FDA issues Emergency Use Authorization for Philips’ new acute care patient monitoring solutions for hospital-based remote m...
June 02 2020 - 8:00AM

FDA issues Emergency Use Authorization for Philips’ new acute care
patient monitoring solutions for hospital-based remote monitoring
June 2, 2020
- Philips’ IntelliVue Patient Monitors MX750/MX850, IntelliVue
4-Slot Module Rack FMX-4 and IntelliVue Active Displays AD75/AD85
receive FDA Emergency Use Authorization for use in the US during
the COVID-19 emergency [1]
- New acute care patient monitoring solution offers an expanded,
real-time view of a patient’s vital signs for bedside and remote
monitoring in the hospital
Amsterdam, the Netherlands – Royal Philips
(NYSE: PHG, AEX: PHIA), a global leader in health technology, today
announced that the US FDA has issued an Emergency Use Authorization
(EUA) for Philips’ IntelliVue Patient Monitors MX750/MX850 and its
IntelliVue Active Displays AD75/AD85, for use in the US during the
COVID-19 health emergency [1]. These patient monitoring solutions
support infection-control protocols and remotely provide critical
patient information for caregivers, capabilities that are much
needed when caring for hospitalized COVID-19 patients.
Philips’ IntelliVue Patient Monitors MX750/MX850 and IntelliVue
Active Displays AD75/AD85 received CE mark in 2019 and are already
being used in hospitals across Europe. The EUA allows Philips to
start delivering the new remote patient monitoring solution to
hospitals in the US, and the company is committed to submitting a
510(k) to FDA for this acute care solution in the course of
2020.
As countries across the globe continue to combat COVID-19, while
gradually resuming elective care, Philips is significantly
increasing its patient monitor production to address the demand for
increased Intensive Care Unit (ICU) capacity. Hospitals and health
systems are increasingly relying on health technology to better
manage the influx of COVID-19 patients they are seeing in Emergency
Rooms (ERs) and ICUs. To minimize staff exposure to the virus that
causes COVID-19 while still delivering quality care, there is a
critical need for patient monitors that enable clinicians to
remotely monitor a patient’s condition.
“As the world continues to battle against COVID-19, we’re
committed to ramping up production of all critical solutions that
can help in this time of crisis,” said Peter Ziese, General
Manager, Monitoring and Analytics at Philips. “This FDA EUA for our
MX750 and MX850 monitors and IntelliVue AD75 and AD85 Active
Displays allows us to do that for these remote patient monitoring
solutions, which are of vital need in the ICU. At Philips, being
able to provide the right information at the right time to
caregivers has always been a top priority. Now more than ever,
there’s an urgent need to make sure those on the frontline have all
the available resources at their disposal.”
The IntelliVue Patient Monitors MX750/MX850 and IntelliVue
Active Displays AD75/AD85 offer advanced functionality and clinical
decision support capabilities such as Philips’ IntelliVue Horizon
Trends information view, which shows deviations in vital signs (for
example, CO2 and heart rate) to contextualize a patient’s
condition, while also supporting infection-control protocols and
access to key information both remotely and at the bedside.
Features such as Philips’ Alarm Advisor and Alarm Reporting help to
reduce alarm fatigue for caregivers, while the smooth glass
surfaces, rounded edges and special surface material of the
monitors and displays facilitates cleaning and disinfection.
The MX750 and MX850 monitors are the latest additions to
Philips’ portfolio of integrated patient monitoring solutions to
support improved clinical and operational workflows. The MX750 and
MX850 also include updated features, including enhancements to
monitor and assess clinical and network device performance, and
additional functionalities to strengthen cybersecurity.
For more information on how Philips is addressing COVID-19
globally, please visit the Philips centralized COVID-19 hub.
[1] The status of the Emergency Use Authorization
can be found on the FDA website.
·The IntelliVue Patient
Monitors MX750/MX850, IntelliVue 4-Slot Module Rack FMX-4 and
IntelliVue Active Displays AD75/AD85 have neither been FDA cleared
or approved for the indication to assist in for monitoring and
recording of, and for generating alarms for, multiple physiological
parameters of adult, pediatric, and neonate patients having or
suspected of having Coronavirus-2019
(COVID-19) ·The IntelliVue
Patient Monitors MX750/MX850, IntelliVue 4-Slot Module Rack FMX-4
and IntelliVue Active Displays AD75/AD85 have been authorized for
the above emergency use by FDA under an EUA
·The IntelliVue Patient
Monitors MX750/MX850, IntelliVue 4-Slot Module Rack FMX-4 and
IntelliVue Active Displays AD75/AD85 have been authorized only for
the duration of the declaration that circumstances exist justifying
the authorization of the emergency use of medical devices under
section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless
the authorization is terminated or revoked sooner.
For further information, please contact:
Steve KlinkPhilips Global Press OfficeTel.: +31 6
10888824E-mail: steve.klink@philips.com
Kathy O’ReillyPhilips Global Press OfficeTel.: + 1
978-221-8919E-mail: kathy.oreilly@philips.comTwitter
@kathyoreilly
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health
technology company focused on improving people's health and
enabling better outcomes across the health continuum from healthy
living and prevention, to diagnosis, treatment and home care.
Philips leverages advanced technology and deep clinical and
consumer insights to deliver integrated solutions. Headquartered in
the Netherlands, the company is a leader in diagnostic imaging,
image-guided therapy, patient monitoring and health informatics, as
well as in consumer health and home care. Philips generated 2019
sales of EUR 19.5 billion and employs approximately 81,000
employees with sales and services in more than 100 countries. News
about Philips can be found at www.philips.com/newscenter.
- Philips IntelliVue MX750, MX850
- Philips IntelliVue Active Display, AD75 and AD85
- Philips Intellivue MX850
Koninklijke Philips NV (EU:PHIA)
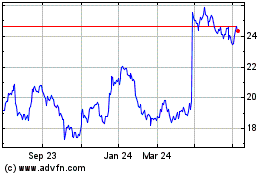
Historical Stock Chart
From Mar 2024 to Apr 2024
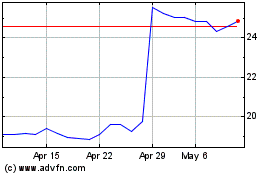
Koninklijke Philips NV (EU:PHIA)
Historical Stock Chart
From Apr 2023 to Apr 2024