be persons
holding or representing a majority in principal amount of the outstanding debt securities of a series. However, if any action
is to be taken relating to a consent or waiver which may be given by the holders of at least a specified percentage in principal
amount of the outstanding debt securities of a series, the persons holding such percentage will constitute a quorum.
Notwithstanding
the foregoing provisions, the indentures provide that if any action is to be taken at a meeting with respect to any request, demand,
authorization, direction, notice, consent, waiver or other action that such indenture expressly provides may be made, given or
taken by the holders of a specified percentage in principal amount of all outstanding debt securities affected by such action,
or of the holders of such series and one or more additional series:
|
|
●
|
there
shall be no minimum quorum requirement for such meeting; and
|
|
|
●
|
the
principal amount of the outstanding debt securities of such series that vote in favor
of such request, demand, authorization, direction, notice, consent, waiver or other action
shall be taken account in determining whether such request, demand, authorization, direction,
notice, consent, waiver or other action has been made, given or taken under such indenture.
|
Subordination
Unless
otherwise provided in the applicable prospectus supplement, subordinated debt securities will be subject to the following subordination
provisions.
Upon
any distribution to our creditors in a liquidation, dissolution or reorganization, the payment of the principal of and interest
on any subordinated debt securities will be subordinated to the extent provided in the applicable indenture in right of payment
to the prior payment in full of all senior debt. However, our obligation to make payments of the principal of and interest on
such subordinated debt securities otherwise will not be affected. No payment of principal or interest will be permitted to be
made on subordinated debt securities at any time if a default on senior debt exists that permits the holders of such senior debt
to accelerate its maturity and the default is the subject of judicial proceedings or we receive notice of the default. After all
senior debt is paid in full and until the subordinated debt securities are paid in full, holders of subordinated debt securities
will be subrogated to the rights of holders of senior debt to the extent that distributions otherwise payable to holders of subordinated
debt securities have been applied to the payment of senior debt. The subordinated indenture will not restrict the amount of senior
debt or other indebtedness of ours. As a result of these subordination provisions, in the event of a distribution of assets upon
insolvency, holders of subordinated debt securities may recover less, ratably, than our general creditors.
The
term “senior debt” will be defined in the applicable indenture as the principal of and interest on, or substantially
similar payments to be made by us in respect of, other outstanding indebtedness, whether outstanding at the date of execution
of the applicable indenture or subsequently incurred, created or assumed. The prospectus supplement may include a description
of additional terms implementing the subordination feature.
No
restrictions will be included in any indenture relating to subordinated debt securities upon the creation of additional senior
debt.
If
this prospectus is being delivered in connection with the offering of a series of subordinated debt securities, the accompanying
prospectus supplement or the information incorporated in this prospectus by reference will set forth the approximate amount of
senior debt outstanding as of the end of our most recent fiscal quarter.
Discharge,
Defeasance and Covenant Defeasance
Unless
otherwise indicated in the applicable prospectus supplement, the indentures allow us to discharge our obligations to holders of
any series of debt securities issued under any indenture when:
|
|
●
|
either
(i) all securities of such series have already been delivered to the applicable trustee
for cancellation; or (ii) all securities of such series have not already been delivered
to the applicable trustee for cancellation but (a) have become due and payable, (b) will
become due and payable within one year, or (c) if redeemable at our option, are to be
redeemed within one year, and we have irrevocably deposited with the applicable trustee,
in trust, funds in such currency or currencies, currency unit or units or composite currency
or currencies in which such debt securities are payable, an amount sufficient to pay
the entire indebtedness on such debt securities in respect of principal and any premium
or make-whole amount, and interest to the date of such deposit if such debt securities
have become due and payable or, if they have not, to the stated maturity or redemption
date;
|
|
|
●
|
we
have paid or caused to be paid all other sums payable; and
|
|
|
●
|
an
officers’ certificate and an opinion of counsel stating the conditions to discharging
the debt securities have been satisfied has been delivered to the trustee.
|
Unless
otherwise indicated in the applicable prospectus supplement, the indentures provide that, upon our irrevocable deposit with the
applicable trustee, in trust, of an amount, in such currency or currencies, currency unit or units or composite currency or currencies
in which such debt securities are payable at stated maturity, or government obligations, or both, applicable to such debt securities,
which through the scheduled payment of principal and interest in accordance with their terms will provide money in an amount sufficient
to pay the principal of, and any premium or make-whole amount, and interest on, such debt securities, and any mandatory sinking
fund or analogous payments thereon, on the scheduled due dates therefor, the issuing company may elect either:
|
|
●
|
to
defease and be discharged from any and all obligations with respect to such debt securities;
or
|
|
|
●
|
to
be released from its obligations with respect to such debt securities under the applicable
indenture or, if provided in the applicable prospectus supplement, its obligations with
respect to any other covenant, and any omission to comply with such obligations shall
not constitute an event of default with respect to such debt securities.
|
Notwithstanding
the above, we may not elect to defease and be discharged from the obligation to pay any additional amounts upon the occurrence
of particular events of tax, assessment or governmental charge with respect to payments on such debt securities and the obligations
to register the transfer or exchange of such debt securities, to replace temporary or mutilated, destroyed, lost or stolen debt
securities, to maintain an office or agency in respect of such debt securities, or to hold monies for payment in trust.
The
indentures only permit us to establish the trust described in the paragraph above if, among other things, we have delivered to
the applicable trustee an opinion of counsel to the effect that the holders of such debt securities will not recognize income,
gain or loss for U.S. federal income tax purposes as a result of such defeasance or covenant defeasance and will be subject to
U.S. federal income tax on the same amounts, in the same manner and at the same times as would have been the case if such defeasance
or covenant defeasance had not occurred. Such opinion of counsel, in the case of defeasance, will be required to refer to and
be based upon a ruling received from or published by the Internal Revenue Service or a change in applicable U.S. federal income
tax law occurring after the date of the indenture. In the event of such defeasance, the holders of such debt securities would
be able to look only to such trust fund for payment of principal, any premium or make-whole amount, and interest.
When
we use the term “government obligations,” we mean securities that are:
|
|
●
|
direct
obligations of the United States or the government that issued the foreign currency in
which the debt securities of a particular series are payable, for the payment of which
its full faith and credit is pledged; or
|
|
|
●
|
obligations
of a person controlled or supervised by and acting as an agency or instrumentality of
the United States or other government that issued the foreign currency in which the debt
securities of such series are payable, the payment of which is unconditionally guaranteed
as a full faith and credit obligation by the United States or such other government,
which are not callable or redeemable at the option of the issuer thereof and shall also
include a depository receipt issued by a bank or trust company as custodian with respect
to any such government obligation or a specific payment of interest on or principal of
any such government obligation held by such custodian for the account of the holder of
a depository receipt. However, except as required by law, such custodian is not authorized
to make any deduction from the amount payable to the holder of such depository receipt
from any amount received by the custodian in respect of the government obligation or
the specific payment of interest on or principal of the government obligation evidenced
by such depository receipt.
|
Unless
otherwise provided in the applicable prospectus supplement, if after we have deposited funds and/or government obligations to
effect defeasance or covenant defeasance with respect to debt securities of any series, (i) the holder of a debt security of such
series is entitled to, and does, elect under the terms of the applicable indenture or the terms of such debt security to receive
payment in a currency, currency unit or composite currency other than that in which such deposit has been made in respect of such
debt security, or (ii) a conversion event occurs in respect of the currency, currency unit or composite currency in which such
deposit has been made, the indebtedness represented by such debt security will be deemed to have been, and will be, fully discharged
and satisfied through the payment of the principal of, and premium or make-whole amount, and interest on, such debt security as
they become due out of the proceeds yielded by converting the amount so deposited in respect of such debt security into the currency,
currency unit or composite currency in which such debt security becomes payable as a result of such election or such cessation
of usage based on the applicable market exchange rate.
When
we use the term “conversion event,” we mean the cessation of use of:
|
|
●
|
a
currency, currency unit or composite currency both by the government of the country that
issued such currency and for the settlement of transactions by a central bank or other
public institutions of or within the international banking community;
|
|
|
●
|
the
European Currency Unit both within the European Monetary System and for the settlement
of transactions by public institutions of or within the European Communities; or
|
|
|
●
|
any
currency unit or composite currency other than the European Currency Unit for the purposes
for which it was established.
|
Unless
otherwise provided in the applicable prospectus supplement, all payments of principal of, and any premium or make-whole amount,
and interest on, any debt security that is payable in a foreign currency that ceases to be used by its government of issuance
shall be made in U.S. dollars.
In
the event that (i) we effect covenant defeasance with respect to any debt securities and (ii) those debt securities are declared
due and payable because of the occurrence of any event of default, the amount in the currency, currency unit or composite currency
in which such debt securities are payable, and government obligations on deposit with the applicable trustee, will be sufficient
to pay amounts due on such debt securities at the time of their stated maturity but may not be sufficient to pay amounts due on
such debt securities at the time of the acceleration resulting from such event of default. However, the issuing company would
remain liable to make payments of any amounts due at the time of acceleration.
The
applicable prospectus supplement may further describe the provisions, if any, permitting such defeasance or covenant defeasance,
including any modifications to the provisions described above, with respect to the debt securities of or within a particular series.
Conversion
Rights
The
terms and conditions, if any, upon which the debt securities are convertible into common stock or other securities of ours will
be set forth in the applicable prospectus supplement. The terms will include whether the debt securities are convertible into
shares of common stock or other securities of ours, the conversion price, or manner of calculation thereof, the conversion period,
provisions as to whether conversion will be at the issuing company’s option or the option of the holders, the events requiring
an adjustment of the conversion price and provisions affecting conversion in the event of the redemption of the debt securities
and any restrictions on conversion.
No
Recourse
There
is no recourse under any obligation, covenant or agreement in the applicable indenture or with respect to any security against
any of our or our successor’s past, present or future shareholders, employees, officers or directors.
DESCRIPTION
OF WARRANTS
We
may issue warrants for the purchase of shares of our common stock or preferred stock or of debt securities. We may issue warrants
independently or together with other securities, and the warrants may be attached to or separate from any offered securities.
Each series of warrants will be issued under a separate warrant agreement to be entered into between us and the investors or a
warrant agent. The following summary of material provisions of the warrants and warrant agreements are subject to, and qualified
in their entirety by reference to, all the provisions of the warrant agreement and warrant certificate applicable to a particular
series of warrants. The terms of any warrants offered under a prospectus supplement may differ from the terms described below.
We urge you to read the applicable prospectus supplement and any related free writing prospectus, as well as the complete warrant
agreements and warrant certificates that contain the terms of the warrants.
The
particular terms of any issue of warrants will be described in the prospectus supplement relating to the issue. Those terms may
include:
|
|
●
|
the
number of shares of common stock or preferred stock purchasable upon the exercise of
warrants to purchase such shares and the price at which such number of shares may be
purchased upon such exercise;
|
|
|
●
|
the
designation, stated value and terms (including, without limitation, liquidation, dividend,
conversion and voting rights) of the series of preferred stock purchasable upon exercise
of warrants to purchase preferred stock;
|
|
|
●
|
the
principal amount of debt securities that may be purchased upon exercise of a debt warrant
and the exercise price for the warrants, which may be payable in cash, securities or
other property;
|
|
|
●
|
the
date, if any, on and after which the warrants and the related debt securities, preferred
stock or common stock will be separately transferable;
|
|
|
●
|
the
terms of any rights to redeem or call the warrants;
|
|
|
●
|
the
date on which the right to exercise the warrants will commence and the date on which
the right will expire;
|
|
|
●
|
U.S.
Federal income tax consequences applicable to the warrants; and
|
|
|
●
|
any
additional terms of the warrants, including terms, procedures, and limitations relating
to the exchange, exercise and settlement of the warrants.
|
Holders
of equity warrants will not be entitled:
|
|
●
|
to
vote, consent or receive dividends;
|
|
|
●
|
receive
notice as stockholders with respect to any meeting of stockholders for the election of
our directors or any other matter; or
|
|
|
●
|
exercise
any rights as stockholders of Advaxis.
|
Each
warrant will entitle its holder to purchase the principal amount of debt securities or the number of shares of preferred stock
or common stock at the exercise price set forth in, or calculable as set forth in, the applicable prospectus supplement. Unless
we otherwise specify in the applicable prospectus supplement, holders of the warrants may exercise the warrants at any time up
to the specified time on the expiration date that we set forth in the applicable prospectus supplement. After the close of business
on the expiration date, unexercised warrants will become void.
A
holder of warrant certificates may exchange them for new warrant certificates of different denominations, present them for registration
of transfer and exercise them at the corporate trust office of the warrant agent or any other office indicated in the applicable
prospectus supplement. Until any warrants to purchase debt securities are exercised, the holder of the warrants will not have
any rights of holders of the debt securities that can be purchased upon exercise, including any rights to receive payments of
principal, premium or interest on the underlying debt securities or to enforce covenants in the applicable indenture. Until any
warrants to purchase common stock or preferred stock are exercised, the holders of the warrants will not have any rights of holders
of the underlying common stock or preferred stock, including any rights to receive dividends or payments upon any liquidation,
dissolution or winding up on the common stock or preferred stock, if any.
DESCRIPTION
OF UNITS
We
may issue units consisting of any combination of the other types of securities offered under this prospectus in one or more series.
We may evidence each series of units by unit certificates that we will issue under a separate agreement. We may enter into unit
agreements with a unit agent. Each unit agent will be a bank or trust company that we select. We will indicate the name and address
of the unit agent in the applicable prospectus supplement relating to a particular series of units.
The
following description, together with the additional information included in any applicable prospectus supplement, summarizes the
general features of the units that we may offer under this prospectus. You should read any prospectus supplement and any free
writing prospectus that we may authorize to be provided to you related to the series of units being offered, as well as the complete
unit agreements that contain the terms of the units. Specific unit agreements will contain additional important terms and provisions
and we will file as an exhibit to the registration statement of which this prospectus is a part, or will incorporate by reference
from another report that we file with the SEC, the form of each unit agreement relating to units offered under this prospectus.
If
we offer any units, certain terms of that series of units will be described in the applicable prospectus supplement, including,
without limitation, the following, as applicable:
|
|
●
|
the
title of the series of units;
|
|
|
●
|
identification
and description of the separate constituent securities comprising the units;
|
|
|
●
|
the
price or prices at which the units will be issued;
|
|
|
●
|
the
date, if any, on and after which the constituent securities comprising the units will
be separately transferable;
|
|
|
●
|
a
discussion of certain U.S. federal income tax considerations applicable to the units;
and
|
|
|
●
|
any
other terms of the units and their constituent securities.
|
GLOBAL
SECURITIES
Book-Entry,
Delivery and Form
Unless
we indicate differently in a prospectus supplement, the securities initially will be issued in book-entry form and represented
by one or more global notes or global securities, or, collectively, global securities. The global securities will be deposited
with, or on behalf of, The Depository Trust Company, New York, New York, as depositary, or DTC, and registered in the name of
Cede & Co., the nominee of DTC. Unless and until it is exchanged for individual certificates evidencing securities under the
limited circumstances described below, a global security may not be transferred except as a whole by the depositary to its nominee
or by the nominee to the depositary, or by the depositary or its nominee to a successor depositary or to a nominee of the successor
depositary.
DTC
has advised us that it is a:
|
|
●
|
limited-purpose
trust company organized under the New York Banking Law;
|
|
|
●
|
“banking
organization” within the meaning of the New York Banking Law;
|
|
|
●
|
member
of the Federal Reserve System;
|
|
|
●
|
“clearing
corporation” within the meaning of the New York Uniform Commercial Code; and
|
|
|
●
|
“clearing
agency” registered pursuant to the provisions of Section 17A of the Exchange Act.
|
DTC
holds securities that its participants deposit with DTC. DTC also facilitates the settlement among its participants of securities
transactions, such as transfers and pledges, in deposited securities through electronic computerized book-entry changes in participants’
accounts, thereby eliminating the need for physical movement of securities certificates. “Direct participants” in
DTC include securities brokers and dealers, including underwriters, banks, trust companies, clearing corporations and other organizations.
DTC is a wholly-owned subsidiary of The Depository Trust & Clearing Corporation, or DTCC. DTCC is the holding company for
DTC, National Securities Clearing Corporation and Fixed Income Clearing Corporation, all of which are registered clearing agencies.
DTCC is owned by the users of its regulated subsidiaries. Access to the DTC system is also available to others, which we sometimes
refer to as indirect participants, that clear through or maintain a custodial relationship with a direct participant, either directly
or indirectly. The rules applicable to DTC and its participants are on file with the SEC.
Purchases
of securities under the DTC system must be made by or through direct participants, which will receive a credit for the securities
on DTC’s records. The ownership interest of the actual purchaser of a security, which we sometimes refer to as a beneficial
owner, is in turn recorded on the direct and indirect participants’ records. Beneficial owners of securities will not receive
written confirmation from DTC of their purchases. However, beneficial owners are expected to receive written confirmations providing
details of their transactions, as well as periodic statements of their holdings, from the direct or indirect participants through
which they purchased securities. Transfers of ownership interests in global securities are to be accomplished by entries made
on the books of participants acting on behalf of beneficial owners. Beneficial owners will not receive certificates representing
their ownership interests in the global securities, except under the limited circumstances described below.
To
facilitate subsequent transfers, all global securities deposited by direct participants with DTC will be registered in the name
of DTC’s partnership nominee, Cede & Co., or such other name as may be requested by an authorized representative of
DTC. The deposit of securities with DTC and their registration in the name of Cede & Co. or such other nominee will not change
the beneficial ownership of the securities. DTC has no knowledge of the actual beneficial owners of the securities. DTC’s
records reflect only the identity of the direct participants to whose accounts the securities are credited, which may or may not
be the beneficial owners. The participants are responsible for keeping account of their holdings on behalf of their customers.
So
long as the securities are in book-entry form, you will receive payments and may transfer securities only through the facilities
of the depositary and its direct and indirect participants. We will maintain an office or agency in the location specified in
the prospectus supplement for the applicable securities, where notices and demands in respect of the securities and the indenture
may be delivered to us and where certificated securities may be surrendered for payment, registration of transfer or exchange.
Conveyance
of notices and other communications by DTC to direct participants, by direct participants to indirect participants and by direct
participants and indirect participants to beneficial owners will be governed by arrangements among them, subject to any legal
requirements in effect from time to time.
Redemption
notices will be sent to DTC. If less than all of the securities of a particular series are being redeemed, DTC’s practice
is to determine by lot the amount of the interest of each direct participant in the securities of such series to be redeemed.
Neither
DTC nor Cede & Co. (or such other DTC nominee) will consent or vote with respect to the securities. Under its usual procedures,
DTC will mail an omnibus proxy to us as soon as possible after the record date. The omnibus proxy assigns the consenting or voting
rights of Cede & Co. to those direct participants to whose accounts the securities of such series are credited on the record
date, identified in a listing attached to the omnibus proxy.
So
long as securities are in book-entry form, we will make payments on those securities to the depositary or its nominee, as the
registered owner of such securities, by wire transfer of immediately available funds. If securities are issued in definitive certificated
form under the limited circumstances described below, we will have the option of making payments by check mailed to the addresses
of the persons entitled to payment or by wire transfer to bank accounts in the United States designated in writing to the applicable
trustee or other designated party at least 15 days before the applicable payment date by the persons entitled to payment, unless
a shorter period is satisfactory to the applicable trustee or other designated party.
Redemption
proceeds, distributions and dividend payments on the securities will be made to Cede & Co., or such other nominee as may be
requested by an authorized representative of DTC. DTC’s practice is to credit direct participants’ accounts upon DTC’s
receipt of funds and corresponding detail information from us on the payment date in accordance with their respective holdings
shown on DTC records. Payments by participants to beneficial owners will be governed by standing instructions and customary practices,
as is the case with securities held for the account of customers in bearer form or registered in “street name.” Those
payments will be the responsibility of participants and not of DTC or us, subject to any statutory or regulatory requirements
in effect from time to time. Payment of redemption proceeds, distributions and dividend payments to Cede & Co., or such other
nominee as may be requested by an authorized representative of DTC, is our responsibility, disbursement of payments to direct
participants is the responsibility of DTC, and disbursement of payments to the beneficial owners is the responsibility of direct
and indirect participants.
Except
under the limited circumstances described below, purchasers of securities will not be entitled to have securities registered in
their names and will not receive physical delivery of securities. Accordingly, each beneficial owner must rely on the procedures
of DTC and its participants to exercise any rights under the securities and the indenture.
The
laws of some jurisdictions may require that some purchasers of securities take physical delivery of securities in definitive form.
Those laws may impair the ability to transfer or pledge beneficial interests in securities.
DTC
may discontinue providing its services as securities depositary with respect to the securities at any time by giving reasonable
notice to us. Under such circumstances, in the event that a successor depositary is not obtained, securities certificates are
required to be printed and delivered.
As
noted above, beneficial owners of a particular series of securities generally will not receive certificates representing their
ownership interests in those securities. However, if:
|
|
●
|
DTC
notifies us that it is unwilling or unable to continue as a depositary for the global
security or securities representing such series of securities or if DTC ceases to be
a clearing agency registered under the Exchange Act at a time when it is required to
be registered and a successor depositary is not appointed within 90 days of the notification
to us or of our becoming aware of DTC’s ceasing to be so registered, as the case
may be;
|
|
|
●
|
we
determine, in our sole discretion, not to have such securities represented by one or
more global securities; or
|
|
|
●
|
an
Event of Default has occurred and is continuing with respect to such series of securities,
|
we
will prepare and deliver certificates for such securities in exchange for beneficial interests in the global securities. Any beneficial
interest in a global security that is exchangeable under the circumstances described in the preceding sentence will be exchangeable
for securities in definitive certificated form registered in the names that the depositary directs. It is expected that these
directions will be based upon directions received by the depositary from its participants with respect to ownership of beneficial
interests in the global securities.
We
have obtained the information in this section and elsewhere in this prospectus concerning DTC and DTC’s book-entry system
from sources that are believed to be reliable, but we take no responsibility for the accuracy of this information.
PLAN
OF DISTRIBUTION
We
may sell the securities from time to time in short or long transactions pursuant to underwritten public offerings, negotiated
transactions, block trades or a combination of these methods or through underwriters or dealers, through agents and/or directly
to one or more purchasers. The securities may be distributed from time to time in one or more transactions:
|
|
●
|
at
a fixed price or prices, which may be changed;
|
|
|
●
|
at
market prices prevailing at the time of sale;
|
|
|
●
|
at
prices related to such prevailing market prices; or
|
|
|
●
|
at
negotiated prices.
|
Each
time that we use this prospectus to sell securities, we will also provide a prospectus supplement that contains the specific terms
of the offering. The prospectus supplement will set forth the terms of the offering of the securities, including:
|
|
●
|
the
name or names of any underwriters, dealers or agents and the amounts of any securities
underwritten or purchased by each of them; and
|
|
|
●
|
the
public offering price of the common stock and the proceeds to us and any discounts, commissions
or concessions allowed or reallowed or paid to dealers.
|
Any
public offering price and any discounts or concessions allowed or reallowed or paid to dealers may be changed from time to time.
If
underwriters are used in the sale of any securities, the securities will be acquired by the underwriters for their own account
and may be resold from time to time in one or more transactions, including negotiated transactions, at a fixed public offering
price or at varying prices determined at the time of sale. The securities may be either offered to the public through underwriting
syndicates represented by managing underwriters, or directly by underwriters. Generally, the underwriters’ obligations to
purchase the securities will be subject to certain conditions precedent. The underwriters will be obligated to purchase all of
the securities if they purchase any of securities.
We
may sell the securities through agents from time to time. The prospectus supplement will name any agent involved in the offer
or sale of the securities and any commissions we pay to them. Generally, any agent will be acting on a best efforts basis for
the period of its appointment.
We
may authorize underwriters, dealers or agents to solicit offers by certain purchasers to purchase the securities from us at the
public offering price set forth in the prospectus supplement pursuant to delayed delivery contracts providing for payment and
delivery on a specified date in the future. The contracts will be subject only to those conditions set forth in the prospectus
supplement, and the prospectus supplement will set forth any commissions we pay for solicitation of these contracts.
Agents
and underwriters may be entitled to indemnification by us against certain civil liabilities, including liabilities under the Securities
Act of 1933, as amended, or to contribution with respect to payments which the agents or underwriters may be required to make
in respect thereof. Agents and underwriters may be customers of, engage in transactions with, or perform services for us in the
ordinary course of business.
All
securities we may offer, other than common stock, will be new issues of securities with no established trading market. Any underwriters
may make a market in these securities, but will not be obligated to do so and may discontinue any market making at any time without
notice. We cannot guarantee the liquidity of the trading markets for any securities.
We
may enter into derivative transactions with third parties, or sell securities not covered by this prospectus to third parties
in privately negotiated transactions. If the applicable prospectus supplement indicates, in connection with those derivatives,
the third parties may sell securities covered by this prospectus and the applicable prospectus supplement, including in short
sale transactions. If so, the third party may use securities pledged by us or borrowed from us or others to settle those sales
or to close out any related open borrowings of securities, and may use securities received from us in settlement of those derivatives
to close out any related open borrowings of securities. The third party in such sale transactions will be an underwriter and will
be identified in the applicable prospectus supplement (or a post-effective amendment).
Upon
written instruction from us, a sales agent party to a distribution agency agreement with us will use its commercially reasonable
efforts to sell on our behalf, as our agent, the securities offered as agreed upon by us and the sales agent. We will designate
the maximum amount of securities to be sold through the sales agent, on a daily basis or otherwise as we and the sales agent agree.
Subject to the terms and conditions of the applicable distribution agency agreement, the sales agent will use its commercially
reasonable efforts to sell, as our sales agent and on our behalf, all of the designated securities. We may instruct the sales
agent not to sell securities if the sales cannot be effected at or above the price designated by us in any such instruction. We
may suspend the offering of securities under any distribution agency agreement by notifying the sales agent. Likewise, the sales
agent may suspend the offering of securities under the applicable distribution agency agreement by notifying us of such suspension.
We
also may sell securities to the sales agent as principal for its own account at a price agreed upon at the time of sale. If we
sell securities to the sales agent as principal, we will enter into a separate agreement setting forth the terms of such transaction.
The
name of any such underwriter or agent involved in the offer and sale of securities, the amounts underwritten and the nature of
its obligations to take our securities will be described in the applicable prospectus supplement.
LEGAL
MATTERS
The
legality and validity of the securities offered from time to time under this prospectus will be passed upon by Goodwin Procter
LLP, New York, New York. Any underwriters will also be advised about the validity of the securities and other legal matters by
their own counsel, which will be named in the prospectus supplement.
EXPERTS
The
financial statements of Advaxis, Inc. as of October 31, 2017 and 2016, and for the years ended October 31, 2017, 2016 and 2015,
have been incorporated by reference herein and in the registration statement in reliance upon the report of Marcum LLP, independent
registered public accounting firm, and upon the authority of said firm as experts in accounting and auditing.
Up
to $20,000,000 of Common Stock
1,084,266
Shares of Common Stock

PROSPECTUS
SUPPLEMENT
July
30, 2020
Ayala Pharmaceuticals (QX) (USOTC:ADXS)
Historical Stock Chart
From Mar 2024 to Apr 2024
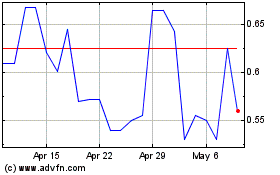
Ayala Pharmaceuticals (QX) (USOTC:ADXS)
Historical Stock Chart
From Apr 2023 to Apr 2024