AMSTERDAM, Feb. 27, 2017 /PRNewswire/ -- Royal Philips
(NYSE:PHG AEX: PHIA), a global leader in health technology, today
announced 510(k) clearance from the U.S. Food and Drug
Administration (FDA) to market its ElastQ Imaging capability,
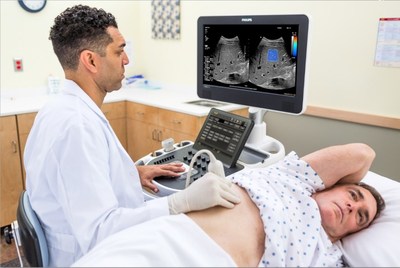
further expanding the functionalities of its EPIQ family of
ultrasound systems. ElastQ Imaging enables simultaneous imaging of
tissue and assessment of its stiffness, which is essential for the
diagnosis of various liver conditions. With ElastQ Imaging,
clinicians have a comprehensive solution to assess and diagnose
liver conditions without the pain or expense of a liver
biopsy. Using shear wave elastography to focus sound waves to
assess soft tissue stiffness, ElastQ Imaging is non-invasive,
reproducible and easily executed. Philips is at the forefront of
imaging technology innovation, and with this new capability, is
addressing a significant need of medical professionals in the
clinical setting.
Liver disease, which includes hepatitis B and C, liver cancer
and cirrhosis, is a growing global health issue due in part to
rising obesity rates and an aging population. Non-alcoholic fatty
liver disease affects approximately 20% of the global population
[1]. According to the World Health Organization (WHO), total deaths
worldwide from cirrhosis and liver cancer rose by 50 million per
year over the last two decades, and many cases continue to go
undetected [2].
To determine the stage of liver disease and damage, a liver
biopsy is typically performed by extracting a small piece of liver
tissue for microscopic examination. Research suggests that instead
of costly and painful biopsy procedures, ultrasound exams using
shear wave elastography could become routine for assessing liver
disease status and may reduce or avoid the need for conventional
liver biopsies [3].
"Philips aims to provide the tools necessary for assessing and
managing chronic conditions that so many people face, and liver
disease is no exception," said Vitor
Rocha, Ultrasound Business Leader at Philips. "We know that
liver disease is a growing health concern around the globe, and we
are committed to pioneering innovations like ElastQ Imaging to
create our ultimate ultrasound liver solution that offers
exceptional clinical performance, further improving patient
care."
Philips ElastQ Imaging shear wave elastography offers:
- Larger field of view or Region of Interest (ROI) than
competitors
- Color-coded quantitative assessment of tissue stiffness
- Real-time feedback and intelligent analysis
- Quantitative measurements with multiple sample points
In addition to ElastQ Imaging, Philips' ultimate ultrasound
liver solution is comprised of a suite of features that make it a
powerful tool for clinicians, including:
- PureWave transducer technology, delivering superb image
quality and increased penetration in technically difficult-to-image
patients
- Contrast enhanced ultrasound, providing confidence in
liver lesion detection and characterization
- Image fusion and navigation with anatomical
intelligence, increasing the power of real-time ultrasound
exams through multimodality fusion and interventional guidance
Improving the exam experience for both patients and clinicians
is a critical part of Philips' mission to improve the quality of
healthcare with patient-centric solutions. The ultimate ultrasound
liver solution combines a routine ultrasound imaging exam of the
liver anatomy with targeted tissue stiffness values, allowing
clinicians to obtain a baseline assessment without the wait time
associated with tissue biopsy. For patients, the non-invasive
ultrasound exam is more comfortable than a traditional liver biopsy
that requires the use of needles. The exam results from ultrasound
are also available instantly, which can help reduce apprehension
patients may feel in waiting for test results.
"There is a significant population at risk for liver disease
that may not even know it," said Richard G.
Barr, MD, PhD, radiologist, Southwoods Imaging, in
Youngstown, Ohio. "As a
Radiologist, I see every day how important liver assessment is
becoming, and I'm hopeful that this solution will help patients get
the diagnosis, monitoring and treatment they need."
For more information on the ultimate ultrasound liver solution
and ElastQ Imaging, visit www.philips.com/epiq. Philips' ultimate
ultrasound solution will be showcased at the European Congress of
Radiology (ECR) 2017 annual meeting in Vienna, Austria, March 1–5. Follow
@PhilipsLiveFrom and visit www.philips.com/ecr for updates
throughout the Congress.
[1] Sattar N, et al. Non-alcoholic fatty liver disease. BMJ
2014;349:g4596. 34 Dyson J et al. Non-alcoholic fatty liver
disease: a practical approach to treatment. Postgrad Med J. 2015
Feb;91(1072):92-101.
[2] (2013, November 4). Global Burden
of Liver Disease Substantial. Medscape. Retrieved from
http://www.medscape.com/viewarticle/813788#vp_1
[3] Ferraioli G, et al. Point shear wave elastography method for
assessing liver stiffness. World J Gastroenterology. 2014 April
28;20(16):4787-4796.
About Royal
Philips
Royal Philips
(NYSE: PHG, AEX: PHIA) is a leading health technology company
focused on improving people's health and enabling better outcomes
across the health continuum from healthy living and prevention, to
diagnosis, treatment and home care. Philips leverages advanced
technology and deep clinical and consumer insights to deliver
integrated solutions. Headquartered in the Netherlands, the company is a leader in
diagnostic imaging, image-guided therapy, patient monitoring and
health informatics, as well as in consumer health and home care.
Philips' health technology portfolio generated 2016 sales of
EUR 17.4 billion and employs
approximately 71,000 employees with sales and services in more than
100 countries. News about Philips can be found at
www.philips.com/newscenter.

To view the original version on PR Newswire,
visit:http://www.prnewswire.com/news-releases/philips-receives-fda-510k-clearance-to-market-its-elastq-imaging-shear-wave-elastography-for-non-invasive-assessment-of-tissue-stiffness-of-the-liver-300413472.html
SOURCE Royal Philips