|
The information in this prospectus supplement is not complete and may be changed. This prospectus supplement is not an offer to sell these securities, and we are not soliciting offers to buy these securities, in any state where the offer or sale is not permitted.
|
SUBJECT
TO COMPLETION, THE DATE OF THIS PROSPECTUS SUPPLEMENT IS MARCH 2, 2018.
Filed Pursuant to Rule 424(b)(3)
Registration No. 333-221477
PROSPECTUS SUPPLEMENT
(to Prospectus dated November 28, 2017)
[___________]

SONOMA PHARMACEUTICALS, INC.
COMMON STOCK
We are offering [______]
shares of common stock, par value $0.0001 per share, or the Shares, at a public offering price of $[__] per Share.
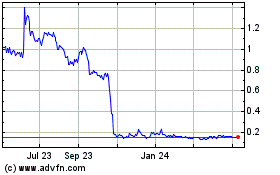
Our common stock is quoted on The Nasdaq
Capital Market under the symbol “SNOA.” On February 28, 2018, the last reported sale price for our common stock was
$3.50 per share.
The aggregate market value of our outstanding
voting common equity held by non-affiliates on February 28, 2018 was $15,673,693 based on a closing stock price of $3.50 on February
28, 2018. The aggregate market value of our outstanding voting common equity held by non-affiliates on January 10, 2018 was $25,122,691
based on a closing stock price of $5.61 from January 10, 2018. During the twelve calendar months prior to and including the date
hereof, we sold securities with an aggregate market value of $1,033,980 pursuant to General Instruction I.B.6. of Form S-3.
Investing in our securities involves
a high degree of risk. Before buying any of our securities, you should carefully consider the risk factors described in “Risk
Factors” on page S-3 of this prospectus supplement.
Neither the Securities and Exchange
Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus
supplement or the accompanying prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
|
|
|
|
|
Per Share
|
|
|
|
Total
|
|
|
Public offering price
|
|
|
$
|
|
|
|
$
|
|
|
|
Placement agent fees (1)
|
|
|
$
|
|
|
|
$
|
|
|
|
Proceeds, before expenses, to us (2)
|
|
|
$
|
|
|
|
$
|
|
|
____________
(1) See “
Plan of
Distribution
” beginning on page S-8 of this prospectus supplement for a description of the compensation payable to the
placement agents, including the issuance of placement agent warrants and reimbursement of expenses.
(2) Assumes maximum offering is
completed.
The placement agents expect to deliver the shares against
payment therefore on or about [______]
,
2018.
|
Dawson James Securities, Inc.
|
Benchmark
|
The date of this prospectus supplement
is [_____], 2018.
TABLE OF CONTENTS
Prospectus Supplement
Prospectus
You should rely only on the information
incorporated by reference or provided in this prospectus supplement, the accompanying prospectus and the registration statement.
We have not authorized anyone else to provide you with different information. If anyone provides you with different or inconsistent
information, you should not rely on it. We are not making an offer to sell these securities in any state where the offer or sale
is not permitted. You should assume that the information in this prospectus supplement, the accompanying prospectus, including
any information incorporated therein by reference, is accurate only as of the dates of those documents. Our business, financial
condition, results of operations and prospects may have changed since those dates.
We are offering to sell, and are
seeking offers to buy, the securities only in jurisdictions where such offers and sales are permitted. The distribution of this
prospectus supplement and the accompanying prospectus and the offering of the securities in certain jurisdictions may be restricted
by law. Persons outside the United States who come into possession of this prospectus supplement and the accompanying prospectus
must inform themselves about and observe any restrictions relating to the offering of the securities and the distribution of this
prospectus supplement and the accompanying prospectus outside the United States. This prospectus supplement and the accompanying
prospectus do not constitute, and may not be used in connection with, an offer to sell, or a solicitation of an offer to buy, any
securities offered by this prospectus supplement and the accompanying prospectus by any person in any jurisdiction in which it
is unlawful for such person to make such an offer or solicitation.
ABOUT THIS PROSPECTUS
SUPPLEMENT
This prospectus supplement and the accompanying
prospectus are part of a “shelf” registration statement on Form S-3 that we filed with the Securities and Exchange
Commission on November 9, 2017, and declared effective on November 27, 2017.
This document is in two parts. The first
part is this prospectus supplement, which describes the terms of this offering and also adds to and updates information contained
in the accompanying prospectus and the documents incorporated by reference into this prospectus supplement and the accompanying
prospectus. The second part is the accompanying prospectus, which gives more general information about the shares of our common
stock and other securities we may offer from time to time under our shelf registration statement, some of which does not apply
to the securities offered by this prospectus supplement. To the extent there is a conflict between the information contained in
this prospectus supplement, on the one hand, and the information contained in the accompanying prospectus or any document incorporated
by reference therein, on the other hand, you should rely on the information in this prospectus supplement.
You should read this prospectus supplement,
the accompanying prospectus, the documents incorporated by reference in this prospectus supplement and the accompanying prospectus
before making an investment decision. You should also read and consider the information in the documents referred to in the sections
of this prospectus supplement entitled “
Where You Can Find More Information
” and “
Incorporation of
Certain Documents by Reference
.”
In this prospectus supplement and the
accompanying prospectus, unless otherwise indicated, the terms “Sonoma,” “we,” “us,” “our,”
and similar terms refer to Sonoma Pharmaceuticals, Inc. and its subsidiaries on a consolidated basis.
THE OFFERING
Common stock outstanding
on February 28, 2018 (1)
|
4,736,936 shares
|
|
|
|
|
Common stock offered by us
|
[____________] shares
|
|
|
|
|
Common stock to be outstanding immediately after this offering assuming all shares of common stock are sold (1)
|
[____________] shares
|
|
|
|
|
Use of proceeds
|
The net proceeds from this offering after deducting estimated commissions and expenses payable by us will be approximately $[______] at an offering price of $[______] per share. We intend to use the net proceeds from this offering for working capital and general corporate purposes. See “
Use of Proceeds
” for a more detailed description of the intended use of proceeds from this offering.
|
|
|
|
|
Risk factors
|
This investment involves a high degree of risk. See “
Risk Factors
” below for factors to consider before deciding to purchase our securities.
|
|
|
|
|
Nasdaq Capital Market
common stock symbol
|
SNOA
|
|
|
(1)
|
Excluding 2,751,000 shares of common stock issuable upon the exercise of outstanding options, restricted stock units, and warrants
as of December 31, 2017.
|
RISK FACTORS
Investing in our securities involves
a high degree of risk. Before investing in our securities, you should carefully consider the risks described below, together with
all of the other information contained in this prospectus supplement and the accompanying prospectus and incorporated by reference
herein and therein, including from our most recent Annual Report on Form 10-K and subsequent filings. Some of these factors relate
principally to our business and the industry in which we operate. Other factors relate principally to your investment in our securities.
The risks and uncertainties described therein and below are not the only risks we face, but those that we consider to be material.
Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also materially and adversely
affect our business and operations. If any of the matters included in the following risks were to occur, our business, financial
condition, results of operations, cash flows or prospects could be materially and adversely affected. In such case, you may lose
all or part of your investment. Please also read carefully the section below entitled “Cautionary Note Regarding Forward-Looking
Statements.”
Additional Risks Related to This
Offering
We will have broad discretion
in how we use the proceeds, and we may use the proceeds in ways in which purchasers in this offering and other stockholders may
disagree.
We intend to use the net proceeds from
this offering for working capital and general corporate purposes. Our management will have broad discretion in the application
of the net proceeds from this offering and could spend the proceeds in ways that do not improve our results of operations or enhance
the value of our common stock. The failure by management to apply these funds effectively could result in financial losses that
could have a material adverse effect on our business or cause the price of our common stock to decline.
Purchasers in this offering may
suffer immediate and substantial dilution in the net tangible book value per share of our common stock.
Because the price per share of common
stock in this offering may be substantially higher than the net tangible book value per share of common stock, purchasers in this
offering may suffer immediate and substantial dilution in the net tangible book value per share of common stock. After giving effect
to the sale of [______] shares of our common stock at a price of $[______] per share, and after deducting commissions and estimated
offering expenses payable by us, purchasers in this offering will experience immediate dilution of $[______] per share, representing
the difference between our as adjusted net tangible book value per share as of December 31, 2017, after giving effect to this offering
and the assumed offering price. See the section entitled “
Dilution
” below for a more detailed illustration of
the dilution you would incur if you participate in this offering.
Purchasers in this offering may
experience future dilution as a result of future equity offerings.
In order to raise additional capital
for the execution of our business plans, we may in the future offer additional shares of our common stock or other securities convertible
into or exchangeable for our common stock at prices that may not be the same as the price per share in this offering. We may sell
shares or other securities in any other offering at a price per share that is less than the price per share paid by purchasers
in this offering, and investors purchasing shares or other securities in the future could have rights superior to existing stockholders.
The price per share at which we sell additional shares of our common stock, or securities convertible or exchangeable into common
stock, in future transactions may be higher or lower than the price per share paid by investors in this offering. In addition,
on December 8, 2017, we entered into an At Market Issuance Sales Agreement, with B. Riley FBR, Inc. under which we may issue and
sell shares of our common stock having an aggregate offering price of up to $5,000,000 from time to time through B. Riley acting
as our sales agent. Since the execution of this agreement, we have sold approximately $1.0 million of shares of our common stock.
Future sales pursuant to this arrangement may result in additional dilution to our existing shareholders.
We have the right to enter into
separate purchase agreements with significant purchasers in this offering, and the remaining purchasers in this offering will not
be able to rely on representations and warranties we agree to in such purchase agreements.
As part of this offering, we have the
right to enter into separate purchase agreements with significant purchasers of shares in this offering. If we exercise this right,
the purchase agreement we enter into will contain certain representations and warranties that we provide only to the purchasers
party to such purchase agreement. However, other purchasers in this offering will not receive the same representations and warranties
and cannot rely on such representations and warranties in making their investment decision.
We are selling the securities
offered in this prospectus on a “best efforts” basis with no minimum offering and may not be able to sell any of the
securities offered herein.
We have engaged the placement agents
to act as a placement agent in connection with this offering. While the placement agents will use their reasonable efforts to arrange
for the sale of the securities, they are under no obligation to purchase any of the securities. As a result, there are no firm
commitments to purchase any of the securities in this offering. Consequently, there is no guarantee that we will be capable of
selling all, or any, of the securities being offered hereby. In addition, we have not specified a minimum offering amount nor have
or will we establish an escrow account in connection with this offering. Because there is no escrow account and no minimum offering
amount, investors could be in a position where they have invested in our company, but we are unable to fulfill our objectives due
to a lack of interest in this offering. Further, because there is no escrow account in operation and no minimum investment amount,
any proceeds from the sale of securities offered by us will be available for our immediate use, despite uncertainty about whether
we would be able to use such funds to effectively implement our business plan. Investor funds will not be returned under any circumstances
whether during or after the offering.
OUR
BUSINESS
We are a specialty pharmaceutical company
dedicated to identifying, developing and commercializing unique, differentiated therapies to millions of patients living with chronic
skin conditions. We believe our products, which are sold throughout the United States and internationally, have improved patient
outcomes for more than five million patients globally by treating and reducing certain topical skin diseases including acne, atopic
dermatitis, scarring, infections, itch, pain and harmful inflammatory responses.
We are focused on the development and
commercialization of therapeutic solutions in medical dermatology to treat skin conditions, such as acne, atopic dermatitis and
scarring. These diseases impact millions of patients worldwide and can have significant, multi-dimensional effects on patients’
quality of life, including their physical, functional and emotional well-being.
Some of our key products in the United States are:
|
|
·
|
Celacyn®
, a prescription hypochlorous acid-based scar management gel clinically proven to soften and flatten raised scars while reducing redness and discoloration.
|
|
|
·
|
Ceramax™ Skin Barrier Cream
helps manage dry itchy skin, minor skin irritations, rashes, and inflammation caused by various skin conditions.
|
|
|
·
|
Mondoxyne™
, a prescription oral tetracycline antibiotic used for the treatment of certain bacterial infections, including acne.
|
|
|
·
|
Alevicyn™
, a prescription hypochlorous acid based atopic dermatitis product line clinically proven to reduce pruritus (itch) and pain associated with various dermatoses.
|
|
|
·
|
Sebuderm™
, a prescription topical gel used as an alternative to corticosteroids for the management of the burning, itching and scaling experienced with seborrhea and seborrheic dermatitis.
|
|
|
·
|
Loyon™
, a prescription liquid containing Cetiol® CC and medical grade dimethicone, intended to manage and relieve erythema and itching for various types of dermatoses.
|
|
|
|
|
|
|
·
|
Microcyn®
(sold under a variety of brand names), a line of products based on electrically charged oxychlorine small molecules designed to target a wide range of pathogens including viruses, fungi, spores and bacteria, including antibiotic-resistant strains.
|
Our key product outside the United States
is:
|
|
·
|
Microcyn®
or
Microdacyn60®
(sold under a variety of brand names), a line of products based on electrically charged oxychlorine small molecules designed to target a wide range of pathogens including viruses, fungi, spores and bacteria, including antibiotic-resistant strains.
|
As of December 31, 2017, we have obtained
20 clearances from the U.S. Food and Drug Administration, or FDA, that permit us to sell our products as medical devices for Section
510(k) of the Federal Food, Drug and Cosmetic Act in the United States.
Outside the United States, we sell products
for dermatological and advanced tissue care with a European Conformity marking (known as Conformité Européenne or
CE) covering 25 of our products, and various approvals in China, Southeast Asia, South Korea, India, Australia, New Zealand, and
the Middle East.
Our Strategy
Our strategy is to in-license, acquire,
develop and commercialize unique, affordable and differentiated therapies that we believe advance the standard of care for patients
with dermatological diseases. The key components of our strategy are to:
|
|
·
|
Expand our Internal U.S. Sales Force:
We continue to hire additional experienced sales people who have established relationships with dermatologists in their territories and we currently have a sales force of 35 sales professionals.
|
|
|
·
|
Develop and Launch New Dermatology Products:
We currently sell nine prescription dermatology products in the United States, and have a strong product pipeline of new products, including an oral antibiotic for severe acne and Ceramax™, which utilizes a “state of the art” skin repair technology.
|
|
|
·
|
In-License and Acquire New Product Candidates:
Since beginning our turn-around strategy in 2014, we have executed multiple transactions resulting in adding new products and product candidates to our growing portfolio. In 2015, we acquired the U.S. marketing rights to Mondoxyne™, an oral antibiotic indicated for severe acne. In 2016, we in-licensed Ceramax™ indicated for various dermatoses, and Loyon® indicated as a descaler of various dermatoses and psoriasis.
|
|
|
·
|
Create a Competitive Pricing Strategy:
We have and will continue to develop a unique product pricing strategy, which we believe solves many of the challenges associated with the prescription dermatology market’s current pricing and rebate programs.
|
|
|
·
|
Develop a Pharmaceutical Line:
We plan to acquire or develop pharmaceutical products with affordable clinical trials to increase our market presence and create innovator patent protection.
|
Our plan is to evolve into a leading
dermatology company, providing innovative and cost-effective solutions to patients, while generating strong, consistent revenue
growth and maximizing long-term shareholder value.
Corporate Information
We incorporated under the laws of the
State of California in April 1999 as Micromed Laboratories, Inc. In August 2001, we changed our name to Oculus Innovative Sciences,
Inc. In December 2006, we reincorporated under the laws of the State of Delaware. On December 6, 2016, we changed our name from
Oculus Innovative Sciences, Inc. to Sonoma Pharmaceuticals, Inc. Our principal executive offices are located at 1129 N. McDowell
Blvd., Petaluma, California, 94954, and our telephone number is (707) 283-0550. We have two active wholly-owned subsidiaries: Oculus
Technologies of Mexico, S.A. de C.V., organized in Mexico; and Sonoma Pharmaceuticals Netherlands, B.V., organized in the Netherlands.
Additional Information
Investors and others should note that
we announce material financial information using our company website (www.sonomapharma.com), our investor relations website (ir.sonomapharma.com),
SEC filings, press releases, public conference calls and webcasts. The information on, or accessible through, our websites is not
incorporated by reference in this prospectus supplement.
CAUTIONARY NOTE
REGARDING FORWARD-LOOKING STATEMENTS
This prospectus supplement, the accompanying
prospectus and the documents incorporated by reference in this prospectus supplement contain forward looking statements. When used
in this prospectus supplement, the accompanying prospectus and the documents incorporated by reference herein or therein, the words
“anticipate,” “intend,” “estimate,” “plan,” “project,” “continue,”
“ongoing,” “potential,” “expect,” “predict,” “believe,” “intend,”
“may,” “can,” “will,” “should,” “could,” “would,” “proposal,”
and similar expressions are intended to identify forward-looking statements.
You should not place undue reliance
on these forward-looking statements. Our actual results could differ materially from those anticipated in the forward-looking statements
for many reasons, including the reasons described in our “
Risk Factors
” section. Although we believe the expectations
reflected in the forward-looking statements are reasonable, they relate only to events as of the date on which the statements are
made. These forward-looking statements speak only as of the date of this prospectus supplement. We expressly disclaim any obligation
or undertaking to update or revise any forward-looking statements contained herein to reflect any change in our expectations with
regard thereto or any change in events, conditions or circumstances on which any such statement is based, except as required by
law. You should, however, review additional disclosures we make in our Annual Reports on Form 10-K, our Quarterly Reports on Form
10-Q, and Current Reports on Form 8-K filed with the SEC.
USE OF PROCEEDS
If we complete the maximum offering,
we estimate that the net proceeds from the sale of [______] shares of common stock that we are offering will be approximately $[______]
after deducting the commissions and estimated offering expenses payable by us.
We intend to use the net proceeds of
this offering for working capital and general corporate purposes. As of the date of this prospectus supplement, we cannot specify
with certainty all of the particular uses for the net proceeds to us from this offering. Accordingly, our management will have
broad discretion in the application of these proceeds.
DILUTION
Purchasers of common stock offered by
this prospectus supplement and the accompanying prospectus will suffer immediate and substantial dilution in the net tangible book
value per share of common stock. Our net tangible book value on December 31, 2017 was approximately $14.3 million, or approximately
$3.07 per share of common stock based upon 4,637,541 shares outstanding as of December 31, 2017. Net tangible book value per share
is determined by dividing our net tangible book value, which consists of tangible assets less total liabilities, by the number
of shares of common stock outstanding on that date.
After giving effect to the sale of our
common stock in the aggregate amount of $[______] at an offering price of $[______] per share and after deducting estimated offering
commissions and offering expenses payable by us, our net tangible book value as of December 31, 2017 would have been approximately
$[______], or $[______] per share of common stock. This represents an immediate increase in net tangible book value of $[______]
per share to existing stockholders and immediate dilution in net tangible book value of $[______] per share to purchasers of our
common stock in this offering at the public offering price. The following table illustrates this calculation on a per share basis:
|
Offering price per share
|
|
|
|
|
|
$
|
|
|
|
Net tangible book value per share as of December 31, 2017
|
|
$
|
3.07
|
|
|
|
|
|
|
Increase in net tangible book value per share attributable to this offering
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
As adjusted net tangible book value per share as of December 31, 2017, after giving effect to this offering
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Dilution in net tangible book value per share to new investors purchasing our common stock in this offering
|
|
|
|
|
|
$
|
|
|
The foregoing table is based on 4,637,541
shares of our common stock outstanding as of December 31, 2017 and excludes:
|
|
·
|
1,385,000
shares of common stock issuable upon exercise of outstanding stock options, at a weighted average exercise price of $12.76 per share, under our equity incentive plans;
|
|
|
·
|
33,000
shares of common stock issuable upon exercise/vesting of restricted stock units;
|
|
|
·
|
1,564,381
additional shares of common stock reserved for future issuance under our equity incentive plans;
|
|
|
·
|
1,333,000 shares of common stock issuable upon exercise of outstanding warrants, with current exercise prices ranging from $5.00 per share to $6.50 per share.
|
To the extent that outstanding options
or warrants outstanding as of December 31, 2017, have been or may be exercised or other shares issued, investors purchasing our
common stock in this offering may experience further dilution. In addition, we may choose to raise additional capital due to market
conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans. To
the extent that additional capital is raised through the sale of equity or convertible debt securities, the issuance of these securities
could result in further dilution to our stockholders.
DESCRIPTION OF
SECURITIES
We have 12,000,000 shares of authorized
common stock, par value $0.001 per share. As of February 28, 2018, there were 4,736,936 shares of common stock issued and outstanding.
All outstanding shares of common stock are fully paid and nonassessable, and the shares of common stock offered, when issued, will
be fully paid and nonassessable.
The transfer agent for our common stock
and our warrants is Computershare, Inc. located at 462 South 4th Street, Suite 1600, Louisville, KY 40202. Its telephone number
is 1-888-647-8901.
PLAN OF DISTRIBUTION
Placement Agency Agreement
In connection with this offering, we will enter
into a placement agency agreement with Dawson James Securities Inc. as the lead placement agent and sole bookrunning manager together
with The Benchmark Company, LLC as the co-placement agent, pursuant to which Dawson James Securities, Inc. and The Benchmark Company,
LLC will agree to act as our placement agents on a best efforts basis in connection with the sale of our common stock. The placement
agents will not purchase or sell any securities offered by us under this prospectus for their own account, nor will they be required
to arrange the purchase or sale of any specific number or dollar amount of the securities, but the placement agents will agree
to act as our agent and to use their reasonable best efforts to arrange for the sale of all of the securities in this offering.
The placement agents may engage selected dealers to assist in the placement of the securities. There is no required minimum number
of securities that must be sold as a condition to completion of this offering.
The placement agency agreement will provide
that the obligations of the placement agents are subject to certain conditions precedent, including, among other things, the absence
of any material adverse change in our business and the receipt of customary legal opinions, letters and certificates. In addition,
we will make certain representations and warranties in the placement agency agreement and we will agree to certain covenants in
the placement agent agreement. The placement agents may be deemed to be underwriters within the meaning of Section 2(a)(11) of
the Securities Act, and any commissions received by them and any profit realized on the resale of the securities sold by them while
acting as principal might be deemed to be underwriting discounts or commissions under the Securities Act. As an underwriter, the
placement agents would be required to comply with the Securities Act and the Exchange Act, including without limitation, Rule 10b-5
and Regulation M under the Exchange Act. These rules and regulations may limit the timing of purchases and sales of shares of common
stock by the placement agents acting as principal. Under these rules and regulations, the placement agents (i) may not engage in
any stabilization activity in connection with our securities; and (ii) may not bid for or purchase any of our securities or attempt
to induce any person to purchase any of our securities, other than as permitted under the Exchange Act, until they have completed
their participation in the distribution.
As part of the offering, we reserve the right
to enter into a securities purchase agreement directly with certain purchasers. If we exercise this right, the securities purchase
agreement will contain customary representations and warranties by us and the purchaser. We may also agree to pay the expenses
of such purchasers in connection with the sale.
The placement agency agreement will provide
that we agree, for a period of 60 days from the date of this offering, that we will not (a) offer, sell, or otherwise transfer
or dispose of, directly or indirectly, any shares of our capital stock or any securities convertible into or exercisable or exchangeable
for shares of our capital stock, except for the exercise of outstanding options and warrants, securities issued for compensation;
or (b) file or caused to be filed any registration statement relating to the offering of any shares of our capital stock or any
securities convertible into or exercisable or exchangeable for shares of our capital stock, other than pursuant to a registration
statement on Form S-8 for employee benefit plans.
Our officers and directors have also
agreed, subject to limited exceptions, for a period of 60 days after the date of the placement agency agreement, not to offer,
sell, contract to sell, pledge, grant any option to purchase, make any short sale or otherwise dispose of, directly or indirectly
any shares of common stock or any securities convertible into or exchangeable for our common stock either owned as of the date
of the placement agency agreement or thereafter acquired without the prior written consent of the placement agent. The placement
agents may, in their sole discretion and at any time or from time to time before the termination of the lock-up period, without
notice, release all or any portion of the securities subject to lock-up agreements.
With certain exceptions, Dawson James Securities,
Inc. shall be entitled to the same fees as provided in this offering with respect to any public or private offering or other financing
or capital-raising transaction of any kind to the extent that such financing or capital is provided to us by investors whom Dawson
had introduced to us or that participated in this offering, if such financing is consummated at any time within the 6-month period
following completion of the offering, subject to any rights already granted to Benchmark. We also granted Dawson James the right
of first refusal to act as lead managing underwriter and sole bookrunner, or as lead placement agent, for any and all future equity,
equity-linked or convertible debt, excluding revolving loan debt offerings, at the market sales or funds from certain purchasers,
during such 6-months period, of the Company, or any successor to or any subsidiary of the Company, subject to rights already granted
to Benchmark.
Upon closing, we will deliver to each purchaser
delivering funds the number of shares of common stock purchased by such purchaser in electronic format.
Dawson James Securities, Inc., its officers
and registered representatives may participate in this offering on the same terms and conditions as the investors in this offering.
Fees and Expenses
Upon the closing of this offering, we have
agreed to pay the Placements Agent (i) a cash fee equal to 8% of the aggregate gross proceeds raised in this offering, and (ii)
warrants to purchase that number of shares of our common stock equal to an aggregate of 5% of the shares of common stock sold
in the offering, in each case excluding any sales of securities to certain purchasers. The warrants will be exercisable at any
time and from time to time, in whole or in part, during the period commencing six months following the commencement of sales in
this offering, and ending five years from such date. The warrants provide for a cashless exercise provision, piggyback registration
rights and customary anti-dilution provisions (for stock dividends and splits and recapitalizations) consistent with FINRA Rule
5110. The warrants and the underlying securities are deemed compensation by FINRA, and are therefore subject to FINRA Rule 5110(g)(1).
In accordance with FINRA Rule 5110(g)(1), neither the warrants nor any securities issued upon exercise of the warrants may be sold,
transferred, assigned, pledged, or hypothecated, or be the subject of any hedging, short sale, derivative, put, or call transaction
that would result in the effective economic disposition of such securities by any person for a period of 180 days immediately following
the date of effectiveness or commencement of sales of the offering pursuant to which the warrants are being issued, except the
transfer of any security: (i) by operation of law or by reason of reorganization of our company; (ii) to any FINRA member firm
participating in this offering and the officers or partners thereof, if all securities so transferred remain subject to the lock-up
restriction described above for the remainder of the time period; (iii) if the aggregate amount of our securities held by either
a placement agent or a related person do not exceed 1% of the securities being offered; (iv) that is beneficially owned on a pro-rata
basis by all equity owners of an investment fund, provided that no participating member manages or otherwise directs investments
by the fund, and participating members in the aggregate do not own more than 10% of the equity in the fund; or (v) the exercise
or conversion of any security, if all securities received remain subject to the lock-up restriction set forth above for the remainder
of the time period.
We have also agreed to reimburse the placement
agents for certain expenses, including up to $65,000 for diligence and legal expenses.
We estimate the total
expenses of this offering which will be payable by us will be approximately $[______]. Assuming we complete the maximum offering,
after deducting the estimated offering expenses, we expect the net proceeds from this offering to be approximately $[____].
Indemnification
We have agreed to indemnify the placement agents
and specified other persons against certain liabilities relating to or arising out of the placement agents’ activities under
the placement agent agreement and to contribute to payments that the placement agent may be required to make in respect of such
liabilities.
Listing
Our shares of common stock are listed on
the Nasdaq Capital Market under the symbol “SNOA.”
Financial Services Agreement
We entered into a financial services
agreement with The Benchmark Company, LLC dated November 1, 2017, as amended. Pursuant to the financial services agreement Benchmark
will provide us with financial advisory services and guidance, such as attaining research coverage, complementing our IR efforts
by organizing road shows and investor meetings, and inviting us to investor conferences. We agreed to pay Benchmark a monthly cash
retainer of $10,000 and expenses up to an amount of $4,000.
LEGAL MATTERS
The validity of the issuance of the
common stock offered by this prospectus will be passed upon for us by Trombly Business Law, PC. Schiff Hardin LLP, Washington
DC, is counsel for the placement agents in connection with this offering.
EXPERTS
The consolidated financial statements
of Sonoma Pharmaceuticals, Inc. appearing in Sonoma Pharmaceuticals, Inc.’s annual report on Form 10-K for
the year ended March 31, 2017, filed on June 28, 2017, have been audited by Marcum LLP, an independent registered public accounting
firm, as set forth in their report included therein, and incorporated herein by reference. Such consolidated financial statements
are incorporated herein by reference in reliance upon such report given on the authority of such firm as experts in accounting
and auditing.
WHERE YOU CAN FIND
MORE INFORMATION
We file annual, quarterly and current
reports, proxy statements and other information with the SEC. You may read and copy the registration statement and any document
we file with the SEC at the SEC’s Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549. You may obtain information
on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. The SEC also maintains a website that contains
reports, proxy and information statements and other information regarding companies, such as ours, that file documents electronically
with the SEC. The address of the SEC’s website is www.sec.gov. The information on the SEC’s website is not part of
this prospectus, and any references to this website or any other website are inactive textual references only.
This prospectus is part of a registration
statement on Form S-3 that we filed with the SEC to register the securities to be offered hereby. This prospectus does not contain
all of the information included in the registration statement, including certain exhibits and schedules. You may obtain the registration
statement and exhibits to the registration statement from the SEC at the address listed above or from the SEC’s website listed
above. In addition to the foregoing, we maintain a website at www.sonomapharma.com. Our website content is made available for informational
purposes only. It should neither be relied upon for investment purposes nor is it incorporated by reference into this prospectus.
We make available at www.sonomapharma.com copies of our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current
Reports on Form 8-K and any amendments to such document as soon as practicable after we electronically file such material with
or furnish such documents to the SEC.
INCORPORATION OF
CERTAIN DOCUMENTS BY REFERENCE
The SEC permits us to “incorporate
by reference” the information contained in documents we file with the SEC, which means that we can disclose important information
to you by referring you to those documents rather than by including them in this prospectus. Information that is incorporated by
reference is considered to be part of this prospectus and you should read it with the same care that you read this prospectus.
Later information that we file with the SEC will automatically update and supersede the information that is either contained, or
incorporated by reference, in this prospectus, and will be considered to be a part of this prospectus from the date those documents
are filed. We have filed with the SEC, and incorporate by reference the following in this prospectus:
|
|
·
|
our Quarterly Report on Form 10-Q for the quarter ended June 30, 2017
, filed on August 8, 2017, our
Quarterly Report on Form 10-Q for the quarter ended September 30, 2017,
filed on November 14, 2017 and
our Quarterly Report on Form 10-Q for the quarter ended December 31, 2017,
filed on February 14, 2018;
|
In addition, all documents that we file
with the SEC pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Securities and Exchange Act of 1934, as amended, after the date
of the initial registration statement of which this prospectus is a part and prior to the effectiveness of the registration statement
as well as all such documents that we file with the SEC after the date of this prospectus and before the termination of the offering
of our securities shall be deemed incorporated by reference into this prospectus and to be a part of this prospectus from the respective
dates of filing such documents. Unless specifically stated to the contrary, none of the information that we disclose under Items
2.02 or 7.01 of any Current Report on Form 8-K that we may from time to time furnish to the SEC will be incorporated by reference
into, or otherwise included in, this prospectus.
You may request a copy of any or all
of the documents incorporated by reference but not delivered with this prospectus, at no cost, by writing or telephoning us at
the following address and number: Investor Relations, Sonoma Pharmaceuticals, Inc., 1129 N. McDowell Blvd., Petaluma, California
94954, telephone (707) 283-0550. We will not, however, send exhibits to those documents, unless the exhibits are specifically
incorporated by reference in those documents.
DISCLOSURE OF COMMISSION
POSITION ON INDEMNIFICATION
Insofar as indemnification for liabilities
arising under the Securities Act, as amended, may be permitted to directors, officers, and controlling persons of the registrant
pursuant to the Company’s constituent documents, or otherwise, the registrant has been advised that in the opinion of the
SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event
that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid
by a director, officer, or controlling person in the successful defense of any action, suit, or proceeding) is asserted by such
director, officer, or controlling person connected with the securities being registered, we will, unless in the opinion of our
counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether
such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication
of such issue.
Filed Pursuant to Rule 424(b)(3
)
Registration No. 333-221477
PROSPECTUS
$75,000,000

SONOMA PHARMACEUTICALS, INC.
Common Stock
Preferred Stock
Warrants
Units
We may, from time to time, offer and
sell common stock, preferred stock or warrants, either separately or in units, in one or more offerings. The preferred stock and
warrants may be convertible into or exercisable or exchangeable for common or preferred stock. We will specify in the accompanying
prospectus supplement more specific information about any such offering. The aggregate initial offering price of all securities
sold under this prospectus will not exceed $75,000,000, including the U.S. dollar equivalent if the public offering of any
such securities is denominated in one or more foreign currencies, foreign currency units or composite currencies.
We may offer these securities independently
or together in any combination for sale directly to investors or through underwriters, dealers or agents. We will set forth the
names of any underwriters, dealers or agents and their compensation in the accompanying prospectus supplement.
This prospectus may not be used to sell
any of these securities unless accompanied by a prospectus supplement.
Our common stock is traded on the Nasdaq
Capital Market under the symbol “SNOA.” On November 24, 2017, the last reported sale price for our common stock was
$5.13 per share. The aggregate market value of our outstanding voting and non-voting equity held by non-affiliates on November
24, 2017 was $21,638,222 based on a share price of $5.13. We have not offered any securities pursuant to General Instruction I.B.6.
of Form S-3 during the 12 calendar months prior to and including the date hereof.
Investing in our securities involves
a high degree of risk. See the section entitled “
Risk Factors
” beginning on page 4.
Neither the Securities and Exchange
Commission nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy
of this prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus is November
28, 2017.
TABLE OF CONTENTS
You should rely only on the information
incorporated by reference or provided in this prospectus, any prospectus supplement and the registration statement. We have not
authorized anyone else to provide you with different information. If anyone provides you with different or inconsistent information,
you should not rely on it. We are not making an offer to sell these securities in any state where the offer or sale is not permitted.
You should assume that the information in this prospectus and any prospectus supplement, or incorporated by reference, is accurate
only as of the dates of those documents. Our business, financial condition, results of operations and prospects may have changed
since those dates.
ABOUT THIS PROSPECTUS
This prospectus is part of a registration
statement that we filed with the Securities and Exchange Commission, or SEC, using a “shelf” registration, or continuous
offering, process. Under this shelf registration process, we may, from time to time, issue and sell any combination of preferred
stock, common stock or warrants, either separately or in units, in one or more offerings with a maximum aggregate offering price
of $75,000,000, including the U.S. dollar equivalent if the public offering of any such securities is denominated in one or more
foreign currencies, foreign currency units or composite currencies.
This prospectus provides you with a
general description of the securities we may offer. Each time we sell securities, we will provide a prospectus supplement that
will contain specific information about the terms of that offering and the offered securities. Any prospectus supplement may also
add, update or change information contained in this prospectus. Any statement that we make in this prospectus will be modified
or superseded by any inconsistent statement made by us in a prospectus supplement. The registration statement we filed with the
SEC includes exhibits that provide more detail of the matters discussed in this prospectus. You should read this prospectus and
the related exhibits filed with the SEC and any prospectus supplement, together with additional information described under the
heading “
Where You Can Find More Information
,” before making your investment decision.
THIS PROSPECTUS MAY NOT BE USED TO CONSUMMATE A SALE OF
SECURITIES UNLESS IT IS ACCOMPANIED BY A PROSPECTUS SUPPLEMENT.
Neither we, nor any agent, underwriter
or dealer has authorized any person to give any information or to make any representation other than those contained or incorporated
by reference in this prospectus, any applicable prospectus supplement prepared by or on behalf of us or to which we have referred
you. This prospectus or any applicable supplement to this prospectus do not constitute an offer to sell or the solicitation of
an offer to buy any securities other than the registered securities to which they relate, nor do this prospectus or any applicable
supplement to this prospectus constitute an offer to sell or the solicitation of an offer to buy securities in any jurisdiction
to any person to whom it is unlawful to make such offer or solicitation in such jurisdiction.
You should not assume that the information
contained in this prospectus or any applicable prospectus supplement is accurate on any date subsequent to the date set forth on
the front of the document or that any information we have incorporated by reference is correct on any date subsequent to the date
of the document incorporated by reference, even though this prospectus or any applicable prospectus supplement is delivered, or
securities are sold, on a later date.
PROSPECTUS SUMMARY
This summary highlights information
contained elsewhere in this prospectus or incorporated by reference. This summary does not contain all of the information you should
consider before buying shares of our common stock, preferred stock, warrants, or units or any combination of these securities.
You should read the entire prospectus carefully, especially the risks of investing in our securities that we describe under “Risk
Factors” and our consolidated financial statements appearing in our annual and periodic reports incorporated in this prospectus
by reference, before deciding to invest in our securities. Unless the context requires otherwise, references to “Sonoma,”
“the Company,” “the Registrant,” “we,” “our” and “us” refer to Sonoma
Pharmaceuticals, Inc.
Company Overview
We are a specialty pharmaceutical company
dedicated to identifying, developing and commercializing unique, differentiated therapies to millions of patients living with chronic
skin conditions. We believe our products, which are sold throughout the United States and internationally, have improved patient
outcomes for more than five million patients globally by treating and reducing certain topical skin diseases including acne, atopic
dermatitis, scarring, infections, itch, pain and harmful inflammatory responses.
Corporate Information
We incorporated under the laws of the
State of California in April 1999 as Micromed Laboratories, Inc. In August 2001, we changed our name to Oculus Innovative Sciences,
Inc. In December 2006, we reincorporated under the laws of the State of Delaware. On December 6, 2016, we changed our name from
Oculus Innovative Sciences, Inc. to Sonoma Pharmaceuticals, Inc. Our principal executive offices are located at 1129 N. McDowell
Blvd., Petaluma, California, 94954, and our telephone number is (707) 283-0550. We have two active wholly-owned subsidiaries: Oculus
Technologies of Mexico, S.A. de C.V., organized in Mexico; and Sonoma Pharmaceuticals Netherlands, B.V., organized in the Netherlands.
The Securities We May Offer
We may offer and sell, from time to
time, in one or more offerings, any combination of shares of our common stock and preferred stock, and warrants to purchase any
of such securities, up to a total aggregate offering price of $75,000,000 under this prospectus, together with any applicable prospectus
supplement, at prices and on terms to be determined by market conditions at the time of the relevant offering. This prospectus
provides you with a general description of the securities we may offer. Each time we offer a type or series of securities under
this prospectus, we will provide a prospectus supplement that will describe the specific amounts, prices and other important terms
of the securities, including, to the extent applicable:
|
|
·
|
designation or classification;
|
|
|
·
|
aggregate offering price;
|
|
|
·
|
rates and times of payment of dividends, if any;
|
|
|
·
|
redemption, conversion, exchange or sinking fund terms, if any;
|
|
|
·
|
conversion or exchange prices or rates, if any, and, if applicable, any provisions for changes to or adjustments in the conversion or exchange prices or rates and in the securities or other property receivable upon conversion or exchange;
|
|
|
·
|
ranking, if applicable;
|
|
|
·
|
restrictive covenants, if any;
|
|
|
·
|
voting or other rights, if any; and
|
|
|
·
|
important United States federal income tax considerations.
|
The prospectus supplement may also add,
update or change information contained in this prospectus or in documents we have incorporated by reference. However, no prospectus
supplement will offer a security that is not registered and described in this prospectus at the time of the effectiveness of the
registration statement of which this prospectus is a part.
This prospectus may not be used to
consummate a sale of securities unless it is accompanied by a prospectus supplement.
We may sell the securities directly
to investors or through underwriters, dealers or agents. We, and our underwriters or agents, reserve the right to accept or reject
all or part of any proposed purchase of securities. If we do offer securities through underwriters or agents, we will include in
the applicable prospectus supplement:
|
|
·
|
the names of those underwriters or agents;
|
|
|
·
|
applicable fees, discounts and commissions to be paid to them;
|
|
|
·
|
details regarding over-allotment options, if any; and
|
|
|
·
|
the estimated net proceeds to us.
|
Risk Factors
Investing in our securities involves
a high degree of risk. You should carefully review the risks and uncertainties described under the heading “Risk Factors”
contained in the applicable prospectus supplement, and under similar headings in our Annual Report on Form 10-K for the year ended
March 31, 2017, as updated by our annual, quarterly and other reports and documents that are incorporated by reference into this
prospectus, before deciding whether to purchase any of the securities being registered pursuant to the registration statement of
which this prospectus is a part. Each of the risk factors could adversely affect our business, operating results and financial
condition, as well as adversely affect the value of an investment in our securities, and the occurrence of any of these risks might
cause you to lose all or part of your investment. Additional risks not presently known to us or that we currently believe are immaterial
may also significantly impair our business operations.
Where You Can Find More Information
We are subject to the information requirements
of the Exchange Act. Accordingly, we file annual, quarterly and current reports, proxy statements as may be required and other
information with the SEC and filed a registration statement on Form S-3 under the Securities Act relating to the securities offered
by this prospectus. This prospectus, which forms part of the registration statement, does not contain all of the information included
in the registration statement. For further information, you should refer to the registration statement and its exhibits.
You may read and copy the registration
statement and any document we file with the SEC at the SEC’s Public Reference Room at 100 F Street, N.E., Washington, D.C.
20549. Please call the SEC at 1-800-SEC-0330 for further information on the operation of the Public Reference Room. You can also
review our filings by accessing the website maintained by the SEC at www.sec.gov. The site contains reports, proxy and information
statements and other information regarding issuers that file electronically with the SEC. In addition to the foregoing, we maintain
a website at www.sonomapharma.com. Our website content is made available for informational purposes only. It should neither be
relied upon for investment purposes nor is it incorporated by reference into this prospectus. We make available at www.sonomapharma.com
copies of our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K and any amendments to
such document as soon as practicable after we electronically file such material with or furnish such documents to the SEC.
OUR COMPANY
We are a specialty pharmaceutical company
dedicated to identifying, developing and commercializing unique, differentiated therapies to millions of patients living with chronic
skin conditions. We believe our products, which are sold throughout the United States and internationally, have improved patient
outcomes for more than five million patients globally by treating and reducing certain topical skin diseases including acne, atopic
dermatitis, scarring, infections, itch, pain and harmful inflammatory responses.
We incorporated under the laws of the
State of California in April 1999 as Micromed Laboratories, Inc. In August 2001, we changed our name to Oculus Innovative Sciences,
Inc. In December 2006, we reincorporated under the laws of the State of Delaware. On December 6, 2016, we changed our name from
Oculus Innovative Sciences, Inc. to Sonoma Pharmaceuticals, Inc. Our principal executive offices are located at 1129 N. McDowell
Blvd., Petaluma, California, 94954, and our telephone number is (707) 283-0550. We have two active wholly-owned subsidiaries: Oculus
Technologies of Mexico, S.A. de C.V., organized in Mexico; and Sonoma Pharmaceuticals Netherlands, B.V., organized in the Netherlands.
RISK FACTORS
Investing in our securities involves
a high degree of risk. You should carefully review the risks and uncertainties described under the heading “Risk Factors”
contained in the applicable prospectus supplement, and under similar headings in our Annual Report on Form 10-K for the year ended
March 31, 2017, as updated by our quarterly and other reports and documents that are incorporated by reference into this prospectus,
before deciding whether to purchase any of the securities being registered pursuant to the registration statement of which this
prospectus is a part. Each of the risk factors could adversely affect our business, operating results and financial condition,
as well as adversely affect the value of an investment in our securities, and the occurrence of any of these risks might cause
you to lose all or part of your investment. Additional risks not presently known to us or that we currently believe are immaterial
may also significantly impair our business operations.
CAUTIONARY NOTE REGARDING
FORWARD-LOOKING STATEMENTS
When used in this prospectus or any
prospectus supplement, the words “expect,” “believe,” “anticipate,” “estimate,”
“may,” “could,” “intend,” and similar expressions are intended to identify forward-looking
statements. These statements are subject to known and unknown risks and uncertainties that could cause actual results to differ
materially from those projected or otherwise implied by the forward-looking statements. These forward-looking statements speak
only as of the date of this prospectus. Given these risks and uncertainties, you should not place undue reliance on these forward-looking
statements. We have discussed many of these risks and uncertainties in greater detail in this prospectus under the heading “Risk
Factors.” Additional cautionary statements or discussions of risks and uncertainties that could affect our results or the
achievement of the expectations described in forward-looking statements may also be contained in the documents we incorporate by
reference into this prospectus.
These forward-looking statements speak
only as of the date of this prospectus. We expressly disclaim any obligation or undertaking to release publicly any updates or
revisions to any forward-looking statements contained herein to reflect any change in our expectations with regard thereto or any
change in events, conditions or circumstances on which any such statement is based. You should, however, review additional disclosures
we make in our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, and Current Reports on
Form 8-K filed with the SEC.
DILUTION
We will set forth in a prospectus supplement
the following information regarding any material dilution of the equity interests of investors purchasing securities in an offering
under this prospectus:
|
|
·
|
|
the net tangible book value per share of our equity securities before and after the offering;
|
|
|
·
|
|
the amount of the increase in such net tangible book value per share attributable to the cash payments made by purchasers in the offering; and
|
|
|
·
|
|
the amount of the immediate dilution from the public offering price which will be absorbed by such purchasers.
|
USE OF PROCEEDS
Unless we state otherwise in the accompanying
prospectus supplement, we intend to use the net proceeds from the sale of the securities offered by this prospectus for general
corporate purposes. General corporate purposes may include additions to working capital, research and development, financing of
capital expenditures, and future acquisitions and strategic investment opportunities. Pending the application of net proceeds,
we expect to invest the net proceeds in interest-bearing securities.
PLAN OF DISTRIBUTION
We may sell the securities offered by
this prospectus to one or more underwriters or dealers for public offering and sale by them or to investors directly or through
agents. The accompanying prospectus supplement will set forth the terms of the offering and the method of distribution and will
identify any firms acting as underwriters, dealers or agents in connection with the offering, including:
|
|
·
|
the name or names of any underwriters, dealers or agents;
|
|
|
·
|
the purchase price of the securities and the proceeds to us from the sale;
|
|
|
·
|
any underwriting discounts and other items constituting compensation to underwriters, dealers or agents;
|
|
|
·
|
any public offering price;
|
|
|
·
|
any discounts or concessions allowed or re-allowed or paid to dealers; and
|
|
|
·
|
any securities exchange or market on which the securities offered in the prospectus supplement may be listed.
|
Only those underwriters identified in
such prospectus supplement are deemed to be underwriters in connection with the securities offered in the prospectus supplement.
The distribution of the securities may
be effected from time to time in one or more transactions at a fixed price or prices, which may be changed, or at prices determined
as the applicable prospectus supplement specifies. The securities may be sold through a rights offering, forward contracts or similar
arrangements. In connection with the sale of the securities, underwriters, dealers or agents may be deemed to have received compensation
from us in the form of underwriting discounts or commissions and also may receive commissions from securities purchasers for whom
they may act as agent. Underwriters may sell the securities to or through dealers, and the dealers may receive compensation in
the form of discounts, concessions or commissions from the underwriters or commissions from the purchasers for whom they may act
as agent. Some of the underwriters, dealers or agents who participate in the securities distribution may engage in other transactions
with, and perform other services for, us or our subsidiaries in the ordinary course of business.
We will provide in the applicable prospectus
supplement information regarding any underwriting discounts or other compensation that we pay to underwriters or agents in connection
with the securities offering, and any discounts, concessions or commissions which underwriters allow to dealers. Underwriters,
dealers and agents participating in the securities distribution may be deemed to be underwriters, and any discounts and commissions
they receive and any profit they realize on the resale of the securities may be deemed to be underwriting discounts and commissions
under the Securities Act of 1933. Underwriters and their controlling persons, dealers and agents may be entitled, under agreements
entered into with us, to indemnification against and contribution toward specific civil liabilities, including liabilities under
the Securities Act.
The securities may or may not be listed
on a national securities exchange. In connection with an offering, the underwriters may purchase and sell securities in the open
market. These transactions may include short sales, stabilizing transactions and purchases to cover positions created by short
sales. Short sales involve the sale by the underwriters of a greater number of securities than they are required to purchase in
an offering. Stabilizing transactions consist of bids or purchases made for the purpose of preventing or retarding a decline in
the market price of the securities while an offering is in progress. The underwriters also may impose a penalty bid. This occurs
when a particular underwriter repays to the underwriters a portion of the underwriting discount received by it because the underwriters
have repurchased securities sold by or for the account of that underwriter in stabilizing or short-covering transactions. These
activities by the underwriters may stabilize, maintain or otherwise affect the market price of the securities. As a result, the
price of the securities may be higher than the price that otherwise might exist in the open market. If these activities are commenced,
they may be discontinued by the underwriters at any time.
DESCRIPTION OF COMMON
STOCK
This section describes the general terms
and provisions of the shares of our common stock. This description is only a summary and is qualified in its entirety by reference
to the description of our common stock incorporated by reference in this prospectus. A copy of our restated certificate of incorporation,
as amended, and our amended and restated bylaws, as amended, has been incorporated by reference from our filings with the SEC as
exhibits to the registration statement of which this prospectus forms a part. Our common stock and the rights of the holders of
our common stock are subject to the applicable provisions of the Delaware General Corporation Law, which we refer to as “Delaware
law,” our restated certificate of incorporation, as amended, our amended and restated bylaws, as amended, the rights of the
holders of our preferred stock, if any, as well as some of the terms of our outstanding indebtedness. See “
Where You Can
Find More Information
.”
We have 12,000,000 shares of authorized
common stock, par value $0.0001 per share. As of November 20, 2017, there were 4,323,831 shares of common stock issued and outstanding.
All outstanding shares of common stock are fully paid and nonassessable, and the shares of common stock offered, when issued, will
be fully paid and nonassessable.
The following description of our common
stock, and any description of our common stock in a prospectus supplement, may not be complete and is subject to, and qualified
in its entirety by reference to, Delaware law and the actual terms and provisions contained in our restated certificate of incorporation
and our amended and restated bylaws, each as amended from time to time.
Voting Rights:
Unless
otherwise provided by law or provided in our restated certificate of incorporation, as amended, each holder of common stock is
entitled to one vote for each share of common stock held on all matters submitted to a vote of stockholders. At a meeting of stockholders
at which a quorum is present, an affirmative vote of the majority of the shares entitled to vote on a matter and that are represented
either in person or by proxy decides all questions, unless the question is one upon which by express provision of law or our restated
certificate of incorporation or of our amended and restated bylaws, a different vote is required.
Dividends
:
Dividends upon
the capital stock of the corporation may be declared by our board of directors at any regular or special meeting or by unanimous
written consent, subject to any restrictions contained under Delaware law or the provisions of the restated certificate of incorporation,
as amended, if any. However, before payment of any dividend, the board of directors may set aside out of the funds of the corporation
available for dividends such sum or sums that the directors from time to time think proper as a reserve or reserves. If and when
a dividend is declared by the board of directors, the holders of shares of common stock shall be entitled to receive out of the
assets of the corporation which are by law available, dividends payable in cash, in property or in shares of capital stock, subject
to the preferential rights of the holders of preferred stock.
Preemptive Rights
:
The
holders of common stock do not have preemptive or conversion rights or other subscription rights.
Redemption
:
Shares of
our common stock are not subject to redemption by operation of a sinking fund or otherwise.
Liquidation Rights
:
Upon
our liquidation, dissolution or winding-up, and after distribution in full of preferential amounts, if any, to be distributed to
the holders of shares of preferred stock, the holders of common stock are entitled, unless otherwise provided by law or provided
in our restated certificate of incorporation, as amended, to receive all of the remaining assets of the corporation of whatever
kind available for distribution to stockholders ratably in proportion to the number of shares of common stock held by them respectively.
Listing:
Our common stock
is traded on the Nasdaq Capital Market under the symbol “SNOA.”
Transfer Agent and Registrar:
The transfer agent for our common stock is Computershare, Inc. located at 462 South 4
th
Street, Suite 1600, Louisville,
KY 40202. Its telephone number is 1-888-647-8901.
DESCRIPTION
OF Preferred Stock
This section describes the general terms
of our preferred stock to which any prospectus supplement may relate. A prospectus supplement will describe the terms relating
to any preferred stock to be offered by us in greater detail and may provide information that is different from terms described
in this prospectus. A copy of our restated certificate of incorporation, as amended, and our amended and restated bylaws, as amended,
has been incorporated by reference from our filings with the SEC as exhibits to the registration statement of which this prospectus
forms a part. A certificate of designation or amendment to the restated certificate of incorporation, as amended, will specify
the terms of the preferred stock being offered, and will be filed or incorporated by reference as an exhibit to the registration
statement before the preferred stock is issued. The following description of our preferred stock, and any description of the preferred
stock in a prospectus supplement may not be complete and is subject to, and qualified in its entirety by reference to, Delaware
law and the actual terms and provisions contained in our restated certificate of incorporation and our amended and restated bylaws,
each as amended from time to time.
Under our restated certificate of incorporation,
as amended, we have the authority to issue 714,286 shares of preferred stock, par value $0.0001 per share, which are issuable in
series on terms to be determined by our board of directors. Accordingly, our board of directors is authorized, without action by
the stockholders, to issue preferred stock from time to time with such dividend, liquidation, conversion, voting, redemption, sinking
fund and other rights and restrictions as it may determine. All shares of any one series of our preferred stock will be identical,
except that shares of any one series issued at different times may differ as to the dates from which dividends may be cumulative,
as described in the applicable prospectus supplement. As of the date of this prospectus, there were no outstanding shares of our
preferred stock.
Terms of Preferred Stock
:
Unless provided in a prospectus supplement, the shares of our preferred stock to be issued will have no preemptive rights.
Any prospectus supplement offering our preferred stock will furnish the following information with respect to the preferred stock
offered by that prospectus supplement:
|
|
·
|
the distinctive designation of each series and the number of shares that will constitute the series;
|
|
|
·
|
the voting rights, if any, of shares of the series and the terms and conditions of the voting rights;
|
|
|
·
|
the dividend rate on the shares of the series, the dates on which dividends are payable, any restriction, limitation or condition upon the payment of dividends, whether dividends will be cumulative, and the dates from and after which dividends shall accumulate;
|
|
|
·
|
the prices at which, and the terms and conditions on which, the shares of the series may be redeemed, if the shares are redeemable;
|
|
|
·
|
the terms and conditions of a sinking or purchase fund for the purchase or redemption of shares of the series, if such a fund is provided;
|
|
|
·
|
any preferential amount payable upon shares of the series in the event of the liquidation, dissolution or winding up of, or upon the distribution of any of our assets; and
|
|
|
·
|
the prices or rates of conversion or exchange at which, and the terms and conditions on which, the shares of the series may be converted or exchanged into other securities, if the shares are convertible or exchangeable.
|
If our board of directors decides to
issue any shares of preferred stock, it may discourage or make more difficult a merger, tender offer, business combination or proxy
contest, assumption of control by a holder of a large block of our securities, or the removal of incumbent management, even if
these events were favorable to the interests of stockholders. Our board of directors, without stockholder approval, may issue preferred
stock with voting and conversion rights and dividend and liquidation preferences that may adversely affect the holders of our other
equity or debt securities.
The particular terms of any series of
preferred stock, and the transfer agent and registrar for that series, will be described in a prospectus supplement. All preferred
stock offered, when issued, will be fully paid and nonassessable. Any material United States federal income tax consequences
and other special considerations with respect to any preferred stock offered under this prospectus will also be described in the
applicable prospectus supplement.
Description
of Warrants
We may issue warrants for the purchase
of preferred stock, common stock, or any combination thereof. We may issue warrants independently or together with any other securities
offered by any prospectus supplement and may be attached to or separate from the other offered securities. Each series of warrants
will be issued under a separate warrant agreement to be entered into by us with a warrant agent. The warrant agent will act solely
as our agent in connection with the warrants and will not assume any obligation or relationship of agency or trust for or with
any holders or beneficial owners of warrants. Further terms of the warrants and the applicable warrant agreements will be set forth
in the applicable prospectus supplement.
The applicable prospectus supplement
relating to any particular issue of warrants will describe the terms of the warrants, including, as applicable, the following:
|
|
·
|
the title of the warrants;
|
|
|
·
|
the aggregate number of the warrants;
|
|
|
·
|
the price or prices at which the warrants will be issued;
|
|
|
·
|
the designation, terms and number of shares of preferred stock or common stock purchasable upon exercise of the warrants;
|
|
|
·
|
the designation and terms of the offered securities, if any, with which the warrants are issued and the number of the warrants issued with each offered security;
|
|
|
·
|
the date, if any, on and after which the warrants and the related preferred stock or common stock will be separately transferable;
|
|
|
·
|
the price at which each share of preferred stock or common stock purchasable upon exercise of the warrants may be purchased;
|
|
|
·
|
the date on which the right to exercise the warrants shall commence and the date on which that right shall expire;
|
|
|
·
|
the minimum or maximum amount of the warrants which may be exercised at any one time;
|
|
|
·
|
information with respect to book-entry procedures, if any;
|
|
|
·
|
a discussion of certain U.S. federal income tax considerations; and
|
|
|
·
|
any other terms of the warrants, including terms, procedures and limitations relating to the exchange and exercise of the warrants.
|
We and the warrant agent may amend or
supplement the warrant agreement for a series of warrants without the consent of the holders of the warrants issued thereunder
to effect changes that are not inconsistent with the provisions of the warrants and that do not materially and adversely affect
the interests of the holders of the warrants.
Description
of Units
The following description, together
with the additional information we include in any applicable prospectus supplement, summarizes the material terms and provisions
of the units that we may offer under this prospectus. Units may be offered independently or together with common or preferred stock,
and warrants offered by any prospectus supplement, and may be attached to or separate from those securities. While the terms we
have summarized below will generally apply to any future units that we may offer under this prospectus, we will describe the particular
terms of any series of units that we may offer in more detail in the applicable prospectus supplement. The terms of any units offered
under a prospectus supplement may differ from the terms described below.
We will incorporate by reference into
the registration statement of which this prospectus forms a part the form of unit agreement, including a form of unit certificate,
if any, that describes the terms of the series of units we are offering before the issuance of the related series of units. The
following summaries of material provisions of the units and the unit agreements are subject to, and qualified in their entirety
by reference to, all the provisions of the unit agreement applicable to a particular series of units. We urge you to read the applicable
prospectus supplements related to the units that we sell under this prospectus, as well as the complete unit agreements that contain
the terms of the units.
We may issue units consisting of one
or more shares of common stock or preferred stock, warrants or any combination of such securities. Each unit will be issued so
that the holder of the unit is also the holder of each security included in the unit.
Additionally, we will describe in the applicable prospectus
supplement the terms of the series of units, including the following:
|
|
·
|
the designation and terms of the units and the securities included in the units;
|
|
|
·
|
any provision for the issuance, payment, settlement, transfer or exchange of the units;
|
|
|
·
|
the date, if any, on and after which the units may be transferable separately;
|
|
|
·
|
whether we will apply to have the units traded on a securities exchange or securities quotation system;
|
|
|
·
|
any material United States federal income tax consequences; and
|
|
|
·
|
how, for United States federal income tax purposes, the purchase price paid for the units is to be allocated among the component securities.
|
CERTAIN PROVISIONS
OF DELAWARE LAW AND OUR CHARTER
AND BYLAWS
The following paragraphs summarize certain
provisions of Delaware law and our restated certificate of incorporation, as amended, and our amended and restated bylaws, as amended.
The summary does not purport to be complete and is subject to and qualified in its entirety by reference to Delaware law and to
our restated certificate of incorporation, as amended, and our amended and restated bylaws, as amended, copies of which are on
file with the SEC as exhibits to reports previously filed by us. See “
Where You Can Find More Information
.”
Delaware Law
We are subject to the provisions of
Section 203 of the Delaware General Corporation Law regulating corporate takeovers. In general, this section prohibits a Delaware
corporation from engaging in any business combination with any interested stockholder for a period of three years following the
date that the stockholder became an interested stockholder, unless:
|
|
·
|
the transaction is approved by the board of directors prior to the time that the interested stockholder became an interested stockholder;
|
|
|
|
|
|
|
·
|
upon consummation of the transaction which resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced; or
|
|
|
|
|
|
|
·
|
at or subsequent to such time that the stockholder became an interested stockholder, the business combination is approved by the board of directors and authorized at a meeting of stockholders by at least two-thirds of the outstanding voting stock that is not owned by the interested stockholder.
|
Section 203 defines “business combination”
to include the following:
|
|
·
|
any merger or consolidation involving the corporation and the interested stockholder;
|
|
|
|
|
|
|
·
|
any sale, lease, exchange, mortgage, pledge, transfer or other disposition of 10% or more of the assets of the corporation involving the interested stockholder;
|
|
|
|
|
|
|
·
|
subject to certain exceptions, any transaction that results in the issuance or transfer by the corporation of any stock of the corporation to the interested stockholder;
|
|
|
|
|
|
|
·
|
any transaction involving the corporation that has the effect, directly or indirectly, of increasing the proportionate share of the stock of any class or series of the corporation beneficially owned by the interested stockholder; or
|
|
|
|
|
|
|
·
|
the receipt by the interested stockholder of the benefit, directly or indirectly, of any loans, advances, guarantees, pledges or other financial benefits provided by or through the corporation.
|
In general, Section 203 defines
an interested stockholder as any entity or person beneficially owning 15% or more of the outstanding voting stock of the corporation
and any entity or person affiliated with or controlling or controlled by any of these entities or persons.
A Delaware corporation may “opt
out” of these provisions either with an express provision in its original certificate of incorporation or in an amendment
to its certificate of incorporation or bylaws approved by its stockholders. However, we have not opted out, and do not currently
intend to opt out of, these provisions. The statute could prohibit or delay mergers or other takeover or change in control attempts
and, accordingly, may discourage attempts to acquire us.
Charter and Bylaws
Our restated certificate of incorporation,
as amended, and our amended and restated bylaws, as amended, provide that:
|
|
·
|
our amended and restated bylaws, as amended, may be amended or repealed only by the approval of at least sixty-six and two-thirds percent (66-2/3%) of the total number of directors then in office or the affirmative vote of the holders of at least sixty-six and two-thirds percent (66-2/3%) of the shares entitled to vote at an election of directors;
|
|
|
|
|
|
|
·
|
no action can be taken by stockholders except at an annual or special meeting of the stockholders called in accordance with our amended and restated bylaws, as amended, and stockholders may not act by written consent;
|
|
|
|
|
|
|
·
|
a stockholder must provide advance notice of stockholder proposals relating to the nomination of candidates for election as directors or new business to be brought before meetings of stockholders;
|
|
|
|
|
|
|
·
|
stockholders may not call special meetings of the stockholders or fill vacancies on the board;
|
|
|
|
|
|
|
·
|
the approval of holders of at least sixty-six and two-thirds percent (66-2/3%) of the shares entitled to vote at an election of directors is required to amend or repeal the provisions of our restated certificate of incorporation, as amended, regarding the inability of stockholders to take action by written consent;
|
|
|
|
|
|
|
·
|
our board of directors is authorized to issue preferred stock without stockholder approval; and
|
|
|
|
|
|
|
·
|
we will indemnify officers and directors against losses that they may incur in investigations and legal proceedings resulting from their services to us, which may include services in connection with takeover defense measures.
|
LEGAL MATTERS
The validity of any securities offered by this prospectus
will be passed upon for us by Trombly Business Law, PC.
EXPERTS
The consolidated financial statements
of Sonoma Pharmaceuticals, Inc. appearing in Sonoma Pharmaceuticals, Inc.’s annual report on Form 10-K for
the year ended March 31, 2017, filed on June 28, 2017, have been audited by Marcum LLP, an independent registered public accounting
firm, as set forth in their report included therein, and incorporated herein by reference. Such consolidated financial statements
are incorporated herein by reference in reliance upon such report given on the authority of such firm as experts in accounting
and auditing.
WHERE YOU CAN FIND
MORE INFORMATION
We file annual, quarterly and current
reports, proxy statements and other information with the SEC. You may read and copy the registration statement and any document
we file with the SEC at the SEC’s Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549. You may obtain information
on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. The SEC also maintains a web site that contains
reports, proxy and information statements and other information regarding companies, such as ours, that file documents electronically
with the SEC. The address of the SEC’s website is www.sec.gov. The information on the SEC’s website is not part of
this prospectus, and any references to this website or any other website are inactive textual references only.
This prospectus is part of a registration
statement on Form S-3 that we filed with the SEC to register the securities to be offered hereby. This prospectus does not contain
all of the information included in the registration statement, including certain exhibits and schedules. You may obtain the registration
statement and exhibits to the registration statement from the SEC at the address listed above or from the SEC’s website listed
above. In addition to the foregoing, we maintain a website at www.sonomapharma.com. Our website content is made available for informational
purposes only. It should neither be relied upon for investment purposes nor is it incorporated by reference into this prospectus.
We make available at www.sonomapharma.com copies of our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current
Reports on Form 8-K and any amendments to such document as soon as practicable after we electronically file such material with
or furnish such documents to the SEC.
INCORPORATION OF
CERTAIN DOCUMENTS BY REFERENCE
The SEC permits us to “incorporate
by reference” the information contained in documents we file with the SEC, which means that we can disclose important information
to you by referring you to those documents rather than by including them in this prospectus. Information that is incorporated by
reference is considered to be part of this prospectus and you should read it with the same care that you read this prospectus.
Later information that we file with the SEC will automatically update and supersede the information that is either contained, or
incorporated by reference, in this prospectus, and will be considered to be a part of this prospectus from the date those documents
are filed. We have filed with the SEC, and incorporate by reference the following in this prospectus:
In addition, all documents that we file
with the SEC pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Securities and Exchange Act of 1934, as amended, after the date
of the initial registration statement of which this prospectus is a part and prior to the effectiveness of the registration statement
as well as all such documents that we file with the SEC after the date of this prospectus and before the termination of the offering
of our securities shall be deemed incorporated by reference into this prospectus and to be a part of this prospectus from the respective
dates of filing such documents. Unless specifically stated to the contrary, none of the information that we disclose under Items
2.02 or 7.01 of any Current Report on Form 8-K that we may from time to time furnish to the SEC will be incorporated by reference
into, or otherwise included in, this prospectus.
You may request a copy of any or all
of the documents incorporated by reference but not delivered with this prospectus, at no cost, by writing or telephoning us at
the following address and number: Investor Relations, Sonoma Pharmaceuticals, Inc., 1129 N. McDowell Blvd., Petaluma, California
94954, telephone (707) 283-0550. We will not, however, send exhibits to those documents, unless the exhibits are specifically
incorporated by reference in those documents.
DISCLOSURE OF COMMISSION
POSITION ON INDEMNIFICATION
Insofar as indemnification for liabilities
arising under the Securities Act, as amended, may be permitted to directors, officers, and controlling persons of the registrant
pursuant to the Company’s constituent documents, or otherwise, the registrant has been advised that in the opinion of the
SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event
that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid
by a director, officer, or controlling person in the successful defense of any action, suit, or proceeding) is asserted by such
director, officer, or controlling person connected with the securities being registered, we will, unless in the opinion of our
counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether
such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication
of such issue.
$__________

COMMON STOCK
Prospectus Supplement
Sonoma Pharmaceuticals (NASDAQ:SNOA)
Historical Stock Chart
From Mar 2024 to Apr 2024
Sonoma Pharmaceuticals (NASDAQ:SNOA)
Historical Stock Chart
From Apr 2023 to Apr 2024