Philips to expand its image-guided therapy devices portfolio through acquisition of Intact Vascular
August 27 2020 - 2:00AM

Philips to expand its image-guided therapy devices portfolio
through acquisition of Intact Vascular
August 27, 2020
Amsterdam, the Netherlands – Royal Philips
(NYSE: PHG, AEX: PHIA), a global leader in health technology, today
announced that it has signed an agreement to acquire Intact
Vascular, Inc., a U.S.-based developer of medical devices for
minimally-invasive peripheral vascular procedures. Intact Vascular
will enhance Philips’ image-guided therapy portfolio, combining
Philips’ interventional imaging platform and diagnostic and
therapeutic devices with Intact Vascular’s unique, specialized
implantable device to optimize the treatment of patients with
Peripheral Artery Disease (PAD).
PAD develops when plaque builds up in arteries and reduces blood
flow to the limbs, most commonly the legs. It affects more than 200
million people worldwide [1]. PAD symptoms include recurrent
fatigue, leg pain, and foot or leg wounds that do not heal or heal
very slowly. Critical Limb Ischemia (CLI) is an advanced stage of
PAD, and is typically associated with high rates of amputation and
mortality.
“Acquiring Intact Vascular will expand our portfolio of
minimally invasive therapy options for Peripheral Artery Disease
with the Tack Endovascular System [2], a much needed implant that
effectively restores blood flow in small limb vessels, promotes
healing and preserves limbs,” said Chris Landon, Senior Vice
President and General Manager Image Guided Therapy Devices at
Philips. “Through the integration of our interventional imaging
systems and diagnostic and therapeutic devices, we will be able to
provide clinicians with a complete procedural solution to optimize
the treatment of patients with this disease.”
Philips’ market leading peripheral vascular portfolio already
includes advanced interventional imaging systems for precision
guidance; intravascular ultrasound (IVUS) catheters to assess the
location of the disease and lesion morphology and guide and confirm
the treatment; peripheral atherectomy devices to remove blockages;
and peripheral therapy devices, such as Philips’ Stellarex
drug-coated balloon, to treat lesions.
Intact Vascular will strengthen this portfolio with the Tack
Endovascular System, which reinforces standard and drug-coated
balloon PAD treatment results. Intact Vascular’s Tack [2] implant
is a first-of-its-kind, minimal-metal, dissection repair device
that provides precision treatment of peripheral arterial
dissections following balloon angioplasty in above-the-knee (ATK)
and below-the-knee (BTK) therapeutic interventions [3]. The Tack
implant leaves less metal behind compared to stents, preserving
future treatment options and ultimately preserving limbs. Moreover,
Intact Vascular’s Tack implant offers a solution for repairing
dissections and optimizing post-angioplasty outcomes in the
challenging CLI patient population.
“We are excited about the strategic fit between our team,
expertise and unique therapeutic device, and Philips’ Image-Guided
Therapy business,” said Bruce Shook, President and CEO of Intact
Vascular. “We share the same vision of complete procedural
solutions to improve existing procedures and expand treatment
options. We look forward to completing the transaction and working
closely with Philips on a seamless transition.”
The transaction, which is subject to customary closing
conditions, is expected to be completed in the third quarter of
2020. Philips will acquire Intact Vascular for an upfront cash
consideration of USD 275 million (approx. EUR 234 million), and
deferred payments for which the company expects to recognize a
provision of USD 85 million (approx. EUR 72 million) upon
completion of the transaction.
Intact Vascular was founded in 2011 and is headquartered in
Wayne, Pennsylvania, in the U.S. On completion of the transaction,
Intact Vascular will become part of Philips’ Image-Guided Therapy
business.
[1] J. Shu J. and G. Santulli; Update on peripheral artery
disease: Epidemiology and evidence-based facts; Atherosclerosis,
2018 Aug; 275: 379-381.[2] Tack Endovascular System and Tack are
trademarks of Intact Vascular, Inc.[3] The Tack Endovascular System
has been approved by the U.S. Food and Drug Administration (FDA)
for ATK and BTK dissection repair interventions.
For further information, please
contact:
Ben ZwirsPhilips Global Press OfficeTel.: +31 6 15213446E-mail:
ben.zwirs@philips.com
Derya GuzelPhilips Investor RelationsTel.: +31 20 59
77222E-mail: derya.guzel@philips.com
About Royal PhilipsRoyal Philips (NYSE: PHG,
AEX: PHIA) is a leading health technology company focused on
improving people's health and enabling better outcomes across the
health continuum from healthy living and prevention, to diagnosis,
treatment and home care. Philips leverages advanced technology and
deep clinical and consumer insights to deliver integrated
solutions. Headquartered in the Netherlands, the company is a
leader in diagnostic imaging, image-guided therapy, patient
monitoring and health informatics, as well as in consumer health
and home care. Philips generated 2019 sales of EUR 19.5 billion and
employs approximately 81,000 employees with sales and services in
more than 100 countries. News about Philips can be found at
www.philips.com/newscenter.
Forward-looking statements This release
contains certain forward-looking statements with respect to the
financial condition, results of operations and business of Philips
and certain of the plans and objectives of Philips with respect to
these items. Examples of forward-looking statements include
statements made about the strategy, estimates of sales growth,
future EBITA, future developments in Philips’ organic business and
the completion of acquisitions and divestments. By their nature,
these statements involve risk and uncertainty because they relate
to future events and circumstances and there are many factors that
could cause actual results and developments to differ materially
from those expressed or implied by these statements.
- Intact Vasculars Tack implant-4F
- Intact Vasculars Tack implant-4F-BTK
- Peripheral hospital procedure01
- Peripheral hospital procedure02
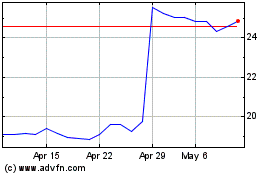
Koninklijke Philips NV (EU:PHIA)
Historical Stock Chart
From Mar 2024 to Apr 2024
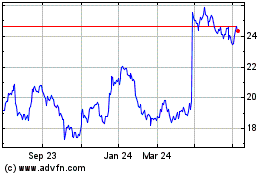
Koninklijke Philips NV (EU:PHIA)
Historical Stock Chart
From Apr 2023 to Apr 2024